Benzodiazepine
| Benzodiazepines |
|---|
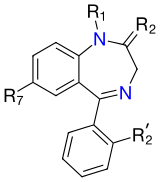 The core structure of benzodiazepines. "R" labels denote common locations of side chains, which give different benzodiazepines their unique properties. |
Benzodiazepines (BZD), sometimes called "benzos", are a class of psychoactive drugs whose core chemical structure is the fusion of a benzene ring and a diazepine ring. The first such drug, chlordiazepoxide (Librium), was discovered accidentally by Leo Sternbach in 1955, and made available in 1960 by Hoffmann–La Roche - which, since 1963, has also marketed the benzodiazepine diazepam (Valium).[1] In 1977 benzodiazepines were globally the most prescribed medications.[2]
Benzodiazepines enhance the effect of the neurotransmitter gamma-aminobutyric acid (GABA) at the GABAA receptor, resulting in sedative, hypnotic (sleep-inducing), anxiolytic (anti-anxiety), anticonvulsant, and muscle relaxant properties. High doses of many shorter-acting benzodiazepines may also cause anterograde amnesia and dissociation.[3] These properties make benzodiazepines useful in treating anxiety, insomnia, agitation, seizures, muscle spasms, alcohol withdrawal and as a premedication for medical or dental procedures.[4] Benzodiazepines are categorized as either short-, intermediate-, or long-acting. Short- and intermediate-acting benzodiazepines are preferred for the treatment of insomnia; longer-acting benzodiazepines are recommended for the treatment of anxiety.[5]
Benzodiazepines are generally viewed as safe and effective for short-term use, although cognitive impairment and paradoxical effects such as aggression or behavioral disinhibition occasionally occur. A minority of people can have paradoxical reactions such as worsened agitation or panic.[6] Long-term use is controversial because of concerns about adverse psychological and physical effects, decreasing effectiveness, and physical dependence and withdrawal.[7][8] As a result of adverse effects associated with the long-term use of benzodiazepines, withdrawal from benzodiazepines, in general, leads to improved physical and mental health.[9][10] The elderly are at an increased risk of suffering from both short- and long-term adverse effects,[9][11] and as a result, all benzodiazepines are listed in the Beers List of inappropriate medications for older adults.[12]
There is controversy concerning the safety of benzodiazepines in pregnancy. While they are not major teratogens, uncertainty remains as to whether they cause cleft palate in a small number of babies and whether neurobehavioural effects occur as a result of prenatal exposure;[13] they are known to cause withdrawal symptoms in the newborn. Benzodiazepines can be taken in overdoses and can cause dangerous deep unconsciousness. However, they are much less toxic than their predecessors, the barbiturates, and death rarely results when a benzodiazepine is the only drug taken; however, when combined with other central nervous system (CNS) depressants such as ethanol and opioids, the potential for toxicity and fatal overdose increases.[14] Benzodiazepines are commonly misused and taken in combination with other drugs of abuse.[15][16][17]
Medical uses
Benzodiazepines possess sedative, hypnotic, anxiolytic, anticonvulsant, muscle relaxant, and amnesic actions,[3][4] which are useful in a variety of indications such as alcohol dependence, seizures, anxiety, panic, agitation, and insomnia. Most are administered orally; however, they can also be given intravenously, intramuscularly, or rectally.[18]:189 In general, benzodiazepines are well-tolerated and are safe and effective drugs in the short term for a wide range of conditions.[19][20] Tolerance can develop to their effects and there is also a risk of dependence, and upon discontinuation a withdrawal syndrome may occur. These factors, combined with other possible secondary effects after prolonged use such as psychomotor, cognitive, or memory impairments, limit their long-term applicability.[21][22] The effects of long-term use or misuse include the tendency to cause or worsen cognitive deficits, depression, and anxiety.[9][11]
Panic disorder
Because of their effectiveness, tolerability, and rapid onset of anxiolytic action, benzodiazepines are frequently used for the treatment of anxiety associated with panic disorder.[23] However, there is disagreement among expert bodies regarding the long-term use of benzodiazepines for panic disorder. The views range from those that hold that benzodiazepines are not effective long-term [24] and that they should be reserved for treatment-resistant cases[25] to that they are as effective in the long term as selective serotonin reuptake inhibitors.[26]
The American Psychiatric Association (APA) guidelines[26] note that, in general, benzodiazepines are well tolerated, and their use for the initial treatment for panic disorder is strongly supported by numerous controlled trials. APA states that there is insufficient evidence to recommend any of the established panic disorder treatments over another. The choice of treatment between benzodiazepines, SSRIs, serotonin–norepinephrine reuptake inhibitors, tricyclic antidepressants, and psychotherapy should be based on the patient's history, preference, and other individual characteristics. Selective serotonin reuptake inhibitors are likely to be the best choice of pharmacotherapy for many patients with panic disorder, but benzodiazepines are also often used, and some studies suggest that these medications are still used with greater frequency than the SSRIs. One advantage of benzodiazepines is that they alleviate the anxiety symptoms much faster than antidepressants, and therefore may be preferred in patients for whom rapid symptom control is critical. However, this advantage is offset by the possibility of developing benzodiazepine dependence. APA does not recommend benzodiazepines for persons with depressive symptoms or a recent history of substance abuse. The APA guidelines state that, in general, pharmacotherapy of panic disorder should be continued for at least a year, and that clinical experience support continuing benzodiazepine treatment to prevent recurrence. Although major concerns about benzodiazepine tolerance and withdrawal have been raised, there is no evidence for significant dose escalation in patients using benzodiazepines long-term. For many such patients stable doses of benzodiazepines retain their efficacy over several years.[26]
Guidelines issued by the UK-based National Institute for Health and Clinical Excellence (NICE), carried out a systematic review using different methodology and came to a different conclusion. They questioned the accuracy of studies that were not placebo-controlled. And, based on the findings of placebo-controlled studies, they do not recommend use of benzodiazepines beyond two to four weeks, as tolerance and physical dependence develop rapidly, with withdrawal symptoms including rebound anxiety occurring after six weeks or more of use.[24][27] Nevertheless, benzodiazepines continue to be prescribed for the long-term treatment of anxiety disorders, although specific antidepressants and psychological therapies are recommended as the first-line treatment options with the anticonvulsant drug pregabalin indicated as a second- or third-line treatment and suitable for long-term use.[28] NICE stated that long-term use of benzodiazepines for panic disorder with or without agoraphobia is an unlicensed indication, does not have long-term efficacy, and is, therefore, not recommended by clinical guidelines. Psychological therapies such as cognitive behavioural therapy are recommended as a first-line therapy for panic disorder; benzodiazepine use has been found to interfere with therapeutic gains from these therapies.[24]
Benzodiazepines are usually administered orally; however, very occasionally lorazepam or diazepam may be given intravenously for the treatment of panic attacks.[18]
Generalized anxiety disorder
Benzodiazepines have robust efficacy in the short-term management of generalized anxiety disorder (GAD), but were not shown to be effective in producing long-term improvement overall.[29] According to National Institute for Health and Clinical Excellence (NICE), benzodiazepines can be used in the immediate management of GAD, if necessary. However, they should not usually be given for longer than 2–4 weeks. The only medications NICE recommends for the longer term management of GAD are antidepressants.[30]
Likewise, Canadian Psychiatric Association (CPA) recommends benzodiazepines alprazolam, bromazepam, lorazepam, and diazepam only as a second-line choice, if the treatment with two different antidepressants was unsuccessful. Although they are second-line agents, benzodiazepines can be used for a limited time to relieve severe anxiety and agitation. CPA guidelines note that after 4–6 weeks the effect of benzodiazepines may decrease to the level of placebo, and that benzodiazepines are less effective than antidepressants in alleviating ruminative worry, the core symptom of GAD. However, in some cases, a prolonged treatment with benzodiazepines as the add-on to an antidepressant may be justified.[31]
A 2015 review found a larger effect with medications than talk therapy.[32] Medications with benefit include serotonin-noradrenaline reuptake inhibitors, benzodiazepines, and selective serotonin reuptake inhibitors.[32]
Insomnia

Benzodiazepines can be useful for short-term treatment of insomnia. Their use beyond 2 to 4 weeks is not recommended due to the risk of dependence. It is preferred that benzodiazepines be taken intermittently and at the lowest effective dose. They improve sleep-related problems by shortening the time spent in bed before falling asleep, prolonging the sleep time, and, in general, reducing wakefulness.[33][34] However, they worsen sleep quality by increasing light sleep and decreasing deep sleep. Other drawbacks of hypnotics, including benzodiazepines, are possible tolerance to their effects, rebound insomnia, and reduced slow-wave sleep and a withdrawal period typified by rebound insomnia and a prolonged period of anxiety and agitation.[35][36]
The list of benzodiazepines approved for the treatment of insomnia is fairly similar among most countries, but which benzodiazepines are officially designated as first-line hypnotics prescribed for the treatment of insomnia can vary distinctly between countries.[34] Longer-acting benzodiazepines such as nitrazepam and diazepam have residual effects that may persist into the next day and are, in general, not recommended.[33]
It is not clear as to whether the new nonbenzodiazepine hypnotics (Z-drugs) are better than the short-acting benzodiazepines. The efficacy of these two groups of medications is similar.[33][36] According to the US Agency for Healthcare Research and Quality, indirect comparison indicates that side-effects from benzodiazepines may be about twice as frequent as from nonbenzodiazepines.[36] Some experts suggest using nonbenzodiazepines preferentially as a first-line long-term treatment of insomnia.[34] However, the UK National Institute for Health and Clinical Excellence did not find any convincing evidence in favor of Z-drugs. NICE review pointed out that short-acting Z-drugs were inappropriately compared in clinical trials with long-acting benzodiazepines. There have been no trials comparing short-acting Z-drugs with appropriate doses of short-acting benzodiazepines. Based on this, NICE recommended choosing the hypnotic based on cost and the patient's preference.[33]
Older adults should not use benzodiazepines to treat insomnia unless other treatments have failed to be effective.[37] When benzodiazepines are used, patients, their caretakers, and their physician should discuss the increased risk of harms, including evidence which shows twice the incidence of traffic collisions among driving patients as well as falls and hip fracture for all older patients.[37]
Seizures
Prolonged convulsive epileptic seizures are a medical emergency that can usually be dealt with effectively by administering fast-acting benzodiazepines, which are potent anticonvulsants. In a hospital environment, intravenous clonazepam, lorazepam, and diazepam are first-line choices, clonazepam due to its stronger and more potent anticonvulsant action, diazepam due to its faster onset and lorazepam for its longer duration of action. In the community, intravenous administration is not practical and so rectal diazepam or (more recently) buccal midazolam are used, with a preference for midazolam as its administration is easier and more socially acceptable.[38][39]
When benzodiazepines were first introduced, they were enthusiastically adopted for treating all forms of epilepsy. However, drowsiness and tolerance become problems with continued use and none are now considered first-line choices for long-term epilepsy therapy.[40] Clobazam is widely used by specialist epilepsy clinics worldwide and clonazepam is popular in the Netherlands, Belgium and France.[40] Clobazam was approved for use in the United States in 2011. In the UK, both clobazam and clonazepam are second-line choices for treating many forms of epilepsy.[41] Clobazam also has a useful role for very short-term seizure prophylaxis and in catamenial epilepsy.[40] Discontinuation after long-term use in epilepsy requires additional caution because of the risks of rebound seizures. Therefore, the dose is slowly tapered over a period of up to six months or longer.[39]
Alcohol withdrawal
Chlordiazepoxide is the most commonly used benzodiazepine for alcohol detoxification,[42] but diazepam may be used as an alternative. Both are used in the detoxification of individuals who are motivated to stop drinking, and are prescribed for a short period of time to reduce the risks of developing tolerance and dependence to the benzodiazepine medication itself.[18]:275 The benzodiazepines with a longer half-life make detoxification more tolerable, and dangerous (and potentially lethal) alcohol withdrawal effects are less likely to occur. On the other hand, short-acting benzodiazepines may lead to breakthrough seizures, and are, therefore, not recommended for detoxification in an outpatient setting. Oxazepam and lorazepam are often used in patients at risk of drug accumulation, in particular, the elderly and those with cirrhosis, because they are metabolized differently from other benzodiazepines, through conjugation.[43][44]
Benzodiazepines are the preferred choice in the management of alcohol withdrawal syndrome, in particular, for the prevention and treatment of the dangerous complication of seizures and in subduing severe delirium.[45] Lorazepam is the only benzodiazepine with predictable intramuscular absorption and it is the most effective in preventing and controlling acute seizures.[46]
Anxiety
Benzodiazepines are sometimes used in the treatment of acute anxiety, as they bring about rapid and marked or moderate relief of symptoms in most individuals;[24] however, they are not recommended beyond 2–4 weeks of use due to risks of tolerance and dependence and a lack of long-term effectiveness. As for insomnia, they may also be used on an irregular/"as-needed" basis, such as in cases where said anxiety is at its worst. Compared to other pharmacological treatments, benzodiazepines are twice as likely to lead to a relapse of the underlying condition upon discontinuation. Psychological therapies and other pharmacological therapies are recommended for the long-term treatment of generalized anxiety disorder. Antidepressants have higher remission rates and are, in general, safe and effective in the short and long term.[24]
