Strontium peroxide
 | |
| Identifiers | |
|---|---|
| 1314-18-7 | |
| EC number | 215-224-6 |
| |
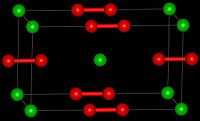
| Jmol-3D images | Image |
| PubChem | 14807 |
| |
| Properties | |
| SrO2 | |
| Molar mass | 119.619 g/mol |
| Appearance | white powder |
| Odor | odorless |
| Density | 4.56 g/cm3 (anhydrous) 1.91 g/cm3 (octahydrate) |
| Melting point | 215 °C (419 °F; 488 K) (decomposes)[1] |
| slightly soluble | |
| Solubility | very soluble in alcohol, ammonium chloride insoluble in acetone |
| Structure | |
| Crystal structure | Tetragonal [2] |
| Space group | D174h, I4/mmm, tI6 |
| 6 | |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Strontium peroxide (SrO2) is a whitish inorganic compound and a peroxide of strontium.
Uses
It is an oxidizing agent used for bleaching. It is used in some pyrotechnic compositions as an oxidizer and a vivid red pyrotechnic colorant. It can also be used as an antiseptic and in tracer munitions.
Manufacturing
Strontium peroxide is created by passing oxygen over heated strontium oxide. In a manner similar to barium peroxide, SrO2 decomposes to SrO and O2 at temperature, although this temperature is far lower than BaO2. As a consequence, SrO2 is harder to manufacture as the lower temperatures kinetically inhibit the peroxidation reaction on the atomic scale.[3]
References
- ↑ Accommodation of Excess Oxygen in Group II Monoxides - S.C. Middleburgh, R.W. Grimes, K.P.D. Lagerlof http://onlinelibrary.wiley.com/doi/10.1111/j.1551-2916.2012.05452.x/abstract
- ↑ Massalimov, I. A.; Kireeva, M. S.; Sangalov, Yu. A. (2002). Inorganic Materials 38 (4): 363. doi:10.1023/A:1015105922260. Missing or empty
|title=(help) - ↑ Accommodation of Excess Oxygen in Group II Monoxides - S.C. Middleburgh, R.W. Grimes, K.P.D. Lagerlof http://onlinelibrary.wiley.com/doi/10.1111/j.1551-2916.2012.05452.x/abstract
See also
| ||||||