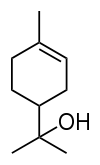
Terpineol
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-(4-Methyl-1-cyclohex-3-enyl)propan-2-ol | |||
| Other names
alpha-Terpineol α-Terpineol p-Menth-1-en-8-ol α,α,4-Trimethylcyclohex-3-ene-1-methanol Terpene alcohol | |||
| Identifiers | |||
| 98-55-5 | |||
| ChEBI | CHEBI:22469 | ||
| ChEMBL | ChEMBL507795 | ||
| ChemSpider | 13850142 | ||
| |||
| Jmol-3D images | Image | ||
| |||
| UNII | 21334LVV8W | ||
| Properties | |||
| Molecular formula |
C10H18O | ||
| Molar mass | 154.25 g·mol−1 | ||
| Density | 0.9338 g/cm3 | ||
| Melting point | 18 °C (64 °F; 291 K) | ||
| Boiling point | 219 °C (426 °F; 492 K) (81 to 82 °C at 4.5 mmHg) | ||
| Hazards | |||
| MSDS | External MSDS | ||
| NFPA 704 | |||
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |||
| | |||
| Infobox references | |||
Terpineol is a naturally occurring monoterpene alcohol that has been isolated from a variety of sources such as cajuput oil, pine oil, and petitgrain oil.[1] There are four isomers, alpha-, beta-, gamma-terpineol, and terpinen-4-ol. beta- and gamma-terpineol differ only by the location of the double bond. Terpineol is usually a mixture of these isomers with alpha-terpineol as the major constituent.
 Terpineols: alpha-, beta-, gamma-, and the 4-terpineol isomer
Terpineols: alpha-, beta-, gamma-, and the 4-terpineol isomer
Terpineol has a pleasant odor similar to lilac and is a common ingredient in perfumes, cosmetics, and flavors. α-Terpineol is one of the two most abundant aroma constituents of lapsang souchong tea; the α-terpineol originates in the pine smoke used to dry the tea.[2] (+)-α-Terpineol is a chemical constituent of scullcap.
Synthesis
Although it is naturally occurring, terpineol is commonly manufactured from the more readily available alpha-pinene.
In one study, an alternative route starting from d-limonene was demonstrated:[3]
 Terpineol synthesisfrom limonene
Terpineol synthesisfrom limonene
Limonene is reacted with trifluoroacetic acid in a Markovnikov addition to a trifluoroacetate intermediate, which is easily hydrolyzed with sodium hydroxide to α-terpineol with 76% selectivity. Side-products are the β-terpineol in a mixture of the cis-isomer, the trans-isomer, and 4-terpineol.
References
- ↑ Merck Index, 11th Edition, 9103
- ↑ Shan-Shan Yao, Wen-Fei Guo, Yi Lu, Yuan-Xun Jiang (2005). "Flavor Characteristics of Lapsang Souchong and Smoked Lapsang Souchong, a Special Chinese Black Tea with Pine Smoking Process". Journal of Agricultural and Food Chemistry 53 (22): 8688–93. doi:10.1021/jf058059i. PMID 16248572.
- ↑ Yuasa, Yoshifumi; Yuasa, Yoko (2006). "A Practical Synthesis ofd-α-Terpineol via Markovnikov Addition ofd-Limonene Using Trifluoroacetic Acid". Organic Process Research & Development 10 (6): 1231–1232. doi:10.1021/op068012d.
External links
- MSDS for alpha-terpineol
-
 Media related to Terpineol at Wikimedia Commons
Media related to Terpineol at Wikimedia Commons