Other indications
Benzodiazepines are often prescribed for a wide range of conditions:
- They can be very useful in intensive care to sedate patients receiving mechanical ventilation or those in extreme distress. Caution is exercised in this situation due to the occasional occurrence of respiratory depression, and it is recommended that benzodiazepine overdose treatment facilities should be available.[47]
- Benzodiazepines are effective as medication given a couple of hours before surgery to relieve anxiety. They also produce amnesia, which can be useful, as patients will not be able to remember any unpleasantness from the procedure.[48] They are also used in patients with dental phobia as well as some ophthalmic procedures like refractive surgery; although such use is controversial and only recommended for those who are very anxious.[49] Midazolam is the most commonly prescribed for this use because of its strong sedative actions and fast recovery time, as well as its water solubility, which reduces pain upon injection. Diazepam and lorazepam are sometimes used. Lorazepam has particularly marked amnesic properties that may make it more effective when amnesia is the desired effect.[18]:693
- Benzodiazepines are well known for their strong muscle-relaxing properties and can be useful in the treatment of muscle spasms,[18]:577–578 although tolerance often develops to their muscle relaxant effects.[9] Baclofen[50] or tizanidine are sometimes used as an alternative to benzodiazepines. Tizanidine has been found to have superior tolerability compared to diazepam and baclofen.[51]
- Benzodiazepines are also used to treat the acute panic caused by hallucinogen intoxication.[52] Benzodiazepines are also used to calm the acutely agitated individual and can, if required, be given via an intramuscular injection.[53] They can sometimes be effective in the short-term treatment of psychiatric emergencies such as acute psychosis as in schizophrenia or mania, bringing about rapid tranquillization and sedation until the effects of lithium or neuroleptics (antipsychotics) take effect. Lorazepam is most commonly used but clonazepam is sometimes prescribed for acute psychosis or mania;[54][55] their long-term use is not recommended due to risks of dependence.[18]:204
- Clonazepam, a benzodiazepine is used to treat many forms of parasomnia.[56] Rapid eye movement behavior disorder responds well to low doses of clonazepam.[57][58] Restless legs syndrome can be treated using clonazepam as a third line treatment option as the use of clonazepam is still investigational.[59][60]
- Benzodiazepines are sometimes used for obsessive–compulsive disorder (OCD), although they are generally believed to be ineffective for this indication; effectiveness was, however, found in one small study.[61] Benzodiazepines can be considered as a treatment option in treatment resistant cases.[62]
- Antipsychotics are generally a first-line treatment for delirium; however, when delirium is caused by alcohol or sedative hypnotic withdrawal, benzodiazepines are a first-line treatment.[63]
- There is some evidence that low doses of benzodiazepines reduce adverse effects of electroconvulsive therapy.[64]
Contraindications
Because of their muscle relaxant action, benzodiazepines may cause respiratory depression in susceptible individuals. For that reason, they are contraindicated in people with myasthenia gravis, sleep apnea, bronchitis, and COPD.[65][66] Caution is required when benzodiazepines are used in people with personality disorders or intellectual disability because of frequent paradoxical reactions.[65][66] In major depression, they may precipitate suicidal tendencies[67] and are sometimes used for suicidal overdoses.[66] Individuals with a history of alcohol, opioid and barbiturate abuse should avoid benzodiazepines, as there is a risk of life-threatening interactions with these drugs.[68]
Pregnancy
In the United States, the Food and Drug Administration has categorized benzodiazepines into either category D or X meaning potential for harm in the unborn has been demonstrated.[69]
Exposure to benzodiazepines during pregnancy has been associated with a slightly increased (from 0.06 to 0.07%) risk of cleft palate in newborns, a controversial conclusion as some studies find no association between benzodiazepines and cleft palate. Their use by expectant mothers shortly before the delivery may result in a floppy infant syndrome, with the newborns suffering from hypotonia, hypothermia, lethargy, and breathing and feeding difficulties.[13][70] Cases of neonatal withdrawal syndrome have been described in infants chronically exposed to benzodiazepines in utero. This syndrome may be hard to recognize, as it starts several days after delivery, for example, as late as 21 day for chlordiazepoxide. The symptoms include tremors, hypertonia, hyperreflexia, hyperactivity, and vomiting and may last for up to three to six months.[13][71] Tapering down the dose during pregnancy may lessen its severity. If used in pregnancy, those benzodiazepines with a better and longer safety record, such as diazepam or chlordiazepoxide, are recommended over potentially more harmful benzodiazepines, such as temazepam[72] or triazolam. Using the lowest effective dose for the shortest period of time minimizes the risks to the unborn child.[73]
Elderly
The benefits of benzodiazepines are least and the risks are greatest in the elderly.[74][75] The elderly are at an increased risk of dependence and are more sensitive to the adverse effects such as memory problems, daytime sedation, impaired motor coordination, and increased risk of motor vehicle accidents and falls,[76] and an increased risk of hip fractures.[77] The long-term effects of benzodiazepines and benzodiazepine dependence in the elderly can resemble dementia, depression, or anxiety syndromes, and progressively worsens over time. Adverse effects on cognition can be mistaken for the effects of old age. The benefits of withdrawal include improved cognition, alertness, mobility, reduced risk incontinence, and a reduced risk of falls and fractures. The success of gradual-tapering benzodiazepines is as great in the elderly as in younger people. Benzodiazepines should be prescribed to the elderly only with caution and only for a short period at low doses.[78][79] Short to intermediate-acting benzodiazepines are preferred in the elderly such as oxazepam and temazepam. The high potency benzodiazepines alprazolam and triazolam and long-acting benzodiazepines are not recommended in the elderly due to increased adverse effects. Nonbenzodiazepines such as zaleplon and zolpidem and low doses of sedating antidepressants are sometimes used as alternatives to benzodiazepines.[79][80]
Long-term use of benzodiazepines has been associated with increased risk of cognitive impairment, but its relationship with dementia remains inconclusive.[81] The association of a past history of benzodiazepine use and cognitive decline is unclear, with some studies reporting a lower risk of cognitive decline in former users, some finding no association and some indicating an increased risk of cognitive decline.[82]
Benzodiazepines are sometimes prescribed to treat behavioral symptoms of dementia. However, like antidepressants, they have little evidence of effectiveness, although antipsychotics have shown some benefit.[83][84] Cognitive impairing effects of benzodiazepines that occur frequently in the elderly can also worsen dementia.[85]
Adverse effects
The most common side-effects of benzodiazepines are related to their sedating and muscle-relaxing action. They include drowsiness, dizziness, and decreased alertness and concentration. Lack of coordination may result in falls and injuries, in particular, in the elderly.[65][86][87] Another result is impairment of driving skills and increased likelihood of road traffic accidents.[88][89] Decreased libido and erection problems are a common side effect. Depression and disinhibition may emerge. Hypotension and suppressed breathing (hypoventilation) may be encountered with intravenous use.[65][86] Less common side effects include nausea and changes in appetite, blurred vision, confusion, euphoria, depersonalization and nightmares. Cases of liver toxicity have been described but are very rare.[18]:183–189[90]
The long-term effects of benzodiazepine use can include cognitive impairment as well as affective and behavioural problems. Feelings of turmoil, difficulty in thinking constructively, loss of sex-drive, agoraphobia and social phobia, increasing anxiety and depression, loss of interest in leisure pursuits and interests, and an inability to experience or express feelings can also occur. Not everyone, however, experiences problems with long-term use.[10][91] Additionally an altered perception of self, environment and relationships may occur.[92]
Cognitive effects
The short-term use of benzodiazepines adversely affects multiple areas of cognition, the most notable one being that it interferes with the formation and consolidation of memories of new material and may induce complete anterograde amnesia.[65] However, researchers hold contrary opinions regarding the effects of long-term administration. One view is that many of the short-term effects continue into the long-term and may even worsen, and are not resolved after stopping benzodiazepine usage. Another view maintains that cognitive deficits in chronic benzodiazepine users occur only for a short period after the dose, or that the anxiety disorder is the cause of these deficits.
While the definitive studies are lacking, the former view received support from a 2004 meta-analysis of 13 small studies.[92][93] This meta-analysis found that long-term use of benzodiazepines was associated with moderate to large adverse effects on all areas of cognition, with visuospatial memory being the most commonly detected impairment. Some of the other impairments reported were decreased IQ, visiomotor coordination, information processing, verbal learning and concentration. The authors of the meta-analysis[93] and a later reviewer[92] noted that the applicability of this meta-analysis is limited because the subjects were taken mostly from withdrawal clinics; the coexisting drug, alcohol use, and psychiatric disorders were not defined; and several of the included studies conducted the cognitive measurements during the withdrawal period.
Paradoxical effects
Paradoxical reactions, such as increased seizures in epileptics,[94] aggression, violence, impulsivity, irritability and suicidal behavior sometimes occur. These reactions have been explained as consequences of disinhibition and the subsequent loss of control over socially unacceptable behavior. Paradoxical reactions are rare in the general population, with an incidence rate below 1% and similar to placebo.[6][95] However, they occur with greater frequency in recreational abusers, individuals with borderline personality disorder, children, and patients on high-dosage regimes.[96][97] In these groups, impulse control problems are perhaps the most important risk factor for disinhibition; learning disabilities and neurological disorders are also significant risks. Most reports of disinhibition involve high doses of high-potency benzodiazepines.[95] Paradoxical effects may also appear after chronic use of benzodiazepines.[98]
Long-term worsening of psychiatric symptoms
While benzodizapines may have short-term benefits for anxiety, sleep and agitation in some patients, long-term (i.e., greater than 2–4 weeks) use can result in a worsening of the very symptoms the medications are meant to treat.[99] Potential explanations include exacerbating cognitive problems that are already common in anxiety disorders, causing or worsening depression and suicidality,[100][101] disrupting sleep architecture by inhibiting deep stage sleep,[102] withdrawal symptoms or rebound symptoms in between doses mimicking or exacerbating underlying anxiety or sleep disorders,[100][101] inhibiting the benefits of psychotherapy by inhibiting memory consolidation and reducing fear extinction,[103][104][105] and reducing coping with trauma/stress and increasing vulnerability to future stress.[99][106] Anxiety, insomnia and irritability may be temporarily exacerbated during withdrawal, but psychiatric symptoms after discontinuation are usually less than even while taking benzodiazepines.[100][107] Fortunately, for those with benzodiazepine-induced problems, functioning significantly improves within 1 year of discontinuation.[100][108]
Reinforcement disorders
Tolerance
The main problem of the chronic use of benzodiazepines is the development of tolerance and dependence. Tolerance manifests itself as diminished pharmacological effect and develops relatively quickly to the sedative, hypnotic, anticonvulsant, and muscle relaxant actions of benzodiazepines. Tolerance to anti-anxiety effects develops more slowly with little evidence of continued effectiveness beyond four to six months of continued use.[9] In general, tolerance to the amnesic effects does not occur.[85] However, controversy exists as to tolerance to the anxiolytic effects with some evidence that benzodiazepines retain efficacy[109] and opposing evidence from a systematic review of the literature that tolerance frequently occurs[19][24] and some evidence that anxiety may worsen with long-term use.[9] The question of tolerance to the amnesic effects of benzodiazepines is, likewise, unclear.[110] Some evidence suggests that partial tolerance does develop, and that, "memory impairment is limited to a narrow window within 90 minutes after each dose".[111]
A major disadvantage of benzodiazepines that tolerance to therapeutic effects develops relatively quickly while many adverse effects persist. Tolerance develops to hypnotic and myorelexant effects within days to weeks, and to anticonvulsant and anxiolytic effects within weeks to months.[99][100] Therefore, benzodiazepines are unlikely to be effective long-term treatments for sleep and anxiety. While BZD therapeutic effects disappear with tolerance, depression and impulsivity with high suicidal risk commonly persist.[100] Several studies have confirmed that long-term benzodiazepines are not significantly different from placebo for sleep[112][113][114] or anxiety.[100][115][116][117] This may explain why patients commonly increase doses over time and many eventually take more than one type of benzodiazepine after the first loses effectiveness.[102][118][119] Additionally, because tolerance to benzodiazepine sedating effects develops more quickly than does tolerance to brainstem depressant effects, those taking more benzodiazepines to achieve desired effects may suffer sudden respiratory depression, hypotension or death.[120] Most patients with anxiety disorders and PTSD have symptoms which persist for at least several months,[120] making tolerance to therapeutic effects a distinct problem for them and necessitating the need for more effective long-term treatment (e.g., psychotherapy, serotonergic antidepressants).
