TRIO (gene)
Triple functional domain protein is a protein that in humans is encoded by the TRIO gene.[1][2]
Interactions
TRIO (gene) has been shown to interact with Filamin[3] and RHOA.[4]
References
- ↑ Debant A, Serra-Pages C, Seipel K, O'Brien S, Tang M, Park SH, Streuli M (July 1996). "The multidomain protein Trio binds the LAR transmembrane tyrosine phosphatase, contains a protein kinase domain, and has separate rac-specific and rho-specific guanine nucleotide exchange factor domains". Proc Natl Acad Sci U S A 93 (11): 5466–71. doi:10.1073/pnas.93.11.5466. PMC 39269. PMID 8643598.
- ↑ "Entrez Gene: TRIO triple functional domain (PTPRF interacting)".
- ↑ Bellanger, J M; Astier C; Sardet C; Ohta Y; Stossel T P; Debant A (December 2000). "The Rac1- and RhoG-specific GEF domain of Trio targets filamin to remodel cytoskeletal actin". Nat. Cell Biol. (ENGLAND) 2 (12): 888–92. doi:10.1038/35046533. ISSN 1465-7392. PMID 11146652.
- ↑ Medley, Q G; Serra-Pagès C; Iannotti E; Seipel K; Tang M; O'Brien S P; Streuli M (November 2000). "The trio guanine nucleotide exchange factor is a RhoA target. Binding of RhoA to the trio immunoglobulin-like domain". J. Biol. Chem. (UNITED STATES) 275 (46): 36116–23. doi:10.1074/jbc.M003775200. ISSN 0021-9258. PMID 10948190.
Further reading
- Taviaux S; Diriong S; Bellanger JM et al. (1997). "Assignment of TRIO, the Trio gene (PTPRF interacting) to human chromosome bands 5p 15.1-->p 14 by in situ hybridization". Cytogenet. Cell Genet. 76 (1–2): 107–8. doi:10.1159/000134524. PMID 9154137.
- Liu X; Wang H; Eberstadt M et al. (1998). "NMR structure and mutagenesis of the N-terminal Dbl homology domain of the nucleotide exchange factor Trio". Cell 95 (2): 269–77. doi:10.1016/S0092-8674(00)81757-2. PMID 9790533.
- Seipel K; Medley QG; Kedersha NL et al. (1999). "Trio amino-terminal guanine nucleotide exchange factor domain expression promotes actin cytoskeleton reorganization, cell migration and anchorage-independent cell growth". J. Cell. Sci. 112 (12): 1825–34. PMID 10341202.
- Medley QG; Serra-Pagès C; Iannotti E et al. (2000). "The trio guanine nucleotide exchange factor is a RhoA target. Binding of RhoA to the trio immunoglobulin-like domain". J. Biol. Chem. 275 (46): 36116–23. doi:10.1074/jbc.M003775200. PMID 10948190.
- Bellanger JM; Astier C; Sardet C et al. (2001). "The Rac1- and RhoG-specific GEF domain of Trio targets filamin to remodel cytoskeletal actin". Nat. Cell Biol. 2 (12): 888–92. doi:10.1038/35046533. PMID 11146652.
- Gao Y; Xing J; Streuli M et al. (2002). "Trp(56) of rac1 specifies interaction with a subset of guanine nucleotide exchange factors". J. Biol. Chem. 276 (50): 47530–41. doi:10.1074/jbc.M108865200. PMID 11595749.
- Strausberg RL; Feingold EA; Grouse LH et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Ota T; Suzuki Y; Nishikawa T et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
- Skowronek KR, Guo F, Zheng Y, Nassar N (2004). "The C-terminal basic tail of RhoG assists the guanine nucleotide exchange factor trio in binding to phospholipids". J. Biol. Chem. 279 (36): 37895–907. doi:10.1074/jbc.M312677200. PMID 15199069.
- Zheng M; Simon R; Mirlacher M et al. (2004). "TRIO Amplification and Abundant mRNA Expression Is Associated with Invasive Tumor Growth and Rapid Tumor Cell Proliferation in Urinary Bladder Cancer". Am. J. Pathol. 165 (1): 63–9. doi:10.1016/S0002-9440(10)63275-0. PMC 1618551. PMID 15215162.
- Yoshizuka N; Moriuchi R; Mori T et al. (2004). "An alternative transcript derived from the trio locus encodes a guanosine nucleotide exchange factor with mouse cell-transforming potential". J. Biol. Chem. 279 (42): 43998–4004. doi:10.1074/jbc.M406082200. PMID 15308664.
- Gerhard DS; Wagner L; Feingold EA et al. (2004). "The Status, Quality, and Expansion of the NIH Full-Length cDNA Project: The Mammalian Gene Collection (MGC)". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928. PMID 15489334.
- Portales-Casamar E; Briançon-Marjollet A; Fromont S et al. (2006). "Identification of novel neuronal isoforms of the Rho-GEF Trio". Biol. Cell 98 (3): 183–93. doi:10.1042/BC20050009. PMID 16033331.
- Tao WA; Wollscheid B; O'Brien R et al. (2005). "Quantitative phosphoproteome analysis using a dendrimer conjugation chemistry and tandem mass spectrometry". Nat. Methods 2 (8): 591–8. doi:10.1038/nmeth776. PMID 16094384.
- Adamowicz M; Radlwimmer B; Rieker RJ et al. (2006). "Frequent amplifications and abundant expression of TRIO, NKD2, and IRX2 in soft tissue sarcomas". Genes Chromosomes Cancer 45 (9): 829–38. doi:10.1002/gcc.20343. PMID 16752383.
- Olsen JV; Blagoev B; Gnad F et al. (2006). "Global, in vivo, and site-specific phosphorylation dynamics in signaling networks". Cell 127 (3): 635–48. doi:10.1016/j.cell.2006.09.026. PMID 17081983.
- Chhatriwala MK, Betts L, Worthylake DK, Sondek J (2007). "The DH and PH Domains of Trio Coordinately Engage Rho GTPases for their Efficient Activation". J. Mol. Biol. 368 (5): 1307–20. doi:10.1016/j.jmb.2007.02.060. PMC 1890047. PMID 17391702.
- Rojas RJ; Yohe ME; Gershburg S et al. (2007). "Gαq Directly Activates p63RhoGEF and Trio via a Conserved Extension of the Dbl Homology-associated Pleckstrin Homology Domain". J. Biol. Chem. 282 (40): 29201–10. doi:10.1074/jbc.M703458200. PMC 2655113. PMID 17606614.
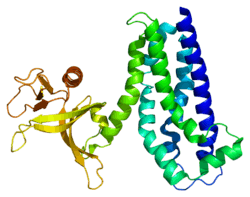
PDB gallery |
|---|
| | 1nty: Crystal structure of the first DH/PH domain of Trio to 1.7 A |
| 2nz8: N-terminal DHPH cassette of Trio in complex with nucleotide-free Rac1 |
|
|
|
|
|---|
| | | | | | |
- Biochemistry overview
- Enzymes overview
- By EC number: 1.1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 10
- 11
- 13
- 14
- 15-18
- 2.1
- 3.1
- 4.1
- 5.1
- 6.1-3
|
|
|
|