Yield (chemistry)
In chemistry, yield, also referred to as reaction yield, is the amount of product obtained in a chemical reaction.[1] The absolute yield can be given as the weight in grams or in moles (molar yield). The percentage yield (or fractional yield or relative yield), which serves to measure the effectiveness of a synthetic procedure, is calculated by dividing the amount of the desired product obtained by the theoretical yield (the unit of measure for both must be the same):
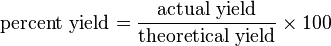
The theoretical yield is the amount predicted by a stoichiometric calculation based on the number of moles of all reactants present. This calculation assumes that only one reaction occurs and that the limiting reactant reacts completely. However the actual yield is very often smaller (the percent yield is less than 100%) for several reasons:[2][3]
- Many reactions are incomplete and the reactants are not completely converted to products. If a reverse reaction occurs, the final state contains both reactants and products in a state of chemical equilibrium.
- Two or more reactions may occur simultaneously, so that some reactant is converted to undesired by-products.
- Losses occur in the separation and purification of the desired product from the reaction mixture.
The ideal or theoretical yield of a chemical reaction would be 100%. According to Vogel's Textbook of Practical Organic Chemistry, yields around 100% are called quantitative, yields above 90% are called excellent, yields above 80% are very good, yields above 70% are good, yields above 50% are fair, and yields below 40% are called poor.[1] It should however be noted that these names are arbitrary and not universally accepted, and for many reactions these expectations may be unrealistically high. Yields may appear to be above 100% when products are impure, as the measured weight of the product will include the weight of any impurities.[3] Purification steps always lower the yield and the reported yields usually refer to the yield of the final purified product.
When more than one reactant participates in a reaction, the yield is usually calculated based on the amount of the limiting reactant, whose amount is less than stoichiometrically equivalent (or just equivalent) to the amounts of all other reactants present. Other reagents present in amounts greater than required to react with all the limiting reagent present are considered excess. As a result the yield should not be automatically taken as a measure for reaction efficiency.
Example
This is an example of an esterification reaction where one molecule acetic acid reacts with one molecule ethanol, yielding one molecule ethyl acetate (a bimolecular second-order reaction of the type A + B → C):
- 120 g acetic acid (60 g/mol, 2.0 mol) was reacted with 230 g ethanol (46 g/mol, 5.0 mol), yielding 132 g ethyl acetate (88 g/mol, 1.5 mol). The yield was 75%.
- The molar amount of the reactants is calculated from the weights (acetic acid: 120 g ÷ 60 g/mol = 2.0 mol; ethanol: 230 g ÷ 46 g/mol = 5.0 mol).
- Ethanol is used in a 2.5-fold excess (5.0 mol ÷ 2.0 mol).
- The theoretical molar yield is 2.0 mol (the molar amount of the limiting compound, acetic acid).
- The molar yield of the product is calculated from its weight (132 g ÷ 88 g/mol = 1.5 mol).
- The % yield is calculated from the actual molar yield and the theoretical molar yield (1.5 mol ÷ 2.0 mol × 100% = 75%).
See also
References
- ↑ 1.0 1.1 Vogel, A.I., Tatchell, A.R., Furnis, B.S., Hannaford, A.J. and P.W.G. Smith. Vogel's Textbook of Practical Organic Chemistry, 5th Edition. Prentice Hall, 1996. ISBN 0582462363.
- ↑ Whitten, K.W., Gailey, K.D. and Davis, R.E. General Chemistry, 4th Edition. Saunders College Publishing, 1992. ISBN 0030723736. p.95
- ↑ 3.0 3.1 Petrucci, R.H., Harwood, W.S. and Herring, F.G. General Chemistry, 8th Edition. Prentice Hall, 2002 ISBN 0130143294. p.125