Withdrawal symptoms and management
Discontinuation of benzodiazepines or abrupt reduction of the dose, even after a relatively short course of treatment (three to four weeks), may result in two groups of symptoms — rebound and withdrawal. Rebound symptoms are the return of the symptoms for which the patient was treated but worse than before. Withdrawal symptoms are the new symptoms that occur when the benzodiazepine is stopped. They are the main sign of physical dependence.[111]
The most frequent symptoms of withdrawal from benzodiazepines are insomnia, gastric problems, tremors, agitation, fearfulness, and muscle spasms.[111] The less frequent effects are irritability, sweating, depersonalization, derealization, hypersensitivity to stimuli, depression, suicidal behavior, psychosis, seizures, and delirium tremens.[121] Severe symptoms usually occur as a result of abrupt or over-rapid withdrawal. Abrupt withdrawal can be dangerous, therefore a gradual reduction regimen is recommended.[8]
Symptoms may also occur during a gradual dosage reduction, but are typically less severe and may persist as part of a protracted withdrawal syndrome for months after cessation of benzodiazepines.[122] Approximately 10% of patients will experience a notable protracted withdrawal syndrome, which can persist for many months or in some cases a year or longer. Protracted symptoms tend to resemble those seen during the first couple of months of withdrawal but usually are of a sub-acute level of severity. Such symptoms do gradually lessen over time, eventually disappearing altogether.[123]
Benzodiazepines have a reputation with patients and doctors for causing a severe and traumatic withdrawal; however, this is in large part due to the withdrawal process being poorly managed. Over-rapid withdrawal from benzodiazepines increases the severity of the withdrawal syndrome and increases the failure rate. A slow and gradual withdrawal customised to the individual and, if indicated, psychological support is the most effective way of managing the withdrawal. Opinion as to the time needed to complete withdrawal ranges from four weeks to several years. A goal of less than six months has been suggested,[8] but due to factors such as dosage and type of benzodiazepine, reasons for prescription, lifestyle, personality, environmental stresses, and amount of available support, a year or more may be needed to withdraw.[9][18]:183–184
Withdrawal is best managed by transferring the physically dependent patient to an equivalent dose of diazepam because it has the longest half-life of all of the benzodiazepines, is metabolised into long-acting active metabolites and is available in low-potency tablets, which can be quartered for smaller doses.[124] A further benefit is that it is available in liquid form, which allows for even smaller reductions.[8] Chlordiazepoxide, which also has a long half-life and long-acting active metabolites, can be used as an alternative.[124][125]
Nonbenzodiazepines are contraindicated during benzodiazepine withdrawal as they are cross tolerant with benzodiazepines and can induce dependence.[9] Alcohol is also cross tolerant with benzodiazepines and more toxic and thus caution is needed to avoid replacing one dependence with another.[124] During withdrawal, fluoroquinolone-based antibiotics are best avoided if possible; they displace benzodiazepines from their binding site and reduce GABA function and, thus, may aggravate withdrawal symptoms.[126] Antipsychotics are not recommended for benzodiazepine withdrawal (or other CNS depressant withdrawal states) especially clozapine, olanzapine or low potency phenothiazines e.g. chlorpromazine as they lower the seizure threshold and can worsen withdrawal effects; if used extreme caution is required.[127]
Withdrawal from long term benzodiazepines is beneficial for most individuals.[98] Withdrawal of benzodiazepines from long-term users, in general, leads to improved physical and mental health particularly in the elderly; although some long term users report continued benefit from taking benzodiazepines, this may be the result of suppression of withdrawal effects.[9][10]
Overdose

Although benzodiazepines are much safer in overdose than their predecessors, the barbiturates, they can still cause problems in overdose.[14] Taken alone, they rarely cause severe complications in overdose;[129] statistics in England showed that benzodiazepines were responsible for 3.8% of all deaths by poisoning from a single drug.[15] However, combining these drugs with alcohol, opiates or tricyclic antidepressants markedly raises the toxicity.[16][130][131] The elderly are more sensitive to the side effects of benzodiazepines, and poisoning may even occur from their long-term use.[132] The various benzodiazepines differ in their toxicity; temazepam appears to be most toxic in overdose and when used with other drugs.[133][134] The symptoms of a benzodiazepine overdose may include; drowsiness, slurred speech, nystagmus, hypotension, ataxia, coma, respiratory depression, and cardiorespiratory arrest.[131]
A reversal agent for benzodiazepines exists, flumazenil (Anexate). Its use as an antidote is not routinely recommended because of the high risk of resedation and seizures.[135] In a double-blind, placebo-controlled trial of 326 patients, 4 patients suffered serious adverse events and 61% became resedated following the use of flumazenil.[136] Numerous contraindications to its use exist. It is contraindicated in patients with a history of long-term use of benzodiazepines, those having ingested a substance that lowers the seizure threshold or may cause an arrhythmia, and in those with abnormal vital signs.[137] One study found that only 10% of the patient population presenting with a benzodiazepine overdose are suitable candidates for treatment with flumazenil.[138]
Interactions
Individual benzodiazepines may have different interactions with certain drugs. Depending on their metabolism pathway, benzodiazepines can be divided roughly into two groups. The largest group consists of those that are metabolized by cytochrome P450 (CYP450) enzymes and possess significant potential for interactions with other drugs. The other group comprises those that are metabolized through glucuronidation, such as lorazepam, oxazepam, and temazepam, and, in general, have few drug interactions.[66]
Many drugs, including oral contraceptives, some antibiotics, antidepressants, and antifungal agents, inhibit cytochrome enzymes in the liver. They reduce the rate of elimination of the benzodiazepines that are metabolized by CYP450, leading to possibly excessive drug accumulation and increased side-effects. In contrast, drugs that induce cytochrome P450 enzymes, such as St John's wort, the antibiotic rifampicin, and the anticonvulsants carbamazepine and phenytoin, accelerate elimination of many benzodiazepines and decrease their action.[68][139] Taking benzodiazepines with alcohol, opioids and other central nervous system depressants potentiates their action. This often results in increased sedation, impaired motor coordination, suppressed breathing, and other adverse effects that have potential to be lethal.[68][139] Antacids can slow down absorption of some benzodiazepines; however, this effect is marginal and inconsistent.[68]
Pharmacology
Mechanism of action

Benzodiazepines work by increasing the efficiency of a natural brain chemical, GABA, to decrease the excitability of neurons. This reduces the communication between neurons and, therefore, has a calming effect on many of the functions of the brain.
GABA controls the excitability of neurons by binding to the GABAA receptor.[140] The GABAA receptor is a protein complex located in the synapses of neurons. All GABAA receptors contain an ion channel that conducts chloride ions across neuronal cell membranes and two binding sites for the neurotransmitter gamma-aminobutyric acid (GABA), while a subset of GABAA receptor complexes also contain a single binding site for benzodiazepines. Binding of benzodiazepines to this receptor complex does not alter binding of GABA. Unlike other positive allosteric modulators that increases ligand binding, benzodiazepine binding acts as a positive allosteric modulator by increasing the total conduction of chloride ions across the neuronal cell membrane when GABA is already bound to its receptor. This increased chloride ion influx hyperpolarizes the neuron's membrane potential. As a result, the difference between resting potential and threshold potential is increased and firing is less likely. Different GABAA receptor subtypes have varying distributions within different regions of the brain and, therefore, control distinct neuronal circuits. Hence, activation of different GABAA receptor subtypes by benzodiazepines may result in distinct pharmacological actions.[141] In terms of the mechanism of action of benzodiazepines, their similarities are too great to separate them into individual categories such as anxiolytic or hypnotic. For example, a hypnotic administered in low doses will produce anxiety-relieving effects, whereas a benzodiazepine marketed as an anti-anxiety drug will at higher doses induce sleep.[142]
The subset of GABAA receptors that also bind benzodiazepines are referred to as benzodiazepine receptors (BzR). The GABAA receptor is a heteromer composed of five subunits, the most common ones being two αs, two βs, and one γ (α2β2γ). For each subunit, many subtypes exist (α1–6, β1–3, and γ1–3). GABAA receptors that are made up of different combinations of subunit subtypes have different properties, different distributions in the brain and different activities relative to pharmacological and clinical effects.[143] Benzodiazepines bind at the interface of the α and γ subunits on the GABAA receptor. Binding also requires that alpha subunits contain a histidine amino acid residue, (i.e., α1, α2, α3, and α5 containing GABAA receptors). For this reason, benzodiazepines show no affinity for GABAA receptors containing α4 and α6 subunits with an arginine instead of a histidine residue.[144] Once bound to the benzodiazepine receptor, the benzodiazepine ligand locks the benzodiazepine receptor into a conformation in which it has a greater affinity for the GABA neurotransmitter. This increases the frequency of the opening of the associated chloride ion channel and hyperpolarizes the membrane of the associated neuron. The inhibitory effect of the available GABA is potentiated, leading to sedatory and anxiolytic effects. For instance, those ligands with high activity at the α1 are associated with stronger hypnotic effects, whereas those with higher affinity for GABAA receptors containing α2 and/or α3 subunits have good anti-anxiety activity.[145]
The benzodiazepine class of drugs also interact with peripheral benzodiazepine receptors. Peripheral benzodiazepine receptors are present in peripheral nervous system tissues, glial cells, and to a lesser extent the central nervous system.[146] These peripheral receptors are not structurally related or coupled to GABAA receptors. They modulate the immune system and are involved in the body response to injury.[147][148] Benzodiazepines also function as weak adenosine reuptake inhibitors. It has been suggested that some of their anticonvulsant, anxiolytic, and muscle relaxant effects may be in part mediated by this action.[149]
Pharmacokinetics
| Benzodiazepine[150] | Half-life (range, h) | Speed of Onset |
|---|---|---|
| Alprazolam | 12–15 | Intermediate |
| Chlordiazepoxide | 10–30 | Intermediate |
| Clonazepam | 10–50 | Slow |
| Diazepam | 20–80 | Fast |
| Lorazepam | 10–20 | Intermediate |
| Oxazepam | 5–10 | Slow |
| Prazepam | 50–200 | Slow |
A benzodiazepine can be placed into one of three groups by its elimination half-life, or time it takes for the body to eliminate half of the dose.[151] Some benzodiazepines have long-acting active metabolites, such as diazepam and chlordiazepoxide, which are metabolised into desmethyldiazepam. Desmethyldiazepam has a half-life of 36–200 hours, and flurazepam, with the main active metabolite of desalkylflurazepam, with a half-life of 40–250 hours. These long-acting metabolites are partial agonists.[5][124]
- Short-acting compounds have a median half-life of 1–12 hours. They have few residual effects if taken before bedtime, rebound insomnia may occur upon discontinuation, and they might cause daytime withdrawal symptoms such as next day rebound anxiety with prolonged usage. Examples are brotizolam, midazolam, and triazolam.
