Carbon-13
| Carbon-13 | |
|---|---|
| General | |
| Name, symbol | Carbon-13,13C |
| Neutrons | 7 |
| Protons | 6 |
| Nuclide data | |
| Natural abundance | 1.109% |
| Isotope mass | 13.003355[1] u |
| Spin | −1⁄2 |
Carbon-13 (13C) is a natural, stable isotope of carbon with a nucleus containing 6 protons and 7 neutrons. One of the environmental isotopes, it makes up about 1.07% of all natural carbon on Earth.[2]
Detection by mass spectrometry
A mass spectrum of an organic compound will usually contain a small peak of one mass unit greater than the apparent molecular ion peak (M) of the whole molecule. This is known as the M+1 peak and comes from the handful of molecules that contain a 13C atom in place of a 12C. A molecule containing one carbon atom will be expected to have an M+1 peak of approximately 1.1% of the size of the M peak, as 1.1% of the molecules will have a 13C rather than a 12C. Similarly, a molecule containing two carbon atoms will be expected to have an M+1 peak of approximately 2.2% of the size of the M peak, as there is double the previous likelihood that any molecule will contain a 13C atom.
In the above, the mathematics and chemistry have been simplified, however it can be used effectively to give the number of carbon atoms for small- to medium-sized organic molecules. In the following formula the result should be rounded to the nearest integer:
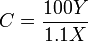
C = number of C atoms X = amplitude of the M ion peak Y = amplitude of the M+1 ion peak
13C-enriched compounds are used in the research of metabolic processes by means of mass spectrometry. Such compounds are safe because they are non-radioactive. In addition, 13C is used to quantify proteins (quantitative proteomics). One important application is in "Stable isotope labeling with amino acids in cell culture" (SILAC). 13C-enriched compounds are used in medical diagnostic tests such as the urea breath test. Analysis in these tests is usually of the ratio of 13C to 12C by Isotope ratio mass spectrometry.
The ratio of 13C to 12C is slightly higher in plants employing C4 carbon fixation than in plants employing C3 carbon fixation. Because the different isotope ratios for the two kinds of plants propagate through the food chain, it is possible to determine if the principal diet of a human or other animal consists primarily of C3 plants or C4 plants by measuring the isotopic signature of their collagen and other tissues. Deliberate increase of proportion of 13C in diet is the concept of i-food, a proposed way to increase longevity.
Uses in science
Due to differential uptake in plants as well as marine carbonates of 13C, it is possible to use these isotopic signature in earth science. In aqueous geochemistry, by analyzing the δ13C value of surface and ground waters the source of the water can be identified. This is because atmospheric, carbonate, and plant derived δ13C values all differ with respect to Pee Dee Belemnite (PDB) standard.
To calculate δ13C:
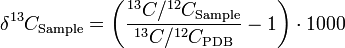
The same calculation can be used to distinguish between biomass produced by C3 and C4 metabolic pathways (e.g. trees versus tropical grasses), which allows for spatiotemporal reconstruction of species and ecosystems range shifts based on the isotopic characterization of soil organic matter.[3][4][5][6][7]
Within C3 plants processes regulating changes in δ13C are well understood, particularly at the leaf level,[8] but also during wood formation.[9][10] Many recent studies combine leaf level isotopic fractionation with annual patterns of wood formation (i.e. tree ring δ13C) to quantify the impacts of climatic variations and atmospheric composition on physiological processes of individual trees and forest stands.[11][12][13] The next phase of understanding, in terrestrial ecosystems at least, seems to be the combination of multiple isotopic proxies to decipher interactions between plants, soils and the atmosphere, and predict how changes in land use will affect climate change.[14][15][16]
Carbon-13 has a non-zero spin quantum number of 1/2, and hence allows the structure of carbon-containing substances to be investigated using Carbon-13 nuclear magnetic resonance.
See also
Notes
- ↑ http://www.sisweb.com/referenc/source/exactmas.htm
- ↑ Carbon
- ↑ SILVA L.C.R, GIORGIS M., ANAND M., ENRICO L., PÉREZ-HARGUINDEGUY N., FALCZUK V., TIESZEN L.L., CABIDO M. (2011) Evidence of shift in C4 species range in central Argentina during the Late Holocene. Plant & Soil 349: 261-279.
