Hydroxylamine
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydroxylamine | |||
| Systematic IUPAC name
Hydroxylamine[1] | |||
| Other names
Aminol Azanol | |||
| Identifiers | |||
| 7803-49-8 | |||
| 3DMet | B01184 | ||
| ChEBI | CHEBI:15429 | ||
| ChEMBL | ChEMBL1191361 | ||
| ChemSpider | 766 | ||
| EC Number | 232-259-2 | ||
| 478 | |||
| Jmol interactive 3D | Image Image | ||
| KEGG | C00192 | ||
| MeSH | Hydroxylamine | ||
| PubChem | 787 | ||
| RTECS number | NC2975000 | ||
| UNII | 2FP81O2L9Z | ||
| |||
| |||
| Properties | |||
| H3NO | |||
| Molar mass | 33.03 g·mol−1 | ||
| Appearance | Vivid white, opaque crystals | ||
| Density | 1.21 g cm−3 (at 20 °C)[2] | ||
| Melting point | 33 °C (91 °F; 306 K) | ||
| Boiling point | 58 °C (136 °F; 331 K) (decomposes) | ||
| log P | −0.758 | ||
| Acidity (pKa) | 5.95 | ||
| Basicity (pKb) | 8.05 | ||
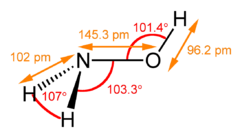
| Structure | |||
| Trigonal at N | |||
| Tetrahedral at N | |||
| 0.67553 D | |||
| Thermochemistry | |||
| 46.47 J K−1 mol−1 | |||
| Std molar entropy (S |
236.18 J K−1 mol−1 | ||
| Std enthalpy of formation (ΔfH |
−39.9 kJ mol−1 | ||
| Hazards | |||
| Safety data sheet | ICSC 0661 | ||
| EU classification (DSD) |
E | ||
| R-phrases | R2, R21/22, R37/38, R40, R41, R43, R48/22, R50 | ||
| S-phrases | (S2), S26, S36/37/39, S61 | ||
| NFPA 704 | |||
| Flash point | 129 °C (264 °F; 402 K) | ||
| 265 °C (509 °F; 538 K) | |||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (Median dose) |
408 mg/kg (oral, mouse); 59–70 mg/kg (intraperitoneal mouse, rat); 29 mg/kg (subcutaneous, rat)[3] | ||
| Related compounds | |||
| Related hydroxylammonium salts |
Hydroxylammonium chloride Hydroxylammonium nitrate Hydroxylammonium sulfate | ||
| Related compounds |
Ammonia | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Hydroxylamine is an inorganic compound with the formula NH2OH. The pure material is a white, unstable crystalline, hygroscopic compound.[4] However, hydroxylamine is almost always provided and used as an aqueous solution. It is used to prepare oximes, an important functional group. It is also an intermediate in biological nitrification. The oxidation of NH3 is mediated by the enzyme hydroxylamine oxidoreductase (HAO).
History
Hydroxylamine was first prepared as hydroxylamine hydrochloride in 1865 by the German chemist Wilhelm Clemens Lossen (1838-1906); he reacted tin and hydrochloric acid in the presence of ethyl nitrate.[5] It was first prepared in pure form in 1891 by the Dutch chemist Lobry de Bruyn and by the French chemist Léon Maurice Crismer (1858-1944).[6][7]
Functional group
Substituted derivatives of hydroxylamine are known. If the hydroxyl hydrogen is substituted, this is called an O-hydroxylamine, if one of the amine hydrogens is substituted, this is called an N-hydroxylamine. Similarly to ordinary amines, one can distinguish primary, secondary and tertiary hydroxylamines, the latter two referring to compounds where two or three hydrogens are substituted, respectively. Examples of compounds containing a hydroxylamine functional group are N-tert-butyl-hydroxylamine or the glycosidic bond in calicheamicin. N,O-Dimethylhydroxylamine is a coupling agent, used to synthesize Weinreb amides.