- Intermediate-acting compounds have a median half-life of 12–40 hours. They may have some residual effects in the first half of the day if used as a hypnotic. Rebound insomnia, however, is more common upon discontinuation of intermediate-acting benzodiazepines than longer-acting benzodiazepines. Examples are alprazolam, estazolam, flunitrazepam, clonazepam, lormetazepam, lorazepam, nitrazepam, and temazepam.
- Long-acting compounds have a half-life of 40–250 hours. They have a risk of accumulation in the elderly and in individuals with severely impaired liver function, but they have a reduced severity of rebound effects and withdrawal. Examples are diazepam, clorazepate, chlordiazepoxide, and flurazepam.
Physical and chemical properties
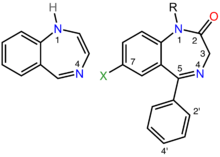

Benzodiazepines share a similar chemical structure, and their effects in humans are mainly produced by the allosteric modification of a specific kind of neurotransmitter receptor, the GABAA receptor, which increases the overall conductance of these inhibitory channels; this results in the various therapeutic effects as well as adverse effects of benzodiazepines.[140] Other less important mechanisms of action are also known.[147][149]
Chemistry
The term benzodiazepine is the chemical name for the heterocyclic ring system (see figure to the right), which is a fusion between the benzene and diazepine ring systems.[153] Under Hantzsch–Widman nomenclature, a diazepine is a heterocycle with two nitrogen atoms, five carbon atom and the maximum possible number of cumulative double bonds. The "benzo" prefix indicates the benzene ring fused onto the diazepine ring.[153]
Benzodiazepine drugs are substituted 1,4-benzodiazepines, although the chemical term can refer to many other compounds that do not have useful pharmacological properties. Different benzodiazepine drugs have different side groups attached to this central structure. The different side groups affect the binding of the molecule to the GABAA receptor and so modulate the pharmacological properties.[140] Many of the pharmacologically active "classical" benzodiazepine drugs contain the 5-phenyl-1H-benzo[e] [1,4]diazepin-2(3H)-one substructure (see figure to the right).[154] Benzodiazepines have been found to mimic protein reverse turns structurally which enable them with their biological activity in many cases.[155][156]
Nonbenzodiazepines also bind to the benzodiazepine binding site on the GABAA receptor and possess similar pharmacological properties. While the nonbenzodiazepines are by definition structurally unrelated to the benzodiazepines, both classes of drugs possess a common pharmacophore (see figure to the lower-right), which explains their binding to a common receptor site.[152]
Common types
- 2-keto compounds:
- clorazepate, diazepam, flurazepam, halazepam, prazepam, and others.[157][158]
- 3-hydroxy compounds:
- 7-nitro compounds:
- Triazolo compounds:
- Imidazo compounds
History
The first benzodiazepine, chlordiazepoxide (Librium), was synthesized in 1955 by Leo Sternbach while working at Hoffmann–La Roche on the development of tranquilizers. The pharmacological properties of the compounds prepared initially were disappointing, and Sternbach abandoned the project. Two years later, in April 1957, co-worker Earl Reeder noticed a "nicely crystalline" compound left over from the discontinued project while spring-cleaning in the lab. This compound, later named chlordiazepoxide, had not been tested in 1955 because of Sternbach's focus on other issues. Expecting the pharmacology results to be negative and hoping to publish the chemistry-related findings, researchers submitted it for a standard battery of animal tests. However, the compound showed very strong sedative, anticonvulsant, and muscle relaxant effects. These impressive clinical findings led to its speedy introduction throughout the world in 1960 under the brand name Librium.[159][160] Following chlordiazepoxide, diazepam marketed by Hoffmann–La Roche under the brand name Valium in 1963, and for a while the two were the most commercially successful drugs. The introduction of benzodiazepines led to a decrease in the prescription of barbiturates, and by the 1970s they had largely replaced the older drugs for sedative and hypnotic uses.[1]
The new group of drugs was initially greeted with optimism by the medical profession, but gradually concerns arose; in particular, the risk of dependence became evident in the 1980s. Benzodiazepines have a unique history in that they were responsible for the largest-ever class-action lawsuit against drug manufacturers in the United Kingdom, involving 14,000 patients and 1,800 law firms that alleged the manufacturers knew of the dependence potential but intentionally withheld this information from doctors. At the same time, 117 general practitioners and 50 health authorities were sued by patients to recover damages for the harmful effects of dependence and withdrawal. This led some doctors to require a signed consent form from their patients and to recommend that all patients be adequately warned of the risks of dependence and withdrawal before starting treatment with benzodiazepines.[161] The court case against the drug manufacturers never reached a verdict; legal aid had been withdrawn and there were allegations that the consultant psychiatrists, the expert witnesses, had a conflict of interest. This litigation led to changes in the British law, making class action lawsuits more difficult.[162]
Although antidepressants with anxiolytic properties have been introduced, and there is increasing awareness of the adverse effects of benzodiazepines, prescriptions for short-term anxiety relief have not significantly dropped.[7] For treatment of insomnia, benzodiazepines are now less popular than nonbenzodiazepines, which include zolpidem, zaleplon and eszopiclone.[163] Nonbenzodiazepines are molecularly distinct, but nonetheless, they work on the same benzodiazepine receptors and produce similar sedative effects.[164]
Society and culture
Legal status
In the United States, benzodiazepines are Schedule IV drugs under the Federal Controlled Substances Act, even when not on the market (for example, nitrazepam and bromazepam). Flunitrazepam is subject to more stringent regulations in certain states and temazepam prescriptions require specially coded pads in certain states.
In Canada, possession of benzodiazepines is legal for personal use. All benzodiazepines are categorized as Schedule IV substances under the Controlled Drugs and Substances Act.[165]
In the United Kingdom, the benzodiazepines are schedule 4 controlled drugs, except for flunitrazepam, temazepam and midazolam, which are schedule 3 controlled drugs and carry stronger penalties for possession and trafficking.[166][167]
In the Netherlands, since October 1993, benzodiazepines, including formulations containing less than 20 mg of temazepam, are all placed on List 2 of the Opium Law. A prescription is needed for possession of all benzodiazepines. Temazepam formulations containing 20 mg or greater of the drug are placed on List 1, thus requiring prescriptions to be written in the List 1 format.[168]
In East Asia and Southeast Asia, temazepam and nimetazepam are often heavily controlled and restricted. In certain countries, triazolam, flunitrazepam, flutoprazepam and midazolam are also restricted or controlled to certain degrees. In Hong Kong, all benzodiazepines are regulated under Schedule 1 of Hong Kong's Chapter 134 Dangerous Drugs Ordinance.[169] Previously only brotizolam, flunitrazepam and triazolam were classed as dangerous drugs.[170]
Internationally, benzodiazepines are categorized as Schedule IV controlled drugs, apart from flunitrazepam which is a Schedule III drug under the Convention on Psychotropic Substances.[171]
Recreational use

Benzodiazepines are considered to be major drugs of abuse.[17] Benzodiazepine abuse is mostly limited to individuals who abuse other drugs, i.e., poly-drug abusers. On the international scene, benzodiazepines are categorized as Schedule IV controlled drugs by the INCB, apart from flunitrazepam which is a Schedule III drug under the Convention on Psychotropic Substances.[172] Some variation in drug scheduling exists in individual countries; for example, in the United Kingdom, midazolam and temazepam are Schedule III controlled drugs. British law requires temazepam (but not midazolam) to be stored in safe custody. Safe custody requirements ensures that pharmacists and doctors holding stock of temazepam must store it in securely fixed double-locked steel safety cabinets and maintain a written register, which must be bound and contain separate entries for temazepam and must be written in ink with no use of correction fluid (although a written register is not required for temazepam in the United Kingdom). Disposal of expired stock must be witnessed by a designated inspector (either a local drug-enforcement police officer or official from health authority).[173][174] Benzodiazepine abuse ranges from occasional binges on large doses, to chronic and compulsive drug abuse of high doses.[175]
Benzodiazepines are used recreationally and by problematic drug misusers. Mortality is higher among poly-drug misusers that also use benzodiazepines. Heavy alcohol use also increases mortality among poly-drug users.[15] Dependence and tolerance, often coupled with dosage escalation, to benzodiazepines can develop rapidly among drug misusers; withdrawal syndrome may appear after as little as three weeks of continuous use. Long-term use has the potential to cause both physical and psychological dependence and severe withdrawal symptoms such as depression, anxiety (often to the point of panic attacks), and agoraphobia.[11] Benzodiazepines and, in particular, temazepam are sometimes used intravenously, which, if done incorrectly or in an unsterile manner, can lead to medical complications including abscesses, cellulitis, thrombophlebitis, arterial puncture, deep vein thrombosis, and gangrene. Sharing syringes and needles for this purpose also brings up the possibility of transmission of hepatitis, HIV, and other diseases. Benzodiazepines are also misused intranasally, which may have additional health consequences. Once benzodiazepine dependence has been established, a clinician usually converts the patient to an equivalent dose of diazepam before beginning a gradual reduction program.[176]
A 1999–2005 Australian police survey of detainees reported preliminary findings that self-reported users of benzodiazepines were less likely than non-user detainees to work full-time and more likely to receive government benefits, use methamphetamine or heroin, and be arrested or imprisoned.[177] Benzodiazepines are sometimes used for criminal purposes; they serve to incapacitate a victim in cases of drug assisted rape or robbery.[178]
Overall, anecdotal evidence suggests that temazepam may be the most psychologically habit-forming (addictive) benzodiazepine. Temazepam abuse reached epidemic proportions in some parts of the world, in particular, in Europe and Australia, and is a major drug of abuse in many Southeast Asian countries. This led authorities of various countries to place temazepam under a more restrictive legal status. Some countries, such as Sweden, banned the drug outright.[179] Temazepam also has certain pharmacokinetic properties of absorption, distribution, elimination, and clearance that make it more apt to abuse compared to many other benzodiazepines.[180][181]
Veterinary use
Benzodiazepines are used in veterinary practice in the treatment of various disorders and conditions. As in humans, they are used in the first-line management of seizures, status epilepticus, and tetanus, and as maintenance therapy in epilepsy (in particular, in cats).[182][183][184] They are widely used in small and large animals (including horses, swine, cattle and exotic and wild animals) for their anxiolytic and sedative effects, as pre-medication before surgery, for induction of anesthesia and as adjuncts to anesthesia.[182][185]
References
- 1 2 Shorter E (2005). "Benzodiazepines". A Historical Dictionary of Psychiatry. Oxford University Press. pp. 41–2. ISBN 0-19-517668-5.
- ↑ Treating Alcohol and Drug Problems in Psychotherapy Practice Doing What Works. New York: Guilford Publications. 2011. p. 47. ISBN 9781462504381.
- 1 2 Page C, Michael C, Sutter M, Walker M, Hoffman BB (2002). Integrated Pharmacology (2nd ed.). C.V. Mosby. ISBN 978-0-7234-3221-0.
- 1 2 Olkkola KT, Ahonen J (2008). "Midazolam and other benzodiazepines". Handb Exp Pharmacol. Handbook of Experimental Pharmacology 182 (182): 335–60. doi:10.1007/978-3-540-74806-9_16. ISBN 978-3-540-72813-9. PMID 18175099.
- 1 2 Dikeos DG, Theleritis CG, Soldatos CR (2008). "Benzodiazepines: effects on sleep". In Pandi-Perumal SR, Verster JC, Monti JM, Lader M, Langer SZ (eds.). Sleep Disorders: Diagnosis and Therapeutics. Informa Healthcare. pp. 220–2. ISBN 0-415-43818-7.
