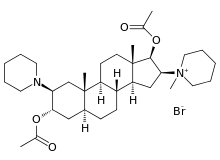
Vecuronium bromide
Vecuronium bromide|
|
| Systematic (IUPAC) name |
|---|
|
[(2S,3S,5S,8R,9S,10S,13S,14S,16S,17S)-17-Acetyloxy-10,13-dimethyl-16-(1-methyl-3,4,5,6-tetrahydro-2H-pyridin-1-yl)-2-(1-piperidyl)-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-yl] acetate bromide |
| Clinical data |
|---|
| AHFS/Drugs.com |
monograph |
|---|
|
- US: C (Risk not ruled out)
-
|
|---|
|
|
|---|
|
Intravenous |
|---|
| Pharmacokinetic data |
|---|
| Bioavailability |
100% (IV) |
|---|
| Metabolism |
liver 30% |
|---|
| Half-life |
51–80 minutes (longer with renal failure) |
|---|
| Excretion |
Fecal (40-75%) and renal (30% as unchanged drug and metabolites) |
|---|
| Identifiers |
|---|
|
50700-72-6  Y Y |
|---|
|
M03AC03 |
|---|
| PubChem |
CID 39764 |
|---|
| UNII |
7E4PHP5N1D  N N |
|---|
| ChEMBL |
CHEMBL1201219  N N |
|---|
| Chemical data |
|---|
| Formula |
C34H57BrN2O4 |
|---|
|
637.73 g/mol |
|---|
 N (what is this?) (verify) N (what is this?) (verify) |
|---|
Vecuronium bromide (Norcuron) is a muscle relaxant in the category of non-depolarizing blocking agents. Vecuronium bromide is indicated as an adjunct to general anesthesia, to facilitate endotracheal intubation and to provide skeletal muscle relaxation during surgery or mechanical ventilation. Although vecuronium bromide is often thought of as a muscle relaxant, it may be more accurate to classify it as a paralyzing agent. It is commercially available as ampoules containing 4 or 10 mg of the drug in powder form which needs to be dissolved in distilled water prior to administration to the patient.
Development
Vecuronium's structure is derived from the same aminosteroid structure as pancuronium, but it is missing the methyl group on the piperidine nitrogen that is attached to the 'A' ring, making it monoquaternary, like D-tubocurarine. It has the same configuration at all ten stereocentres as pancuronium, and is a single-isomer preparation.
Clinical pharmacology
Vecuronium operates by competing for the cholinoceptors at the motor end plate thereby exerting its muscle-relaxing properties which are used adjunctively to general anesthesia. Under balanced anesthesia, the time to recovery to 25% of control (clinical duration) is approximately 25 to 40 minutes after injection and recovery is usually 95% complete approximately 45 to 65 minutes after injection of intubating dose. The neuromuscular blocking action of vecuronium is slightly enhanced in the presence of potent inhalation anesthetics. If vecuronium is first administered more than 5 minutes after the start of the inhalation of enflurane, isoflurane, or halothane, or when a steady state has been achieved, the intubating dose of vecuronium may be decreased by approximately 15%.
Non-medical usage
Vecuronium bromide can be used as part of a drug cocktail that prisons in the United States use as a means to put a condemned prisoner to death. Vecuronium bromide is used to paralyze the prisoner and stop his or her breathing, in conjunction with a sedative and potassium chloride to stop the prisoner's heart. Injections of vecuronium bromide without proper sedation allow the person to be fully awake but unable to move in response to pain.[1]
In 2001, Japanese nurse Daisuke Mori was reported to have murdered 10 patients using vecuronium bromide.[2] He was convicted of murder and was sentenced to life imprisonment.[3]
References
|
|---|
| Peripherally acting
(primarily antinicotinic,
NMJ block) | Non-depolarizing | |
|---|
| Depolarizing | |
|---|
| | |
|---|
|
|---|
| | Centrally acting | | |
|---|
| Benzodiazepines | |
|---|
| Nonbenzodiazepines | |
|---|
| Thienodiazepines | |
|---|
| Quinazolines | |
|---|
| Anticholinergics
(Antimuscarinics) | |
|---|
| Other | |
|---|
|
|---|
| | Directly acting | |
|---|
| |
|---|
| | Description |
- Anatomy
- head
- neck
- arms
- chest and back
- diaphragm
- abdomen
- genital area
- legs
- Muscle tissue
- Physiology
|
|---|
| | Disease |
- Myopathy
- Soft tissue
- Connective tissue
- Congenital
- abdomen
- muscular dystrophy