- ↑ SILVA L.C.R., STERNBERG L. da S.L., HAIDAR R.F., VALE G.D. (2010) Deciphering earth mound origins in central Brazil. Plant & Soil 336: 3–14
- ↑ SILVA L.C.R., HARIDASAN M., STERNBERG L.S.L., FRANCO A.C., HOFFMANN W.A. (2010) Not all forests are expanding into central Brazilian savannas. Plant & Soil 333: 431-443.
- ↑ SILVA L.C.R., ANAND M. (2011) Mechanisms of Araucaria (Atlantic) forest expansion into southern Brazilian grasslands. Ecosystems 14: 1354-1371.
- ↑ SILVA L.C.R., STERNBERG L.S.L., HARIDASAN M., HOFFMANN W.A., MIRALLES-WILHELM F., FRANCO A.C. (2008) Expansion of gallery forests into central Brazilian savannas. Global Change Biology 14:1-11.
- ↑ Farquhar, G.D., Ehleringer, J.R. & Hubick, K.T. (1989) Carbon isotope discrimination and photosynthesis. Annual Review of Plant Physiology and Plant Molecular Biology, 40, 503-537.
- ↑ McCarroll D, Loader NJ (2004) Stable isotopes in tree rings. Quaternary Science Reviews 23: 771–801 doi:10.1016/j.quascirev.2003.06.017. . doi: 10.1016/j.quascirev.2003.06.017.
- ↑ Ewe SML, Da Silveira Lobo Sternberg L, Busch DE (1999) Water-use patterns of woody species in pineland and hammock communities of South Florida. Forest Ecology and Management 118: 139–148 doi:10.1016/S0378-1127(98)00493-9. . doi: 10.1016/S0378-1127(98)00493-9.
- ↑ SILVA L.C.R., ANAND M., OLIVEIRA J.M., PILLAR V.D. (2009) Past century changes in Araucaria angustifolia (Bertol.) Kuntze water use efficiency and growth in forest and grassland ecosystems of southern Brazil: implications for forest expansion. Global Change Biology 15: 2387–2396.
- ↑ SILVA L.C.R., ANAND M. (2013) Probing for the influence of atmospheric CO2 and climate change on forest ecosystems across biomes. Global Ecology and Biogeography.DOI:10.1111/j.1466-8238.2012.00783.x
- ↑ SILVA L.C.R., HORWATH W.R. (2013) Explaining global changes in water use efficiency: Why have we overestimated forest responses to rising atmospheric CO2? PLoS One 8(1): e53089. doi:10.1371/journal.pone.0053089
- ↑ GÓMEZ-GUERRERO A., SILVA L.C.R., HORWATH W.R., BARRERA-REYES M., KISHCHUK B., VELÁZQUEZ-MARTÍNEZ A., TRINIDAD-HERNÁNDEZ T., PLASCENCIA-ESCALANTE O. (2013) Growth decline and divergent tree-ring isotopic composition (δ13C and δ18O) contradict predictions of CO2 stimulation in high altitudinal forests. Global Change Biology. DOI: 10.1111/gcb.12170
- ↑ ROSSATTO D.R., HOFFMANN W.A., SILVA L.C.R., HARIDASAN M., STERNBERG L.S.L., FRANCO A.C. (2013) Seasonal variation in leaf traits between congeneric savanna and forest trees in Central Brazil: implications for forest expansion into savanna. Trees DOI 10.1007/s00468-013-0864-2
- ↑ SILVA L.C.R., CORRÊA R.S., DOANE T., PEREIRA E., HORWATH W.R. (2013) Unprecedented carbon accumulation in mined soils: the synergistic effect of resource input and plant species invasion. Ecological Applications. http://dx.doi.org/10.1890/12-1957.1
| Lighter: carbon-12 |
Carbon-13 is an isotope of carbon |
Heavier: carbon-14 |
| Decay product of: boron-13, nitrogen-13 |
Decay chain of carbon-13 |
Decays to: stable |