- 1 2 Saïas T, Gallarda T (2008). "[Paradoxical aggressive reactions to benzodiazepine use: a review]". Encephale (in French) 34 (4): 330–6. doi:10.1016/j.encep.2007.05.005. PMID 18922233.
- 1 2 Lader M (2008). "Effectiveness of benzodiazepines: do they work or not?". Expert Rev Neurother (PDF) 8 (8): 1189–91. doi:10.1586/14737175.8.8.1189. PMID 18671662.
- 1 2 3 4 Lader M, Tylee A, Donoghue J (2009). "Withdrawing benzodiazepines in primary care". CNS Drugs 23 (1): 19–34. doi:10.2165/0023210-200923010-00002. PMID 19062773.
- 1 2 3 4 5 6 7 8 9 Ashton H (2005). "The diagnosis and management of benzodiazepine dependence" (PDF). Current Opinion in Psychiatry 18 (3): 249–55. doi:10.1097/01.yco.0000165594.60434.84. PMID 16639148.
- 1 2 3 Ashton H (2004). "Benzodiazepine dependence". In Haddad P, Dursun S, Deakin B (eds.). Adverse Syndromes and Psychiatric Drugs: A Clinical Guide. Oxford University Press. pp. 239–60. ISBN 978-0-19-852748-0.
- 1 2 3 McIntosh A, Semple D, Smyth R, Burns J, Darjee R (2005). "Depressants". Oxford Handbook of Psychiatry (1st ed.). Oxford University Press. p. 540. ISBN 0-19-852783-7.
- ↑ American Geriatrics Society 2012 Beers Criteria Update Expert Panel (2012). "American Geriatrics Society updated Beers Criteria for potentially inappropriate medication use in older adults" (PDF). U.S. Department of Health & Human Services. guideline.gov. Agency for Healthcare Research and Quality.
- 1 2 3 American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Obstetrics (April 2008). "ACOG Practice Bulletin no. 92: Use of psychiatric medications during pregnancy and lactation". Obstet Gynecol 111 (4): 1001–20. doi:10.1097/AOG.0b013e31816fd910. PMID 18378767. Lay summary.
- 1 2 Fraser AD (1998). "Use and abuse of the benzodiazepines". Ther Drug Monit 20 (5): 481–9. doi:10.1097/00007691-199810000-00007. PMID 9780123.
- 1 2 3 Charlson F, Degenhardt L, McLaren J, Hall W, Lynskey M (2009). "A systematic review of research examining benzodiazepine-related mortality". Pharmacoepidemiol Drug Saf 18 (2): 93–103. doi:10.1002/pds.1694. PMID 19125401.
- 1 2 White JM, Irvine RJ (1999). "Mechanisms of fatal opioid overdose". Addiction 94 (7): 961–72. doi:10.1046/j.1360-0443.1999.9479612.x. PMID 10707430.
- 1 2 Lader MH (1999). "Limitations on the use of benzodiazepines in anxiety and insomnia: are they justified?". E ur Neuropsychopharmacol 9 (Suppl 6): S399–405. doi:10.1016/S0924-977X(99)00051-6. PMID 10622686.
- 1 2 3 4 5 6 7 8 Royal Pharmaceutical Society of Great Britain (2009). British National Formulary (BNF 57). BMJ Group and RPS Publishing. ISBN 978-0-85369-845-6.
- 1 2 Perugi G, Frare F, Toni C (2007). "Diagnosis and treatment of agoraphobia with panic disorder". CNS Drugs 21 (9): 741–64. doi:10.2165/00023210-200721090-00004. PMID 17696574.
- ↑ Tesar GE (1990). "High-potency benzodiazepines for short-term management of panic disorder: the U.S. experience". J Clin Psychiatry 51 (Suppl): 4–10; discussion 50–3. PMID 1970816.
- ↑ Faught E (2004). "Treatment of refractory primary generalized epilepsy". Rev Neurol Dis 1 (Suppl 1): S34–43. PMID 16400293.
- ↑ Allgulander C, Bandelow B, Hollander E, Montgomery SA, Nutt DJ, Okasha A, Pollack MH, Stein DJ, Swinson RP (2003). "WCA recommendations for the long-term treatment of generalized anxiety disorder". CNS Spectr 8 (Suppl 1): 53–61. PMID 14767398.
- ↑ Stevens JC, Pollack MH (2005). "Benzodiazepines in clinical practice: consideration of their long-term use and alternative agents". Journal of Clinical Psychiatry 66 (Suppl 2): 21–27. PMID 15762816.
The frequent use of benzodiazepines for the treatment of anxiety is likely a reflection of their effectiveness, rapid onset of anxiolytic effect, and tolerability.
- 1 2 3 4 5 6 McIntosh A, Cohen A, Turnbull N; et al. (2004). "Clinical guidelines and evidence review for panic disorder and generalised anxiety disorder" (PDF). National Collaborating Centre for Primary Care. Retrieved 2009-06-16.
- ↑ Bandelow B, Zohar J, Hollander E, Kasper S, Möller HJ (October 2002). "World Federation of Societies of Biological Psychiatry (WFSBP) Guidelines for the Pharmacological Treatment of Anxiety, Obsessive-Compulsive and Posttraumatic Stress Disorders". The World Journal of Biological Psychiatry (Informa Healthcare) 3 (4): 171–99. doi:10.3109/15622970209150621. PMID 12516310.
- 1 2 3 Work Group on Panic Disorder (January 2009). "APA Practice Guideline for the Treatment of Patients With Panic Disorder, Second Edition" (PDF). Retrieved July 12, 2009.
- ↑ Barbui C, Cipriani A (2009). "Proposal for the inclusion in the WHO Model List of Essential Medicines of a selective serotonin-reuptake inhibitor for Generalised Anxiety Disorder" (PDF). WHO Collaborating Centre for Research and Training in Mental Health. Retrieved 2009-06-23.
- ↑ Cloos JM, Ferreira V (2009). "Current use of benzodiazepines in anxiety disorders". Current Opinion in Psychiatry 22 (1): 90–95. doi:10.1097/YCO.0b013e32831a473d. PMID 19122540.
- ↑ Martin JL, Sainz-Pardo M, Furukawa TA, Martín-Sánchez E, Seoane T, Galán C (September 2007). "Benzodiazepines in generalized anxiety disorder: heterogeneity of outcomes based on a systematic review and meta-analysis of clinical trials". J. Psychopharmacol. (Oxford) 21 (7): 774–82. doi:10.1177/0269881107077355. PMID 17881433.
- ↑ "Clinical Guideline 22 (amended). Anxiety: management of anxiety (panic disorder, with or without agoraphobia, and generalised anxiety disorder) in adults in primary, secondary and community care" (PDF). National Institute for Health and Clinical Excellence. 2007. pp. 23–25. Retrieved 2009-08-08.
- ↑ Canadian Psychiatric Association (July 2006). "Clinical practice guidelines. Management of anxiety disorders". Can J Psychiatry (PDF) 51 (8 Suppl 2): 51S–55S. PMID 16933543. Retrieved 2009-08-08.
- 1 2 Bandelow B, Reitt M, Röver C, Michaelis S, Görlich Y, Wedekind D (2015). "Efficacy of treatments for anxiety disorders: a meta-analysis". International Clinical Psychopharmacology 30 (4): 183–92. doi:10.1097/YIC.0000000000000078. PMID 25932596.
- 1 2 3 4 "Technology Appraisal Guidance 77. Guidance on the use of zaleplon, zolpidem and zopiclone for the short-term management of insomnia" (PDF). National Institute for Clinical Excellence. April 2004. Retrieved 2009-07-26.
- 1 2 3 Ramakrishnan K, Scheid DC (August 2007). "Treatment options for insomnia". American Family Physician 76 (4): 517–26. PMID 17853625.
- ↑ Carlstedt RA (13 December 2009). Handbook of Integrative Clinical Psychology, Psychiatry, and Behavioral Medicine: Perspectives, Practices, and Research. Springer Publishing Company. pp. 128–30. ISBN 0-8261-1094-0.
- 1 2 3 Buscemi N, Vandermeer B, Friesen C, Bialy L, Tubman M, Ospina M, Klassen TP, Witmans M (June 2005). "Manifestations and Management of Chronic Insomnia in Adults. Summary, Evidence Report/Technology Assessment: Number 125" (PDF). Agency for Healthcare Research and Quality.
- 1 2 American Geriatrics Society. "Five Things Physicians and Patients Should Question". Choosing Wisely: an initiative of the ABIM Foundation (American Geriatrics Society). Retrieved August 1, 2013, which cites
- Finkle WD, Der JS, Greenland S, Adams JL, Ridgeway G, Blaschke T, Wang Z, Dell RM, VanRiper KB (2011). "Risk of Fractures Requiring Hospitalization After an Initial Prescription for Zolpidem, Alprazolam, Lorazepam, or Diazepam in Older Adults". Journal of the American Geriatrics Society 59 (10): 1883–1890. doi:10.1111/j.1532-5415.2011.03591.x. PMID 22091502.
- Allain H, Bentué-Ferrer D, Polard E, Akwa Y, Patat A (2005). "Postural instability and consequent falls and hip fractures associated with use of hypnotics in the elderly: A comparative review". Drugs & aging 22 (9): 749–765. doi:10.2165/00002512-200522090-00004. PMID 16156679.
- "American Geriatrics Society Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults". Journal of the American Geriatrics Society 60 (4): 616–631. 2012. doi:10.1111/j.1532-5415.2012.03923.x. PMC 3571677. PMID 22376048.
- ↑ Scottish Intercollegiate Guidelines Network (2005). "Diagnosis and management of epilepsy in adults" (PDF). pp. 17–9. Retrieved 2009-06-05.
- 1 2 Stokes T, Shaw EJ, Juarez-Garcia A, Camosso-Stefinovic J, Baker R (October 2004). Clinical Guidelines and Evidence Review for the Epilepsies: diagnosis and management in adults and children in primary and secondary care (PDF). London: Royal College of General Practitioners. pp. 61, 64–65. Retrieved 2009-06-02.
- 1 2 3 Shorvon SD (March 2009). "Drug treatment of epilepsy in the century of the ILAE: the second 50 years, 1959–2009". Epilepsia 50 (Suppl 3): 93–130. doi:10.1111/j.1528-1167.2009.02042.x. PMID 19298435.
- ↑ Stokes T, Shaw EJ, Juarez-Garcia A, Camosso-Stefinovic J, Baker R (October 2004). "Clinical Guidelines and Evidence Review for the Epilepsies: diagnosis and management in adults and children in primary and secondary care (Appendix B)" (PDF). London: Royal College of General Practitioners. p. 432. Retrieved 2009-06-02.
- ↑ Ashworth M, Gerada C (1997). "ABC of mental health. Addiction and dependence—II: Alcohol". BMJ 315 (7104): 358–60. doi:10.1136/bmj.315.7104.358. PMC 2127236. PMID 9270461.
- ↑ Kraemer KL, Conigliaro J, Saitz R (1999). "Managing alcohol withdrawal in the elderly". Drugs Aging 14 (6): 409–25. doi:10.2165/00002512-199914060-00002. PMID 10408740.
- ↑ Prater CD, Miller KE, Zylstra RG (September 1999). "Outpatient detoxification of the addicted or alcoholic patient". American Family Physician 60 (4): 1175–83. PMID 10507746.
- ↑ Ebell MH (April 2006). "Benzodiazepines for alcohol withdrawal". American Family Physician 73 (7): 1191. PMID 16623205.
- ↑ Peppers MP (1996). "Benzodiazepines for alcohol withdrawal in the elderly and in patients with liver disease". Pharmacotherapy 16 (1): 49–57. doi:10.1002/j.1875-9114.1996.tb02915.x. PMID 8700792.