Production
NH2OH can be produced via several routes. The main route is via the Raschig process: aqueous ammonium nitrite is reduced by HSO3− and SO2 at 0 °C to yield a hydroxylamido-N,N-disulfonate anion:
- NH4NO2 + 2 SO2 + NH3 + H2O → 2 NH4+ + N(OH)(OSO2)22−
This anion is then hydrolyzed to give (NH3OH)2SO4:
- N(OH)(OSO2)22− + H2O → NH(OH)(OSO2)− + HSO4−
- 2 NH(OH)(OSO2)− + 2 H2O → (NH
3OH)
2SO
4 + SO2−
4
Solid NH2OH can be collected by treatment with liquid ammonia. Ammonium sulfate, (NH
4)
2SO
4, a side-product insoluble in liquid ammonia, is removed by filtration; the liquid ammonia is evaporated to give the desired product.[4]
The net reaction is:
- 2NO−
2 + 4SO
2 + 6H
2O + 6NH
3 → 4SO2−
4 + 6NH+
4 + 2NH
2OH
Hydroxylammonium salts can then be converted to hydroxylamine by neutralization:
- (NH3OH)Cl + NaOBu → NH2OH + NaCl + BuOH[4]
Julius Tafel discovered that hydroxylamine hydrochloride or sulfate salts can be produced by electrolytic reduction of nitric acid with HCl or H2SO4 respectively:[8][9]
- HNO3 + 3H2 → NH2OH + 2H2O
Hydroxylamine can also be produced by the reduction of nitrous acid or potassium nitrite with bisulfite:
- HNO2 + 2 HSO3− → N(OH)(OSO2)22− + H2O → NH(OH)(OSO2)− + HSO4−
- NH(OH)(OSO2)− + H3O+ (100 °C/1 h) → NH3(OH)+ + HSO4−
Reactions
Hydroxylamine reacts with electrophiles, such as alkylating agents, which can attach to either the oxygen or the nitrogen atoms:
- R-X + NH2OH → R-ONH2 + HX
- R-X + NH2OH → R-NHOH + HX
The reaction of NH2OH with an aldehyde or ketone produces an oxime.
- R2C=O + NH2OH∙HCl , NaOH → R2C=NOH + NaCl + H2O
This reaction is useful in the purification of ketones and aldehydes: if hydroxylamine is added to an aldehyde or ketone in solution, an oxime forms, which generally precipitates from solution; heating the precipitate with an inorganic acid then restores the original aldehyde or ketone.[10]
Oximes, e.g., dimethylglyoxime, are also employed as ligands.
NH2OH reacts with chlorosulfonic acid to give hydroxylamine-O-sulfonic acid, a useful reagent for the synthesis of caprolactam.
- HOSO2Cl + NH2OH → NH2OSO2OH + HCl
The hydroxylamine-O-sulfonic acid, which should be stored at 0 °C to prevent decomposition, can be checked by iodometric titration.
Hydroxylamine (NH2OH), or hydroxylamines (R-NHOH) can be reduced to amines.[11]
- NH2OH (Zn/HCl) → NH3
- R-NHOH (Zn/HCl) → R-NH2
Hydroxylamine explodes with heat:
- 4 NH2OH + O2 → 2 N2 + 6 H2O
The high reactivity comes in part from the partial isomerisation of the NH2OH structure to ammonia oxide (also known as azane oxide), with structure NH3+O−. [12]
Uses
Hydroxylamine and its salts are commonly used as reducing agents in myriad organic and inorganic reactions. They can also act as antioxidants for fatty acids.
 Conversion from cyclohexanone to caprolactam involving the Beckmann Rearrangement can be understood with this scheme.
Conversion from cyclohexanone to caprolactam involving the Beckmann Rearrangement can be understood with this scheme.
In the synthesis of nylon 6, cyclohexanone (1) is first converted to its oxime (2); treatment of this oxime with acid induces the Beckmann rearrangement to give caprolactam (3):
The nitrate salt, hydroxylammonium nitrate, is being researched as a rocket propellant, both in water solution as a monopropellant and in its solid form as a solid propellant.
This has also been used in the past by biologists to introduce random mutations by switching base pairs from G to A, or from C to T. This is to probe functional areas of genes to elucidate what happens if their functions are broken. Nowadays other mutagens are used. Hydroxylamine can also be used to highly selectively cleave asparaginyl-glycine peptide bonds in peptides and proteins. It also bonds to and permanently disables (poisons) heme-containing enzymes. It is used as an irreversible inhibitor of the oxygen-evolving complex of photosynthesis on account of its similar structure to water.
 This route also involves the Beckmann Rearrangement, like the conversion from cyclohexanone to caprolactam.
