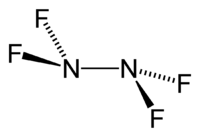
Tetrafluorohydrazine
 | |
 | |
| Names | |
|---|---|
| IUPAC name
1,1,2,2-tetrafluorohydrazine | |
| Other names
dinitrogen tetrafluoride, perfluorohydrazine, UN 1955 | |
| Identifiers | |
| 10036-47-2 | |
| ChemSpider | 23228 |
| |
| Jmol-3D images | Image |
| PubChem | 24845 |
| |
| Properties | |
| N2F4 | |
| Molar mass | 104.01 g mol−1 |
| Melting point | −164.5 °C (−264.1 °F; 108.6 K) [1] |
| Boiling point | −73 °C (−99 °F; 200 K)[1] |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Tetrafluorohydrazine or dinitrogen tetrafluoride, N2F4, is a colourless, reactive inorganic gas. It is a fluorinated analog of hydrazine. It is a highly hazardous chemical. In the presence of organic materials, it reacts explosively.
Tetrafluorohydrazine is manufactured from nitrogen trifluoride using an iron catalyst or iron(II) fluoride. It is used in some chemical syntheses, as a precursor or a catalyst.
Tetrafluorohydrazine was used as a high-energy liquid oxidizer in some never-flown rocket fuel formulas. In 1959 it was considered new for this application.[2]
References
- ↑ 1.0 1.1 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0080379419.
- ↑ Tetrafluorohydrazine at DTIC.mil