- ↑ Devlin JW, Roberts RJ (July 2009). "Pharmacology of commonly used analgesics and sedatives in the ICU: benzodiazepines, propofol, and opioids". Crit Care Clin 25 (3): 431–49, vii. doi:10.1016/j.ccc.2009.03.003. PMID 19576523.
- ↑ Broscheit J, Kranke P (February 2008). "[The preoperative medication: background and specific indications for the selection of the drugs]". Anasthesiol Intensivmed Notfallmed Schmerzther 43 (2): 134–43. doi:10.1055/s-2008-1060547. PMID 18293248.
- ↑ Berthold C (May 2007). "Enteral sedation: safety, efficacy, and controversy". Compend Contin Educ Dent 28 (5): 264–71; quiz 272, 282. PMID 17607891.
- ↑ Mañon-Espaillat R, Mandel S (1999). "Diagnostic algorithms for neuromuscular diseases". Clin Podiatr Med Surg 16 (1): 67–79. PMID 9929772.
- ↑ Kamen L, Henney HR, Runyan JD (February 2008). "A practical overview of tizanidine use for spasticity secondary to multiple sclerosis, stroke, and spinal cord injury". Curr Med Res Opin 24 (2): 425–39. doi:10.1185/030079908X261113. PMID 18167175.
- ↑ Wyatt JP, Illingworth RN, Robertson CE, Clancy MJ, Munro PT (2005). "Poisoning". Oxford Handbook of Accident and Emergency Medicine (2nd ed.). Oxford University Press. pp. 173–208. ISBN 978-0-19-852623-0.
- ↑ Zimbroff DL (2008). "Pharmacological control of acute agitation: focus on intramuscular preparations". CNS Drugs 22 (3): 199–212. doi:10.2165/00023210-200822030-00002. PMID 18278976.
- ↑ Curtin F, Schulz P (2004). "Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis". J Affect Disord 78 (3): 201–8. doi:10.1016/S0165-0327(02)00317-8. PMID 15013244.
- ↑ Gillies D, Beck A, McCloud A, Rathbone J, Gillies D (2005). Gillies, Donna, ed. "Benzodiazepines alone or in combination with antipsychotic drugs for acute psychosis". Cochrane Database of Systematic Reviews (4): CD003079. doi:10.1002/14651858.CD003079.pub2. PMID 16235313.
- ↑ Schenck CH, Arnulf I, Mahowald MW (June 2007). "Sleep and Sex: What Can Go Wrong? A Review of the Literature on Sleep Related Disorders and Abnormal Sexual Behaviors and Experiences". Sleep 30 (6): 683–702. PMC 1978350. PMID 17580590.
- ↑ Ferini-Strambi L, Zucconi M (September 2000). "REM sleep behavior disorder". Clin Neurophysiol. 111 Suppl 2: S136–40. doi:10.1016/S1388-2457(00)00414-4. PMID 10996567.
- ↑ Silber MH (February 2001). "Sleep disorders". Neurol Clin 19 (1): 173–86. doi:10.1016/S0733-8619(05)70011-6. PMID 11471763.
- ↑ Grupo Brasileiro de Estudos em Síndrome das Pernas Inquietas (GBE-SPI) (September 2007). "Síndrome das pernas inquietas: diagnóstico e tratamento. Opinião de especialistas brasileiros" [Restless legs syndrome: diagnosis and treatment. Opinion of Brazilian experts]. Arq. Neuro-Psiquiatr. (in Portuguese) 65 (3a): 721–7. doi:10.1590/S0004-282X2007000400035. ISSN 0004-282X. PMID 17876423.
- ↑ Trenkwalder C, Hening WA, Montagna P, Oertel WH, Allen RP, Walters AS, Costa J, Stiasny-Kolster K, Sampaio C (December 2008). "Treatment of restless legs syndrome: an evidence-based review and implications for clinical practice" (PDF). Mov Disord 23 (16): 2267–302. doi:10.1002/mds.22254. PMID 18925578.
- ↑ Schatzberg AF, Nemeroff CB, eds. (May 6, 2009). The American Psychiatric Publishing Textbook of Psychopharmacology (Fourth ed.). American Psychiatric Publishing. p. 470. ISBN 978-1-58562-309-9.
- ↑ Bandelow B (September 2008). "The medical treatment of obsessive-compulsive disorder and anxiety". CNS Spectr 13 (9 Suppl 14): 37–46. PMID 18849910.
- ↑ Attard A, Ranjith G, Taylor D (August 2008). "Delirium and its treatment". CNS Drugs 22 (8): 631–44. doi:10.2165/00023210-200822080-00002. PMID 18601302.
- ↑ Gallegos J, Vaidya P, D'Agati D, Jayaram G, Nguyen T, Tripathi A, Trivedi JK, Reti IM (June 2012). "Decreasing adverse outcomes of unmodified electroconvulsive therapy: suggestions and possibilities". J ECT 28 (2): 77–81. doi:10.1097/YCT.0b013e3182359314. PMID 22531198.
- 1 2 3 4 5 Ballenger JC (2000). "Benzodiazepine receptors agonists and antagonists". In Sadock VA, Sadock BJ, Kaplan HI (eds.). Kaplan & Sadock's Comprehensive Textbook of Psychiatry (7th ed.). Lippincott Williams & Wilkins. pp. 2317–23. ISBN 0-683-30128-4.
- 1 2 3 4 Meyler L, Aronson JK, ed. (2006). Meyler's Side Effects of Drugs: the International Encyclopedia of Adverse Drug Reactions and Interactions (15th ed.). Elsevier. pp. 429–43. ISBN 0-444-50998-4.
- ↑ Committee on Safety of Medicines (1988). "Benzodiazepines, dependence and withdrawal symptoms" (PDF). Medicines and Healthcare products Regulatory Agency. Retrieved 2009-05-28.
- 1 2 3 4 Moody D (2004). "Drug interactions with benzodiazepines". In Raymon LP, Mozayani A (eds.). Handbook of Drug Interactions: a Clinical and Forensic Guide. Humana. pp. 3–88. ISBN 1-58829-211-8.
- ↑ Roach SS, Ford SM (2006). "Sedatives and hypnotics". Introductory Clinical Pharmacology (8th ed.). Lippincott Williams & Wilkins. p. 236. ISBN 978-0-7817-7595-3.
- ↑ Dolovich LR, Addis A, Vaillancourt JM, Power JD, Koren G, Einarson TR (September 1998). "Benzodiazepine use in pregnancy and major malformations or oral cleft: meta-analysis of cohort and case-control studies" (PDF). BMJ 317 (7162): 839–43. doi:10.1136/bmj.317.7162.839. PMC 31092. PMID 9748174.
- ↑ American Academy of Pediatrics Committee on Drugs (1998). "Neonatal drug withdrawal" (PDF). Pediatrics 101 (6): 1079–88. PMID 9614425.
- ↑ Temazepam-Rxlist Pregnancy Category@
- ↑ Iqbal MM, Sobhan T, Ryals T (2002). "Effects of commonly used benzodiazepines on the fetus, the neonate and the nursing infant" (PDF). Psychiatr Serv 53 (1): 39–49. doi:10.1176/appi.ps.53.1.39. PMID 11773648.
- ↑ Tariq SH, Pulisetty S (2008). "Pharmacotherapy for insomnia". Clin Geriatr Med 24 (1): 93–105, vii. doi:10.1016/j.cger.2007.08.009. PMID 18035234.
- ↑ Bain KT (2006). "Management of chronic insomnia in elderly persons". Am J Geriatr Pharmacother 4 (2): 168–92. doi:10.1016/j.amjopharm.2006.06.006. PMID 16860264.
- ↑ Allain H, Bentué-Ferrer D, Polard E, Akwa Y, Patat A (2005). "Postural instability and consequent falls and hip fractures associated with use of hypnotics in the elderly: a comparative review". Drugs Aging 22 (9): 749–65. doi:10.2165/00002512-200522090-00004. PMID 16156679.
- ↑ Khong TP, de Vries F, Goldenberg JS, Klungel OH, Robinson NJ, Ibáñez L, Petri H (July 2012). "Potential impact of benzodiazepine use on the rate of hip fractures in five large European countries and the United States". Calcif. Tissue Int. 91 (1): 24–31. doi:10.1007/s00223-012-9603-8. PMC 3382650. PMID 22566242.
- ↑ Bogunovic OJ, Greenfield SF (2004). "Practical geriatrics: Use of benzodiazepines among elderly patients" (PDF). Psychiatr Serv 55 (3): 233–5. doi:10.1176/appi.ps.55.3.233. PMID 15001721.
- 1 2 Jackson SG, Jansen P, Mangoni A (22 May 2009). Prescribing for Elderly Patients. Wiley. pp. 47–48. ISBN 0-470-02428-3.
- ↑ Rosenthal TC, Williams M, Naughton BJ (2006). Office care geriatrics. Philadelphia: Lippincott Williams Wilkins. pp. 260–262. ISBN 0-7817-6196-4.
- ↑ Hulse GK, Lautenschlager NT, Tait RJ, Almeida OP (2005). "Dementia associated with alcohol and other drug use". Int Psychogeriatr 17 (Suppl 1): S109–27. doi:10.1017/S1041610205001985. PMID 16240487.
- ↑ Verdoux H, Lagnaoui R, Begaud B (2005). "Is benzodiazepine use a risk factor for cognitive decline and dementia? A literature review of epidemiological studies". Psychol Med 35 (3): 307–15. doi:10.1017/S0033291704003897. PMID 15841867.
- ↑ Snowden M, Sato K, Roy-Byrne P (2003). "Assessment and treatment of nursing home residents with depression or behavioral symptoms associated with dementia: a review of the literature". J Am Geriatr Soc 51 (9): 1305–17. doi:10.1046/j.1532-5415.2003.51417.x. PMID 12919245.
- ↑ Wang PS, Brookhart MA, Setoguchi S, Patrick AR, Schneeweiss S (2006). "Psychotropic medication use for behavioral symptoms of dementia". Curr Neurol Neurosci Rep 6 (6): 490–5. doi:10.1007/s11910-006-0051-6. PMID 17074284.
- 1 2 Longo LP, Johnson B (April 2000). "Addiction: Part I. Benzodiazepines—side effects, abuse risk and alternatives". American Family Physician 61 (7): 2121–8. PMID 10779253.
- 1 2 Tasman A, Lieberman JA (2006). Handbook of Psychiatric Drugs. Wiley. p. 151. ISBN 0-470-02821-1.
- ↑ Stone KL, Ensrud KE, Ancoli-Israel S (September 2008). "Sleep, insomnia and falls in elderly patients". Sleep Med. 9 Suppl 1: S18–22. doi:10.1016/S1389-9457(08)70012-1. PMID 18929314.
- ↑ Rapoport MJ, Lanctôt KL, Streiner DL, Bédard M, Vingilis E, Murray B, Schaffer A, Shulman KI, Herrmann N (2009). "Benzodiazepine use and driving: a meta-analysis". J Clin Psychiatry 70 (5): 663–73. doi:10.4088/JCP.08m04325. PMID 19389334.
- ↑ Orriols L, Salmi LR, Philip P, Moore N, Delorme B, Castot A, Lagarde E (2009). "The impact of medicinal drugs on traffic safety: a systematic review of epidemiological studies". Pharmacoepidemiol Drug Saf 18 (8): 647–58. doi:10.1002/pds.1763. PMC 2780583. PMID 19418468.
- ↑ "benzodiazepines-oral" at medicinenet.com
- ↑ Hammersley D, Beeley L (1996). "The effects of medication on counselling". In Palmer S, Dainow S, Milner P. Counselling: The BACP Counselling Reader 1. Sage. pp. 211–4. ISBN 978-0-8039-7477-7.
- 1 2 3 Stewart SA (2005). "The effects of benzodiazepines on cognition" (PDF). J Clin Psychiatry 66 (Suppl 2): 9–13. PMID 15762814.
- 1 2 Barker MJ, Greenwood KM, Jackson M, Crowe SF (2004). "Cognitive effects of long-term benzodiazepine use: a meta-analysis". CNS Drugs 18 (1): 37–48. doi:10.2165/00023210-200418010-00004. PMID 14731058.
