Spiro compound
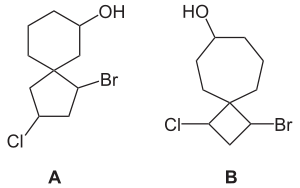
A spiro compound is a bicyclic organic compound with rings connected through just one atom. The rings can be different in nature or identical. The connecting atom is also called the spiroatom, most often a quaternary carbon ("spiro carbon"). All spiro compounds have the infix spiro followed by square brackets containing the number of atoms in the smaller ring and the number of atoms in the larger ring excluding the spiroatom itself; the numbers being separated by a dot. For example compound A is called 1-bromo-3-chlorospiro[4.5]decan-7-ol and compound B is called 1-bromo-3-chlorospiro[3.6]decan-7-ol. The spiro compound consisting of a cyclohexane ring and a cyclopentane ring is called spiro[4.5]decane. This nomenclature was proposed by Adolf von Baeyer in 1900.[1]
An example of a spiro compound with a trivial name: spiropentadiene.
Synthetic and natural spiro carbocycles attract attention of organic chemists owing to their unique structural features and numerous possible reactions in which they undergo carbon–carbon bond cleavage. For example, natural compounds such as fredericamycine, acorenone B, β-vetivone, ACAT-inhibitor, and isocomene isolated from plants contain spirocarbonatoms; the synthetic spirans include non-trivial compounds such as [5.5.5.5.]fenestrane, triangulanes, and [6.5]coronane. The properties of spiro carbocycles such as high reactivity are combined with fairly high thermal stability, and the existing synthetic difficulties usually faced by researchers when choosing the synthetic routes to target compounds stimulate many organic chemists to develop rational preparative approaches to various spiro carbocycles. There are quite a few published reviews, papers and patents that mainly present classical methods for the synthesis based on multistep synthetic strategies and expensive and exotic reagents. Meanwhile, the application of techniques of metal complex catalysis to organic and organometallic synthesis markedly facilitated the synthetic routes to spirans and made these syntheses promising for not only laboratory practice but also industry. During the last 10-15 years, considerable progress has been made, and a series of original methods and effective reagents for the preparation of various spiro carbocycles have been reported. Therefore, we attempted to give a critical account and general survey of the publications available from the literature on the synthesis of spirans using metal complex catalysts. [2]
Acetals
Acetals of cyclic ketones with diols or dithiols are spiro compounds. An example is spirapril, with a five-membered ring formed from 1,2-ethanedithiol.
Polyspiro compounds
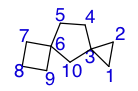

A polyspiro compound is connected by two or more spiroatoms making up three or more rings.
When naming polyspiro compounds, the prefixes di-, tri-, tetra-, etc. are first added to the front of the name to indicate the amount of spiroatoms, the single atoms between each ring. The atoms of the compound are numbered systematically. A polyspiro compound is numbered starting from a ring that holds one connecting spiroatom to another ring, i.e. a terminal ring; there can be two or more terminal rings to a polyspiro compound, so long as each of these rings has only one connecting spiroatom. In the terminal ring chosen to start the numbering, an atom of the ring next, and connected to the spiroatom, is labeled 1. Each following atom is numbered one higher than the previous—going around the compound.
Of the two or more terminal rings of any polyspiro compound, the terminal ring to start the numbering is chosen in such a way that the first spiroatom to be numbered, after numbering every other atom within the chosen ring, is the lowest possible number that can be assigned to a spiroatom. For example, suppose there is a compound where there are two terminal rings, 6 atoms in one ring, and 8 in another. The 6-atom ring is chosen to start the numbering, because after each other atom within the ring is numbered, the spiroatom is numbered 6, whereas the spiroatom of the 8-atom ring would be numbered 8. The direction of the numbering is determined by the following spiroatom (after the first numbered spiroatom of that terminal ring has been numbered/passed). If the following spiroatom in one direction assigned a number that is lower than the number it would be assigned in the opposite direction, then the direction of numbering that assigns the next spiroatom the lowest number is picked. The magnitude of the rest of the numbers given to the rest of the spiroatoms in the compound is irrelevant.
In some cases the first two, three, four, or more numbers that are assigned the same spiroatoms, in either numbering direction are the same. The first numbering direction to assign a particular spiroatom a lower number than the opposite direction assigns this spiroatom, is chosen. For example if a numbering system in one direction, for each spiroatom, is 3, 5, 7, 10; and for another direction the numbers of the spiroatoms are 3, 5, 7, 9, then the second mentioned numbering is chosen. In this case, in the first numbering direction there would be two atoms between the spiroatoms 7 and 10, and in the opposite numbering direction, there would be one atom between spiroatoms 7 and 9. The magnitude of any following spiroatom numbers, after the fourth one mentioned (numbered 9 in the first direction) in this case is irrelevant.
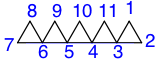
In polyspiro nomenclature the first number within the brackets is the amount of atoms starting from the atom numbered 1 to the last atom on the first ring before the first numbered spiroatom. The rest of the numbers represent the amount of atoms between the spiroatoms, or the amount of atoms at a terminal ring, going in the direction of the numbering. Each number is separated by a period. In the example to the left there are two atoms (numbered 1 and 2) before the first spiroatom (3). Between spiroatoms 3 and 4 there are zero, and likewise between 5 and 4, and 6 and 5. At the second terminal ring there are two; there is one between 6 and 5, 5 and 4, and 4 and 3. This gives the sequence [2.0.0.0.2.1.1.1]. The total number of atoms determines the acyclic alkane name that follows, in this case, undecane. For clarity, superscripts of the spiroatom numbers appear after the number of atoms that are the last to encounter the spiroatom, going in the direction of the numbering.
In the example to the right, there are two spiroatoms, so the name starts with dispiro-. In the first terminal ring, there are two atoms, showing so far the name dispiro[2. Between the next two spiroatoms there is one atom, and four at the other terminal ring, revealing the name dispiro[2.1.35. The superscript for the second spiroatom, numbered 5, was added as this is the last time it will be encountered after the three atoms at the terminal ring, following the path of numbered atoms. After adding atoms 9 and 10 between spiroatoms 3 and 5, then adding the name of the alkane that has the same amount of atoms as this spiro compound, decane, the name dispiro[2.1.35.23]decane is revealed. The superscript for spiroatom 3 is added after the atoms 9 and 10 are represented (by a 2) since after these two atoms are encountered the spiroatom 3 is encountered for the last time.
Chirality
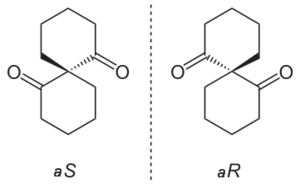
Some spiro compounds exhibit axial chirality. Spiroatoms can be centers of chirality even when they lack the required four different substituents normally observed in chirality. When two rings are identical the priority is determined by a slight modification of the CIP system assigning a higher priority to one ring extension and a lower priority to an extension in the other ring. When rings are dissimilar the regular rules apply.
Spiro forms of lactones and oxazines are frequently used as leuco dyes, frequently displaying chromism - reversible change between their colorless and color form.
References
- ↑ von Baeyer, Adolf (1900). "Systematik und Nomenclatur Bicyclischer Kohlenwasserstoffe". Berichte der deutschen chemischen Gesellschaft 33 (3): 3771–3775. doi:10.1002/cber.190003303187.
- ↑ D'yakonov, Vladimir (2014). "Metal Complex Catalysis in the Synthesis of Spirocarbocycles". Chemical Reviews 114 (11): 5775–5814. doi:10.1021/cr400291c.