Tin(IV) sulfide
 | |
| Names | |
|---|---|
| IUPAC name
Tin(IV) sulfide | |
| Other names
Tin disulfide, Stannic sulfide, Mosaic gold | |
| Identifiers | |
| 1315-01-1 | |
| ChEBI | CHEBI:50886 |
| EC-number | 215-252-9 |
| |
| Jmol-3D images | Image Image (S=Sn=S) |
| PubChem | 73977 15238661 (S=Sn=S) |
| |
| UNII | YVY89V9BUH |
| Properties | |
| Molecular formula |
S2Sn |
| Molar mass | 182.84 g·mol−1 |
| Appearance | Gold-yellow powder |
| Odor | Odorless |
| Density | 4.5 g/cm3[1] |
| Melting point | 600 °C (1,112 °F; 873 K) decomposes[1] |
| Insoluble | |
| Solubility | Soluble in aq. alkalis, decompose in aqua regia[1] Insoluble in alkyl acetates, acetone[2] |
| Structure | |
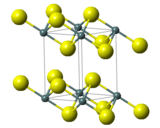
| Crystal structure | Rhombohedral, hP3[3] |
| Space group | P3m1, No. 164[3] |
| Point group | 3 2/m[3] |
| Lattice constant | a = 3.65 Å, c = 5.88 Å[3] |
| Lattice constant | α = 90°, β = 90°, γ = 120° |
| Octahedral (Sn4+)[3] | |
| Hazards | |
| GHS pictograms |  [4] [4] |
| GHS signal word | Warning |
| H302, H312, H315, H319, H332, H335[4] | |
| P261, P280, P301+312, P302+352, P304+340, P305+351+338, P332+313[4] | |
| NFPA 704 | |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Tin(IV) sulfide is a compound with the formula SnS2. The compound crystallizes in the cadmium iodide motif, with the Sn(IV) situated in "octahedral holes' defined by six sulfide centers.[5] It occurs naturally as the rare mineral berndtite.[6] It is useful as semiconductor material with band gap 2.2 eV.
Reactions
The compound precipitates as a brown solid upon the addition of H2S to solutions of tin(IV) species. This reaction is reversed at low pH. Crystalline SnS2 has a bronze colour and is used in decorative coating[7] where it is known as mosaic gold.
The material also reacts with sulfide salts to give a series of thiostannates with the formula [SnS2]m[S]n2n−. A simplified equation for this depolymerization reaction is
- SnS2 + S2− → 1/x{SnS32−}x.
References
- ↑ 1.0 1.1 1.2 Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ↑ Comey, Arthur Messinger; Hahn, Dorothy A. (1921-02). A Dictionary of Chemical Solubilities: Inorganic (2nd ed.). New York: The MacMillan Company. p. 1080. Check date values in:
|date=(help) - ↑ 3.0 3.1 3.2 3.3 3.4 Voort, G.F. Vander, ed. (2004). "Crystal Structure*" (PDF). ASM Handbook (ASM International) 9 (Metallography and Microstructures): 29–43. doi:10.1361/asmhba0003722 (inactive 2015-02-01).
- ↑ 4.0 4.1 4.2
- ↑ Wells, A.F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press. ISBN 0-19-855370-6.
- ↑ Vaughan, D. J.; Craig, J. R. "Mineral Chemistry of Metal Sulfides" Cambridge University Press, Cambridge: 1978. ISBN 0-521-21489-0.
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
External links
| Wikimedia Commons has media related to Tin(IV) sulfide. |
| ||||||||||
