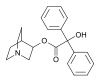
3-Quinuclidinyl benzilate
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1-azabicyclo[2.2.2]oct-3-yl 2-hydroxy-2,2-diphenylacetate | |||
| Identifiers | |||
| 6581-06-2 | |||
| ChEMBL | ChEMBL12980 | ||
| ChemSpider | 21577 | ||
| 3260 | |||
| Jmol interactive 3D | Image | ||
| MeSH | Quinuclidinyl+benzilate | ||
| PubChem | 23056 | ||
| |||
| |||
| Properties | |||
| C21H23NO3 | |||
| Molar mass | 337.41 g/mol | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
3-Quinuclidinyl benzilate (QNB) — IUPAC name 1-azabicyclo[2.2.2]oct-3-yl 2-hydroxy-2,2-diphenylacetate; US Army code EA-2277; NATO code BZ; Soviet code Substance 78 — is an odorless military incapacitating agent.[1]
BZ is a glycolate anticholinergic compound related to atropine, scopolamine, hyoscyamine, and other deliriants. Dispersal would be as an aerosolized solid (primarily for inhalation) or as agent dissolved in one or more solvents for ingestion or percutaneous absorption.
Acting as a competitive inhibitor of acetylcholine at postsynaptic and postjunctional muscarinic receptor sites in smooth muscle, exocrine glands, autonomic ganglia, and the brain, BZ decreases the effective concentration of acetylcholine seen by receptors at these sites. Thus, BZ causes PNS effects that in general are the opposite of those seen in nerve agent poisoning. CNS effects include stupor, confusion, and confabulation with concrete and panoramic illusions and hallucinations, and with regression to primitive, involuntary behaviors such as floccillation and disrobing.
Physostigmine, which increases the concentration of acetylcholine in synapses and in neuromuscular and neuroglandular junctions, is a specific antidote.
Production of BZ is controlled under schedule 2 of the Chemical Weapons Convention of 1997.
History
Invention and research
BZ was invented by the Swiss pharmaceutical company Hoffman-LaRoche in 1951.[2] The company was investigating anti-spasmodic agents, similar to tropine, for treating gastrointestinal ailments when the chemical was discovered.[2] It was then investigated for possible use in ulcer treatment, but was found unsuitable. At this time the United States military investigated it along with a wide range of possible nonlethal, psychoactive incapacitating agents including psychedelic drugs such as LSD and THC, dissociative drugs such as ketamine and phencyclidine, potent opioids such as fentanyl, as well as several glycolate anticholinergics.[3][4] By 1959 the United States Army showed significant interest in deploying it as a chemical warfare agent.[2] It was originally designated "TK", but when it was standardized by the Army in 1961 it received the NATO code name "BZ".[2] The agent commonly became known as "Buzz" because of this abbreviation and the effects it had on the mental state of the human volunteers intoxicated with it in research studies at Edgewood Arsenal in Maryland.[2] As described in retired Army psychiatrist James Ketchum’s autobiographical book Chemical Warfare: Secrets Almost Forgotten (2006), work proceeded in 1964 when a general envisioned a scheme to incapacitate an entire trawler with aerosolized BZ; this effort was dubbed Project DORK.[5] BZ was ultimately weaponized for delivery in the M44 generator cluster and the M43 cluster bomb, until all such stocks were destroyed in 1989 as part of a general downsizing of the US chemical warfare program.