This route also involves the Beckmann Rearrangement, like the conversion from cyclohexanone to caprolactam.
An alternative industrial synthesis of paracetamol developed by Hoechst–Celanese involves the conversion of ketone to a ketoxime with hydroxylamine.
Some non-chemical uses include removal of hair from animal hides and photography developing solutions.[2] In the semiconductor industry, hydroxylamine is often a component in the "resist stripper", which removes photoresist after lithography.
Safety
Hydroxylamine may explode on heating. The nature of the explosive hazard is not well understood. At least two factories dealing in hydroxylamine have been destroyed since 1999 with loss of life.[13] It is known, however, that ferrous and ferric iron salts accelerate the decomposition of 50% NH2OH solutions.[14] Hydroxylamine and its derivatives are more safely handled in the form of salts.
It is an irritant to the respiratory tract, skin, eyes, and other mucous membranes. It may be absorbed through the skin, is harmful if swallowed, and is a possible mutagen.[15]
See also
References
- ↑ "Hydroxylamine - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.
- 1 2 Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ↑ Martel, B.; Cassidy, K. (2004). Chemical Risk Analysis: A Practical Handbook. Butterworth–Heinemann. p. 362. ISBN 1-903996-65-1.
- 1 2 3 Greenwood and Earnshaw. Chemistry of the Elements. 2nd Edition. Reed Educational and Professional Publishing Ltd. pp. 431–432. 1997.
- ↑ W. C. Lossen (1865) "Ueber das Hydroxylamine" (On hydroxylamine), Zeitschrift für Chemie, 8 : 551-553. From p. 551: "Ich schlage vor, dieselbe Hydroxylamin oder Oxyammoniak zu nennen." (I propose to call it hydroxylamine or oxyammonia.)
- ↑ C. A. Lobry de Bruyn (1891) "Sur l'hydroxylamine libre" (On free hydroxylamine), Recueil des travaux chimiques des Pays-Bas, 10 : 100-112.
- ↑ L. Crismer (1891) "Préparation de l'hydroxylamine cristallisée" (Preparation of crystalized hydroxylamine), Bulletin de la Société chimique de Paris, series 3, 6 : 793-795.
- ↑ James Hale, Arthur (1919). The Manufacture of Chemicals by Electrolysis (1st ed.). New York: D. Van Nostrand Co. p. 32. Retrieved 5 June 2014.
- ↑ Osswald, Philipp; & Geisler, Walter; (1941). Process of preparing hydroxylamine hydrochloride (US2242477) (PDF). U.S. Patent Office.
- ↑ Ralph Lloyd Shriner, Reynold C. Fuson, and Daniel Y. Curtin, The Systematic Identification of Organic Compounds: A Laboratory Manual, 5th ed. (New York: Wiley, 1964), chapter 6.
- ↑ Smith, Michael and Jerry March. March's advanced organic chemistry : reactions, mechanisms, and structure. New York. Wiley. p. 1554. 2001.
- ↑ "Ammonia oxide makes up some 20% of an aqueous solution of hydroxylamine.". National Center for Biotechnology Information. Chemical Communications (Cambridge, England). 28 February 2010. Retrieved 2 December 2014.
- ↑ Japan Science and Technology Agency Failure Knowledge Database.
- ↑ Cisneros, L. O.; Rogers, W. J.; Mannan, M. S.; Li, X.; Koseki, H. (2003). "Effect of Iron Ion in the Thermal Decomposition of 50 mass% Hydroxylamine/Water Solutions". J. Chem. Eng Data 48 (5): 1164–1169. doi:10.1021/je030121p.
- ↑ MSDS Sigma-Aldrich
Further reading
- Hydroxylamine
- Walters, Michael A. and Andrew B. Hoem. "Hydroxylamine." e-Encyclopedia of Reagents for Organic Synthesis. 2001.
- Schupf Computational Chemistry Lab
- M. W. Rathke A. A. Millard "Boranes in Functionalization of Olefins to Amines: 3-Pinanamine" Organic Syntheses, Coll. Vol. 6, p. 943; Vol. 58, p. 32. (preparation of hydroxylamine-O-sulfonic acid).
External links
- Calorimetric studies of hydroxylamine decomposition
- Chemical company BASF info
- MSDS
- Deadly detonation of hydroxylamine at Concept Sciences facility


