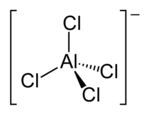
Tetrachloroaluminate
 | |
| Names | |
|---|---|
| IUPAC name
Tetrachloroaluminate(1–) | |
| Properties | |
| AlCl4− | |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Tetrachloroaluminate [AlCl4]− is an anion formed from aluminium and chlorine. The anion has a tetrahedral shape, similar to carbon tetrachloride where carbon is replaced with aluminium. Some tetrachloroaluminates are soluble in organic solvents, creating an ionic non-aqueous solution, making them suitable as component of electrolytes for batteries. E.g. lithium tetrachloroaluminate is used in some lithium batteries.