Alleged use
In February 1998, the British Ministry of Defence accused Iraq of having stockpiled large amounts of a glycolate anticholinergic incapacitating agent known as Agent 15.[6] Agent 15 is an alleged Iraqi incapacitating agent that is likely to be chemically either identical to BZ or closely related to it. Agent 15 was reportedly stockpiled in large quantities prior to and during the Persian Gulf War. However after the war the CIA concluded that Iraq had not stockpiled or weaponised Agent 15.[7][8]
In January 2013, an unidentified U.S. administration official, referring to an undisclosed U.S. State Department cable, claimed that "Syrian contacts made a compelling case that Agent 15, a hallucinogenic chemical similar to BZ,[9] was used in Homs".[10] However in response to these reports U.S. National Security Council spokesman stated "The reporting we have seen from media sources regarding alleged chemical weapons incidents in Syria has not been consistent with what we believe to be true about the Syrian chemical weapons program".[8][11] The chemical was also allegedly used in the August 2013 Ghouta attacks.[12]
Fictional use
In the film Jacob's Ladder, the eponymous character is told that the horrific events he experienced on his final day in Vietnam were the product of an experimental drug called "the Ladder", which was used on troops without their knowledge. At the end of the film, a message is displayed saying that reports of testing of BZ on U.S. soldiers during the Vietnam War were denied by the Pentagon. Director Adrian Lyne said a part of the inspiration for this motif was Martin A. Lee's book Acid Dreams: The CIA, LSD and Sixties Rebellion, but noted that "nothing in the book suggests that the drug BZ—a super-hallucinogen that has a tendency to elicit maniac behavior—was used on U.S. troops.".[13]
Physiochemical characteristics
BZ is odorless. It is stable in most solvents, with a half-life of three to four weeks in moist air; even heat-producing munitions can disperse it. It is extremely persistent in soil and water and on most surfaces. It is slightly soluble in water; soluble in dilute acids, trichloroethylene, dimethylformamide, most organic solvents, insoluble with aqueous alkali.[14]
Detection and protection
BZ is odorless and nonirritating with delayed symptoms several hours after contact.[1] In the field the only immediate indications of its use may be the white smoke emanating from delivered weapons. Though detection methods have been developed for BZ, these have not been standardized for field use and are limited to laboratory analysis or specialized monitoring in industrial facilities.
Protection from BZ means blocking it from entry into the body. At dosages adequate for a lung effect there is little risk of absorption through the skin or contact hazards from aerosols that have settled out onto surfaces. The amount of BZ that may settle out on surfaces from an aerosol is too small to represent a hazard from secondary aerosols. Therefore, the most appropriate protective response is to don a protective mask with a good quality aerosol filter. Even improvised respiratory protection (e.g., several folded pieces of cloth over the nose and mouth) may render BZ employment ineffectual.
There is the possibility that BZ could be employed for a skin effect by adding to a skin penetrating solvent, or used for a secondary aerosol through contaminating terrain with bulk micro-pulverized BZ. However, both of these employment schemes are unlikely owing to the high cost and uncontrolled dose (potentially lethal). In any situation where BZ is present in liquid or bulk powder form, adequate skin protection with impermeable protective clothing and gloves is warranted.
Toxicokinetics
BZ is dispersed as an aerosol. It may be micropulverized for dissemination by a disperser (90% dissemination efficiency), or mixed with a pyrotechnic burning mixture for dissemination in burning munitions (70% dissemination efficiency). Alternatively, it may be dissolved in a solvent such as DMSO to enhance percutaneous absorption, though experiments before this proved unsatisfactory for military purposes.
Bioavailability via ingestion and by inhalation of particles 1 micrometer in size approximates 80%, and 40 to 50%, respectively, of a parenterally delivered dose of BZ. Percutaneous absorption of BZ dissolved in propylene glycol yields, after a latent period of up to 24 hours, serum levels approximately 5 to 10% of those achieved with intravenous or intramuscular administration.
Following absorption, BZ is systemically distributed to most organs and biological tissues of the body. Its ability to reach synapses and neuromuscular and neuroglandular junctions throughout the body is responsible for its PNS effects, whereas its ability to cross the blood–brain barrier confers upon it the ability to cause CNS effects. Atropine and hyoscyamine both cross the placenta and can be found in small quantities in breast milk; whether this is also true for BZ is unclear.
Metabolism of BZ would be expected to occur primarily in the liver, with elimination of unchanged agent and metabolites chiefly in the urine.