- Neoplasms and cancer
- Injury
- Symptoms and signs
|
|---|
| | Treatment |
- Procedures
- Drugs
- anti-inflammatory
- muscle relaxants
|
|---|
|
|
|
|---|
| | |
|---|
| | mACh |
- Antagonists: 3-Quinuclidinyl benzilate
- 4-DAMP
- Aclidinium bromide
- Anisodamine
- Anisodine
- Antihistamines (first-generation) (e.g., brompheniramine, chlorphenamine, cyproheptadine, dimenhydrinate, diphenhydramine, doxylamine, mepyramine (pyrilamine), phenindamine, pheniramine, promethazine, tripelennamine, triprolidine)
- Atropine
- Atropine methonitrate
- Atypical antipsychotics (e.g., clozapine, olanzapine, quetiapine, zotepine)
- Benactyzine
- Benzatropine (benztropine)
- Benzydamine
- BIBN 99
- Biperiden
- Bornaprine
- CAR-226,086
- CAR-301,060
- CAR-302,196
- CAR-302,282
- CAR-302,368
- CAR-302,537
- CAR-302,668
- CS-27349
- Cyclobenzaprine
- Cyclopentolate
- Darifenacin
- DAU-5884
- Dimethindene
- Dexetimide
- DIBD
- Dicyclomine (dicycloverine)
- Ditran
- EA-3167
- EA-3443
- EA-3580
- EA-3834
- Etanautine
- Etybenzatropine (ethybenztropine)
- Flavoxate
- Himbacine
- HL-031,120
- Ipratropium bromide
- J-104,129
- Hyoscyamine
- Mamba toxin 3
- Mamba toxin 7
- Mazaticol
- Mebeverine
- Methoctramine
- Metixene
- N-Ethyl-3-piperidyl benzilate
- N-Methyl-3-piperidyl benzilate
- Orphenadrine
- Otenzepad
- Oxybutynin
- PBID
- PD-102,807
- PD-0298029
- Phenglutarimide
- Phenyltoloxamine
- Pirenzepine
- Piroheptine
- Procyclidine
- Profenamine
- RU-47,213
- SCH-57,790
- SCH-72,788
- SCH-217,443
- Scopolamine (hyoscine)
- Solifenacin
- Telenzepine
- Tetracyclic antidepressants (e.g., amoxapine, maprotiline, mianserin, mirtazapine)
- Tiotropium bromide
- Tolterodine
- Tricyclic antidepressants (e.g., amitriptyline, butriptyline, clomipramine, desipramine, dosulepin (dothiepin), doxepin, imipramine, lofepramine, nortriptyline, protriptyline, trimipramine)
- Trihexyphenidyl
- Tripitamine
- Tropatepine
- Tropicamide
- Typical antipsychotics (e.g., chlorpromazine, loxapine, thioridazine)
- WIN-2299
- Xanomeline
- Zamifenacin
|
|---|
| | nACh |
- Agonists: 5-HIAA
- A-84,543
- A-366,833
- A-582,941
- A-867,744
- ABT-202
- ABT-418
- ABT-560
- ABT-894
- Acetylcholine
- Altinicline
- Anabasine
- Anatoxin-a
- AR-R17779
- Butinoline
- Butyrylcholine
- Carbachol
- Choline
- Cotinine
- Cytisine
- Decamethonium
- Desformylflustrabromine
- Dianicline
- Dimethylphenylpiperazinium
- Epibatidine
- Epiboxidine
- Ethanol
- Ethoxysebacylcholine
- EVP-4473
- EVP-6124
- Galantamine
- GTS-21
- Ispronicline
- Ivermectin
- Levamisole
- Lobeline
- MEM-63,908 (RG-3487)
- Morantel
- Nicotine (tobacco)
- NS-1738
- PHA-543,613
- PHA-709,829
- PNU-120,596
- PNU-282,987
- Pozanicline
- Rivanicline
- RJR-2429
- Sazetidine A
- SB-206553
- Sebacylcholine
- SIB-1508Y
- SIB-1553A
- SSR-180,711
- Suberyldicholine
- Suxamethonium (succinylcholine)
- TC-1698
- TC-1734
- TC-1827
- TC-2216
- TC-5214
- TC-5619
- TC-6683
- Tebanicline
- Tropisetron
- UB-165
- Varenicline
- WAY-317,538
- XY-4083
|
|---|
|
| | | | | |
|---|
| | ChAT |
- 1-(-Benzoylethyl)pyridinium
- 2-(α-Naphthoyl)ethyltrimethylammonium
- 3-Chloro-4-stillbazole
- 4-(1-Naphthylvinyl)pyridine
- Acetylseco hemicholinium-3
- Acryloylcholine
- AF64A
- B115
- BETA
- CM-54,903
- N,N-Dimethylaminoethylacrylate
- N,N-Dimethylaminoethylchloroacetate
|
|---|
| | AChE | |
|---|
| | BChE |
Note: Many of the AChE inhibitors listed above also act as BChE inhibitors.
|
|---|
|
| | | | |
|---|
| | Description |
- Anatomy
- meninges
- cortex
- association fibers
- commissural fibers
- lateral ventricles
- basal ganglia
- diencephalon
- mesencephalon
- pons
- cerebellum
- medulla
- spinal cord
- Physiology
- Development
|
|---|
| | Disease |
- Cerebral palsy
- Meningitis
- Demyelinating diseases
- Seizures and epilepsy
- Headache
- Stroke
- Sleep
- Congenital
- Injury
- Neoplasms and cancer
- Other
- Symptoms and signs
- head and neck
- eponymous
- lesions
- Tests
|
|---|
| | Treatment |
- Procedures
- Drugs
- general anesthetics
- analgesics
- addiction
- epilepsy
- cholinergics
- migraine
- Parkinson's
- vertigo
- other
|
|---|
|
|