- ↑ Riss J, Cloyd J, Gates J, Collins S (August 2008). "Benzodiazepines in epilepsy: pharmacology and pharmacokinetics". Acta Neurol Scand 118 (2): 69–86. doi:10.1111/j.1600-0404.2008.01004.x. PMID 18384456.
- 1 2 Paton C (2002). "Benzodiazepines and disinhibition: a review" (PDF). Psychiatr Bull R Coll Psychiatr 26 (12): 460–2. doi:10.1192/pb.26.12.460.
- ↑ Bond AJ (1998). "Drug-induced behavioural disinhibition: incidence, mechanisms and therapeutic implications". CNS Drugs 9 (1): 41–57. doi:10.2165/00023210-199809010-00005.
- ↑ Drummer OH (2002). "Benzodiazepines—effects on human performance and behavior". Forensic Sci Rev 14 (1–2): 1–14.
- 1 2 Ashton H (2007). "Drug dependency: benzodiazepines". In Ayers S, Baum A, McManus C, Newman S (eds.). Cambridge Handbook of Psychology, Health and Medicine (2nd ed.). Cambridge University Press. pp. 675–8. ISBN 978-0-521-87997-2.
- 1 2 3 Guina, Jeffrey; Rossetter, Sarah R.; DeRHODES, Bethany J.; Nahhas, Ramzi W.; Welton, Randon S. (2015-07-01). "Benzodiazepines for PTSD: A Systematic Review and Meta-Analysis". Journal of Psychiatric Practice 21 (4): 281–303. doi:10.1097/PRA.0000000000000091. ISSN 1538-1145. PMID 26164054.
- 1 2 3 4 5 6 7 Michelini S, Cassano GB, Frare F; et al. (2016). "Long-term use of benzodiazepines: tolerance, dependence and clinical problems in anxiety and mood disorders.". Pharmacopsychiatry 29:127-134.
- 1 2 Lydiard, R. B.; Brawman-Mintzer, O.; Ballenger, J. C. (1996-08-01). "Recent developments in the psychopharmacology of anxiety disorders". Journal of Consulting and Clinical Psychology 64 (4): 660–668. doi:10.1037/0022-006x.64.4.660. ISSN 0022-006X. PMID 8803355.
- 1 2 Tasman A, Kay J, Lieberman JA (Eds.) (2008). Psychiatry (3rd ed.). Chichester, England: John Wiley & Sons. pp. 1186–1200, 2603–2615. ISBN 978-0470065716.
- ↑ Gelpin, E.; Bonne, O.; Peri, T.; Brandes, D.; Shalev, A. Y. (1996-09-01). "Treatment of recent trauma survivors with benzodiazepines: a prospective study". The Journal of Clinical Psychiatry 57 (9): 390–394. ISSN 0160-6689. PMID 9746445.
- ↑ Rosen, Craig S.; Greenbaum, Mark A.; Schnurr, Paula P.; Holmes, Tyson H.; Brennan, Penny L.; Friedman, Matthew J. (2013-12-01). "Do benzodiazepines reduce the effectiveness of exposure therapy for posttraumatic stress disorder?". The Journal of Clinical Psychiatry 74 (12): 1241–1248. doi:10.4088/JCP.13m08592. ISSN 1555-2101. PMID 24434093.
- ↑ Wilhelm, F. H.; Roth, W. T. (1997-09-01). "Acute and delayed effects of alprazolam on flight phobics during exposure". Behaviour Research and Therapy 35 (9): 831–841. doi:10.1016/s0005-7967(97)00033-8. ISSN 0005-7967. PMID 9299803.
- ↑ Matar, Michael A.; Zohar, Joseph; Kaplan, Zeev; Cohen, Hagit (2009-04-01). "Alprazolam treatment immediately after stress exposure interferes with the normal HPA-stress response and increases vulnerability to subsequent stress in an animal model of PTSD". European Neuropsychopharmacology: The Journal of the European College of Neuropsychopharmacology 19 (4): 283–295. doi:10.1016/j.euroneuro.2008.12.004. ISSN 1873-7862. PMID 19167197.
- ↑ Curran, H. V.; Collins, R.; Fletcher, S.; Kee, S. C. Y.; Woods, B.; Iliffe, S. (2003-10-01). "Older adults and withdrawal from benzodiazepine hypnotics in general practice: effects on cognitive function, sleep, mood and quality of life". Psychological Medicine 33 (7): 1223–1237. doi:10.1017/s0033291703008213. ISSN 0033-2917. PMID 14580077.
- ↑ Pary R, Lewis S (2008). "Prescribing benzodiazepines in clinical practice". Resident and Staff Physician, 54(1), 8-17.
- ↑ Nardi AE, Perna G (May 2006). "Clonazepam in the treatment of psychiatric disorders: an update". Int Clin Psychopharmacol 21 (3): 131–42. doi:10.1097/01.yic.0000194379.65460.a6. PMID 16528135.
- ↑ Otto MW, Bruce SE, Deckersbach T (2005). "Benzodiazepine use, cognitive impairment, and cognitive-behavioral therapy for anxiety disorders: issues in the treatment of a patient in need" (PDF). J Clin Psychiatry 66 (Suppl 2): 34–8. PMID 15762818.
- 1 2 3 Chouinard G (2004). "Issues in the clinical use of benzodiazepines: potency, withdrawal, and rebound" (PDF). J Clin Psychiatry 65 (Suppl 5): 7–12. PMID 15078112.
- ↑ Curran, H. V.; Collins, R.; Fletcher, S.; Kee, S. C. Y.; Woods, B.; Iliffe, S. (2003-10-01). "Older adults and withdrawal from benzodiazepine hypnotics in general practice: effects on cognitive function, sleep, mood and quality of life". Psychological Medicine 33 (7): 1223–1237. doi:10.1017/s0033291703008213. ISSN 0033-2917. PMID 14580077.
- ↑ "Holbrook AM. %282004%29. Treating insomnia. - PubMed - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2015-12-10.
- ↑ Poyares, Dalva; Guilleminault, Christian; Ohayon, Maurice M.; Tufik, Sergio (2004-06-01). "Chronic benzodiazepine usage and withdrawal in insomnia patients". Journal of Psychiatric Research 38 (3): 327–334. doi:10.1016/j.jpsychires.2003.10.003. ISSN 0022-3956. PMID 15003439.
- ↑ Friedman MJ (1998). "Pharmacotherapy for posttraumatic stress disorder: a status report". Clinical Neurosciences, 52(Suppl.), S115-S121.
- ↑ Heather N, Bowie A, Ashton H, McAvoy B, Spencer I, Brodie J, Giddings D (2004). "Randomised controlled trial of two brief interventions against long-term benzodiazepine use: outcome of intervention". Addiction Research and Theory 12 (2): 141–154.
- ↑ Bandelow, Borwin; Zohar, Joseph; Hollander, Eric; Kasper, Siegfried; Möller, Hans-Jürgen; Zohar, Joseph; Hollander, Eric; Kasper, Siegfried (2008-01-01). "World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders - first revision". The World Journal of Biological Psychiatry: The Official Journal of the World Federation of Societies of Biological Psychiatry 9 (4): 248–312. doi:10.1080/15622970802465807. ISSN 1562-2975. PMID 18949648. Missing
|last6=in Authors list (help) - ↑ Ashton, Heather (2005-05-01). "The diagnosis and management of benzodiazepine dependence". Current Opinion in Psychiatry 18 (3): 249–255. doi:10.1097/01.yco.0000165594.60434.84. ISSN 0951-7367. PMID 16639148.
- ↑ Morin, Charles M.; Bélanger, Lynda; Bastien, Célyne; Vallières, Annie (2005-01-01). "Long-term outcome after discontinuation of benzodiazepines for insomnia: a survival analysis of relapse". Behaviour Research and Therapy 43 (1): 1–14. doi:10.1016/j.brat.2003.12.002. ISSN 0005-7967. PMID 15531349.
- 1 2 American Psychiatric Association (2013). Diagnostic and statistical manual of mental disorders, fifth edition. Arlington, VA: American Psychiatric Association.
- ↑ Harrison PC, Gelder MG, Cowen P (2006). "The misuse of alcohol and drugs". Shorter Oxford Textbook of Psychiatry (5th ed.). Oxford University Press. pp. 461–2. ISBN 0-19-856667-0.
- ↑ Judith Collier, Murray Longmore, Keith Amarakone (31 January 2013). "Psychiatry". Oxford Handbook of Clinical Specialties. OUP Oxford. p. 368. ISBN 978-0-19-150476-1.
- ↑ Ashton H (1991). "Protracted withdrawal syndromes from benzodiazepines". J Subst Abuse Treat 8 (1–2): 19–28. doi:10.1016/0740-5472(91)90023-4. PMID 1675688.
- 1 2 3 4 Ashton CH (2002). Benzodiazepines: how they work & how to withdraw (aka The Ashton Manual). Retrieved 2009-05-27.
- ↑ Lal R, Gupta S, Rao R, Kattimani S (2007). "Emergency management of substance overdose and withdrawal" (PDF). Substance Use Disorder. World Health Organization (WHO). p. 82. Retrieved 2009-06-06.
Generally, a longer-acting benzodiazepine such as chlordiazepoxide or diazepam is used and the initial dose titrated downward
- ↑ Ford C, Law F (July 2014). "Guidance for the use and reduction of misuse of benzodiazepines and other hypnotics and anxiolytics in general practice" (PDF). smmgp.org.uk.
- ↑ Ebadi M (23 October 2007). "Alphabetical presentation of drugs". Desk Reference for Clinical Pharmacology (2nd ed.). USA: CRC Press. p. 512. ISBN 978-1-4200-4743-1.
- ↑ Overdose Death Rates. By National Institute on Drug Abuse (NIDA).
- ↑ Gaudreault P, Guay J, Thivierge RL, Verdy I (1991). "Benzodiazepine poisoning. Clinical and pharmacological considerations and treatment". Drug Saf 6 (4): 247–65. doi:10.2165/00002018-199106040-00003. PMID 1888441.
- ↑ Robin Mantooth (28 January 2010). "Toxicity, benzodiazepine". eMedicine. Retrieved 2010-10-02.
- 1 2 Ramrakha P, Moore K (2004). "Chapter 14: Drug overdoses". Oxford Handbook of Acute Medicine (2nd ed.). Oxford University Press. pp. 791–838 (798). ISBN 0-19-852072-7.
- ↑ Klein-Schwartz W, Oderda GM (1991). "Poisoning in the elderly. Epidemiological, clinical and management considerations". Drugs Aging 1 (1): 67–89. doi:10.2165/00002512-199101010-00008. PMID 1794007.
- ↑ Buckley NA, Dawson AH, Whyte IM, O'Connell DL (1995). "Relative toxicity of benzodiazepines in overdose". BMJ 310 (28): 219–221. doi:10.1136/bmj.310.6974.219. PMC 2548618. PMID 7866122.
- ↑ Serfaty M, Masterton G (1994). "Fatal poisonings attributed to benzodiazepines in Britain during the 1980s". British Journal of Psychiatry 163 (1): 128–9. doi:10.1192/bjp.163.3.386. PMID 8104653.
- ↑ Seger DL (2004). "Flumazenil—treatment or toxin". J Toxicol Clin Toxicol 42 (2): 209–16. doi:10.1081/CLT-120030946. PMID 15214628.
- ↑ "Treatment of benzodiazepine overdose with flumazenil. The flumazenil in benzodiazepine intoxication multicenter study group". Clin Ther 14 (6): 978–95. 1992. PMID 1286503.
- ↑ Spivey WH (1992). "Flumazenil and seizures: analysis of 43 cases". Clin Ther 14 (2): 292–305. PMID 1611650.
- ↑ Goldfrank LR (2002). Goldfrank's Toxicologic Emergencies. McGraw-Hill. ISBN 0-07-136001-8.
- 1 2 Norman TR, Ellen SR, Burrows GD (1997). "Benzodiazepines in anxiety disorders: managing therapeutics and dependence" (PDF). Med J Aust 167 (9): 490–5. PMID 9397065.