Toxicity
The characteristic that makes BZ an incapacitating rather than a toxic chemical warfare agent is its high safety margin (ICt50/LCt50) of around 40-fold (range 32 to 384 fold). It has an ID50 of 0.00616 mg per person (i.v.) with a probit slope of 9.2. The respiratory ICt50 (median incapacitating dosage) for BZ is 110 mg·min/m³ (mild activity—15 l/min rate of breathing), whereas the LCt50 is often estimated to be around 3,800–41,300 mg·min/m³.[15]
Toxicodynamics (mechanism of action)
The agent BZ and other anticholinergic glycolates act as competitive inhibitors of the neurotransmitter acetylcholine neurons (1) at postjunctional muscarinic receptors in cardiac and smooth muscle and in exocrine (ducted) glands and (2) at postsynaptic receptors in neurons. As the concentration of BZ at these sites increases, the proportion of receptors available for binding to acetylcholine decreases and the end organ "sees" less acetylcholine. (One way of visualizing this process is to imagine BZ coating the surface of the end organ and preventing acetylcholine from reaching its receptors.) Because BZ has little to no agonist activity with respect to acetylcholine, high concentrations of BZ essentially substitute a "dud" for acetylcholine at these sites and lead to clinical effects reflective of understimulation of end organs.
See also
- Benactyzine—Also made from Benzilic acid, but esterified with 2-(diethylamino)ethanol instead of 3-Quinuclidinol.
References
- 1 2 QNB: Incapacitating Agent. Emergency Response Safety and Health Database. National Institute for Occupational Safety and Health. Accessed April 20, 2009.
- 1 2 3 4 5 Kirby, Reid. "Paradise Lost: The Psycho Agents", The CBW Conventions Bulletin, May 2006, Issue no. 71, pp. 2-3, accessed December 11, 2008.
- ↑ Possible Long-Term Health Effects of Short-Term Exposure To Chemical Agents, Volume 2: Cholinesterase Reactivators, Psychochemicals and Irritants and Vesicants. (1984)
- ↑ Ketchum - Chemical Warfare: Secrets Almost Forgotten (2006)
- ↑ Army’s Hallucinogenic Weapons Unveiled
- ↑ Iraqi `zombie gas' arsenal revealed
- ↑ Intelligence Update: Chemical Warfare Agent Issues (Report). CIA. April 2002. Retrieved 28 January 2013.
We assess that Iraq never went beyond research with Agent 15—a hallucinogenic chemical similar to BZ—or any other psychochemical. Agent 15 became an issue after a 9 February 1998 British press release claimed that the UK had information, thought to be reliable, that Iraq had large quantities of this chemical agent in the 1980s. UNSCOM and intelligence information indicated that Iraq researched a number of psychochemicals, including Agent 15, BZ, and PCP; however, UNSCOM indicated it saw no evidence of Iraqi importation of large quantities, weaponization, procurement of militarily significant quantities of precursors, or industrial production of these agents
- 1 2 Jeffrey Lewis (25 January 2013). "Why everyone's wrong about Assad's zombie gas". Foreign Policy. Retrieved 28 January 2013.
- ↑ Intelligence Update: Chemical Warfare Agent Issues
- ↑ Rogin, Josh (15 January 2013). "Secret State Department cable: Chemical weapons used in Syria". Foreign Policy The Cable. Retrieved 16 January 2013.
- ↑ "U.S. plays down media report that Syria used chemical weapons". Reuters. 16 January 2013. Retrieved 28 January 2013.
- ↑ "Suffering in Syria is clear, but cause and culprits are murky". Edition.cnn.com. Retrieved 2013-08-24.
- ↑ Hartl, John (1990-11-01). "Adrian Lyne Met A Metaphysical Challenge". The Seattle Times. Retrieved 2010-02-06.
- ↑ US Army FM 3-9
- ↑
- Goodman E (2010). Ketchum J, Kirby R, ed. Historical Contributions to the Human Toxicology of Atropine. Eximdyne. p. 120. ISBN 978-0-9677264-3-4.
-
 This article incorporates public domain material from websites or documents of the United States Army.
This article incorporates public domain material from websites or documents of the United States Army.
External links
- Erowid—BZ Vault
- Tear Gases and Chemical Agents—Agent BZ
- eMedicine—Incapacitating Agents: 3-Quinuclidinyl Benzilate
- Possible Abuse of BZ by Insurgents in Iraq
- Center for Disease Control—BZ Incapacitating Agent
- Paradise Lost: The Psycho Agents by Reid Kirby
- Department of Defense—Agent BZ use in Hawaii April through June 1966.
- Quinuclidinyl benzilate
| ||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||