- 1 2 3 Olsen RW, Betz H (2006). "GABA and glycine". In Siegel GJ, Albers RW, Brady S, Price DD (eds.). Basic Neurochemistry: Molecular, Cellular and Medical Aspects (7th ed.). Elsevier. pp. 291–302. ISBN 0-12-088397-X.
- ↑ Rudolph U, Möhler H (2006). "GABA-based therapeutic approaches: GABAA receptor subtype functions". Current Opinion in Pharmacology 6 (1): 18–23. doi:10.1016/j.coph.2005.10.003. PMID 16376150.
- ↑ Puri BK, Tyrer P (28 August 1998). "Clinical psychopharmacology". Sciences Basic to Psychiatry (2nd ed.). Churchill Livingstone. pp. 155–156. ISBN 978-0-443-05514-0. Retrieved 11 July 2009.
- ↑ Johnston GA (1996). "GABAA receptor pharmacology". Pharmacol Ther 69 (3): 173–98. doi:10.1016/0163-7258(95)02043-8. PMID 8783370.
- ↑ Wafford KA, Macaulay AJ, Fradley R, O'Meara GF, Reynolds DS, Rosahl TW (2004). "Differentiating the role of gamma-aminobutyric acid type A (GABAA) receptor subtypes". Biochem Soc Trans 32 (Pt3): 553–6. doi:10.1042/BST0320553. PMID 15157182.
- ↑ Hevers W, Lüddens H (1998). "The diversity of GABAA receptors. Pharmacological and electrophysiological properties of GABAA channel subtypes". Mol Neurobiol 18 (1): 35–86. doi:10.1007/BF02741459. PMID 9824848.
- ↑ Arvat E, Giordano R, Grottoli S, Ghigo E (2002). "Benzodiazepines and anterior pituitary function". J Endocrinol Invest 25 (8): 735–47. doi:10.1007/bf03345110. PMID 12240908.
- 1 2 Zavala F (1997). "Benzodiazepines, anxiety and immunity". Pharmacol Ther 75 (3): 199–216. doi:10.1016/S0163-7258(97)00055-7. PMID 9504140.
- ↑ Zisterer DM, Williams DC (1997). "Peripheral-type benzodiazepine receptors". Gen Pharmacol 29 (3): 305–14. doi:10.1016/S0306-3623(96)00473-9. PMID 9378234.
- 1 2 Narimatsu E, Niiya T, Kawamata M, Namiki A (2006). "[The mechanisms of depression by benzodiazepines, barbiturates and propofol of excitatory synaptic transmissions mediated by adenosine neuromodulation]". Masui (in Japanese) 55 (6): 684–91. PMID 16780077.
- ↑ O'Brien (2005). "Benzodiazepine Use, Abuse, and Dependence" (PDF). J Clin Psychiatry: 66. Retrieved 5 September 2013.
- ↑ Cardinali DP, Monti JM (2006). "Chronopharmacology and its implication to the pharmacology of sleep". In Pandi-Perumal SR, Monti JM. Clinical pharmacology of sleep. Basel: Birkhäuser. pp. 211–3. ISBN 9783764374402.
- 1 2 Madsen U, Bräuner-Osborne H, Greenwood JR, Johansen TN, Krogsgaard-Larsen P, Liljefors T, Nielsen M, Frølund B (2005). "GABA and Glutamate receptor ligands and their therapeutic potential in CNS disorders". In Gad SC. Drug Discovery Handbook. Hoboken, N.J: Wiley-Interscience/J. Wiley. pp. 797–907. ISBN 0-471-21384-5.
- 1 2 Panico R, Powell WH, Richer JC, eds. (1993). A Guide to IUPAC Nomenclature of Organic Compounds. IUPAC/Blackwell Science. pp. 40–3. ISBN 0-632-03488-2.; Moss GP (1998). "Nomenclature of fused and bridged fused ring systems (IUPAC Recommendations 1998)" (PDF). Pure Appl Chem 70 (1): 143–216. doi:10.1351/pac199870010143.
- ↑ CAS registry number:2898-08-0 1,3-dihydro-5-phenyl-2H-1,4-benzodiazepin-2-one; other names: Ro 05-2921, dechlorodemethyldiazepam.
- ↑ Ripka WC, De Lucca GV, Bach AC, Pottorf RS, Blaney JM (1993). "Protein β-turn mimetics I. Design, synthesis, and evaluation in model cyclic peptides". Tetrahedron 49 (17): 3593–3608. doi:10.1016/S0040-4020(01)90217-0.
- ↑ Hata M, Marshall GR (2006). "Do benzodiazepines mimic reverse-turn structures?". Journal of Computer-Aided Molecular Design 20 (5): 321–331. doi:10.1007/s10822-006-9059-x. PMID 16972167.
- 1 2 3 4 5 Juergens, MD, Steven M. "Understanding Benzodiazepines" (PDF). California Society of Addiction Medicine. Retrieved 25 April 2012.
- 1 2 3 4 5 Carlo P, Finollo R, Ledda A, Brambilla G (January 1989). "Absence of liver DNA fragmentation in rats treated with high oral doses of 32 benzodiazepine drugs". Fundamental and Applied Toxicology 12 (1): 34–41. doi:10.1016/0272-0590(89)90059-6. PMID 2925017.
- ↑ Sternbach LH (1979). "The benzodiazepine story". Journal of Medicinal Chemistry 22 (1): 1–7. doi:10.1021/jm00187a001. PMID 34039.
During this cleanup operation, my co-worker, Earl Reeder, drew my attention to a few hundred milligrams of two products, a nicely crystalline base and its hydrochloride. Both the base, which had been prepared by treating the quinazoline N-oxide 11 with methylamine, and its hydrochloride had been made sometime in 1955. The products were not submitted for pharmacological testing at that time because of our involvement with other problems
- ↑ Miller NS, Gold MS (1990). "Benzodiazepines: reconsidered". Adv Alcohol Subst Abuse 8 (3–4): 67–84. doi:10.1300/J251v08n03_06. PMID 1971487.
- ↑ King MB (1992). "Is there still a role for benzodiazepines in general practice?". Br J Gen Pract 42 (358): 202–5. PMC 1372025. PMID 1389432.
- ↑ Peart R (1999-06-01). "Memorandum by Dr Reg Peart". Minutes of Evidence. Select Committee on Health, House of Commons, UK Parliament. Retrieved 2009-05-27.
- ↑ Jufe GS (Jul–Aug 2007). "[New hypnotics: perspectives from sleep physiology]". Vertex 18 (74): 294–9. PMID 18265473.
- ↑ Lemmer B (2007). "The sleep–wake cycle and sleeping pills". Physiol Behav 90 (2–3): 285–93. doi:10.1016/j.physbeh.2006.09.006. PMID 17049955.
- ↑ "Controlled Drugs and Substances Act". Canadian Department of Justice. Retrieved 2009-05-27.
- ↑ Blackpool NHS Primary Care Trust (1 May 2008). "Medicines Management Update" (PDF). United Kingdom National Health Service. Retrieved 2009-05-27.
- ↑ "List of Drugs Currently Controlled Under The Misuse of Drugs Legislation" (PDF). Misuse of Drugs Act UK. British Government. Retrieved 2009-05-27.
- ↑ "Opium Law" (PDF). Dutch Government. 29 November 2004. Archived from the original (PDF) on October 20, 2008. Retrieved 2009-05-27.
- ↑ Hong Kong Government. "DANGEROUS DRUGS ORDINANCE – SCHEDULE 1". Hong Kong Ordinances. Hong Kong: hklii.org.
- ↑ Lee, K. K.; Chan, T. Y.; Chan, A. W.; Lau, G. S.; Critchley, J. A. (1995). "Use and abuse of benzodiazepines in Hong Kong 1990–93 – The impact of regulatory changes". Journal of Toxicology. Clinical Toxicology 33 (6): 597–602. doi:10.3109/15563659509010615. PMID 8523479.
- ↑ International Narcotics Control Board (August 2003). "List of psychotropic substances under international control" (pdf). incb.org. Retrieved 2008-12-17.
- ↑ International Narcotics Control Board (2003). "List of psychotropic substances under international control" (PDF). Retrieved 2008-12-17.
- ↑ Home Office (2005). Explanatory memorandum to the misuse of drugs and the misuse of drugs (supply to addicts) (amendment) regulations 2005.No.2864. Accessed 20–10–03
- ↑ "List of drugs currently controlled under the misuse of drugs legislation" (PDF). UK Government Home Office. 2010-10-25. Retrieved 2011-01-30.
- ↑ Karch SB (20 December 2006). Drug Abuse Handbook (2nd ed.). United States of America: CRC Press. p. 217. ISBN 978-0-8493-1690-6.
- ↑ Gerada C, Ashworth M (1997). "ABC of mental health. Addiction and dependence—I: Illicit drugs". BMJ 315 (7103): 297–300. doi:10.1136/bmj.315.7103.297. PMC 2127199. PMID 9274553.
- ↑ Loxley W (2007). "Benzodiazepine use and harms among police detainees in Australia" (PDF). Trends Issues Crime Crim Justice (Canberra, A.C.T.: Australian Institute of Criminology) (336). ISSN 0817-8542. Retrieved 2009-06-10.
- ↑ Kintz P (2007). "Bioanalytical procedures for detection of chemical agents in hair in the case of drug-facilitated crimes". Anal Bioanal Chem 388 (7): 1467–74. doi:10.1007/s00216-007-1209-z. PMID 17340077.
- ↑ "Benzodiazepine abuse". Benzo.org.uk. Retrieved 2011-11-28.
- ↑ Farré M, Camí J (December 1991). "Pharmacokinetic considerations in abuse liability evaluation". Br J Addict. 86 (12): 1601–6. doi:10.1111/j.1360-0443.1991.tb01754.x. PMID 1786493.
- ↑ Busto U, Sellers EM (1986). "Pharmacokinetic determinants of drug abuse and dependence. A conceptual perspective". Clin Pharmacokinet. 11 (2): 144–153. doi:10.2165/00003088-198611020-00004. PMID 3514044.
- 1 2 Kahn CM, Line S, Aiello SE, ed. (2005). The Merck Veterinary Manual (9th ed.). Wiley. ISBN 978-0-911910-50-6.
- ↑ Frey HH (1989). "Anticonvulsant drugs used in the treatment of epilepsy". Probl Vet Med 1 (4): 558–77. PMID 2520134.
- ↑ Podell M (1996). "Seizures in dogs". Vet Clin North Am Small Anim Pract 26 (4): 779–809. PMID 8813750.
- ↑ Gross ME (2001). "Tranquilizers, α2-adrenergic agonists, and related agents". In Adams RH (ed.). Veterinary Pharmacology and Therapeutics (8th ed.). Iowa State University Press. pp. 325–33. ISBN 0-8138-1743-9.
External links
- National Institute on Drug Abuse: "NIDA for Teens: Prescription Depressant Medications".* Ashton CH (2002). Benzodiazepines: how they work & how to withdraw (aka The Ashton Manual). The Ashton Manual. Retrieved 2009-06-09.
- Ashton CH (2007). "Benzodiazepine equivalence table". Retrieved 2009-06-09.
- Fruchtengarten L (April 1998). Ruse M, ed. "Benzodiazepines". Poisons Information Monograph (Group monograph) G008. IPCS INCHEM. Retrieved 2009-06-09.
- Longo LP, Johnson B (April 2000). "Addiction: Part I. Benzodiazepines—side effects, abuse risk and alternatives". American Family Physician 61 (7): 2121–8. PMID 10779253.
- "The complete story of the benzodiazepines". The Eaton T. Fores Research Center. 2005. Archived from the original on June 5, 2009. Retrieved 2009-06-09.
- "Benzodiazepines". Drugs.com.
- Benzodiazepines – information from mental health charity The Royal College of Psychiatrists
- "Benzodiazepines". RxList.
- "benzodiazepines-oral" at medicinenet.com
| ||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|