Rifabutin
 | |
| Systematic (IUPAC) name | |
|---|---|
|
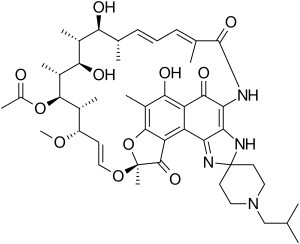
(9S,12E,14S,15R,16S,17R,18R,19R,20S, 21S,22E,24Z)-6,16,18,20-tetrahydroxy-1'- isobutyl-14-methoxy-7,9,15,17,19,21,25- hepta-methyl-spiro[9,4-(epoxypentadeca [1,11,13]trienimino)-2H-furo-[2',3':7,8]-naphth [1,2-d]imidazol-2,4'-piperidin]-5,10,26-(3H,9H)- trione-16-acetate | |
| Clinical data | |
| Trade names | Mycobutin |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a693009 |
| |
| |
| Oral | |
| Pharmacokinetic data | |
| Bioavailability | 85% |
| Protein binding | 85% |
| Metabolism | Hepatic |
| Half-life | 28 to 62 hours (mean) |
| Excretion | Renal and fecal |
| Identifiers | |
|
72559-06-9 | |
| J04AB04 | |
| PubChem | CID 6323490 |
| DrugBank |
DB00615 |
| ChemSpider |
10482168 |
| UNII |
1W306TDA6S |
| KEGG |
D00424 |
| ChEBI |
CHEBI:45367 |
| ChEMBL |
CHEMBL444633 |
| Chemical data | |
| Formula | C46H62N4O11 |
| 847.005 g/mol | |
|
SMILES
| |
| |
| | |
Rifabutin (Rfb) is a bactericidal antibiotic drug primarily used in the treatment of tuberculosis. The drug is a semi-synthetic derivative of rifamycin S. Its effect is based on blocking the DNA-dependent RNA-polymerase of the bacteria. It is effective against Gram-positive and some Gram-negative bacteria, but also against the highly resistant Mycobacteria, e.g. Mycobacterium tuberculosis, M. leprae, and M. avium intracellulare.
It is on the World Health Organization's List of Essential Medicines, a list of the most important medication needed in a basic health system.[1]
Medical uses
Rifabutin is now recommended as first-line treatment for tuberculosis,[2] but Rifampicin is more widely used because of its cheaper cost.
Rifabutin is used in the treatment of mycobacterium avium complex disease, a bacterial infection most commonly encountered in late-stage AIDS patients. Its main usefulness lies in the fact that it has lesser drug interactions than rifampicin; therefore HIV infected patients on HAART are usually given rifabutin for treatment of TB.
Rifabutin is well tolerated in patients with HIV-related tuberculosis (TB), but new findings suggest that patients with low CD4 cell counts have a high risk of treatment failure or relapse due to acquired rifamycin resistance. Since patients co-infected with TB and HIV / AIDS are likely to get TB treated first, when the CD4 is suppressed at the time TB treatment begins, doctors and patients should be aware of a possible rifamycin resistance.
Rifabutin is also being investigated in trials for treating Crohn's Disease as part of the anti-MAP therapy. In a Phase 3 study administering sub-therapeutic doses of Rifabutin in combination therapy to patients not identified with MAP infections, it was associated with significant short term benefits.[3][4]
It has also found to be useful in the treatment of (Chlamydia) Chlamydophila pneumoniae (Cpn) Infection. See external link.
History
Scientists at the Italian drug company Achifar discovered rifabutin in 1975. (Eventually Archifar became part of Farmitalia Carlo Erba, a unit of the conglomerate Montedison.) This company's Adria Laboratories subsidiary filed for Food and Drug Administration (FDA) approval of rifabutin under the brand name Mycobutin in the early 1990s and the drug gained FDA approval in December 1992.
Rifabutin (RFB) is primarily bactericidal antibiotic drug used to treat tuberculosis. Its effect on bacteria is based on the DNA-dependent RNA polymerase blocking drug rifamycin S., a semi-synthetic derivative. It is effective, for example, in highly resistant mycobacteria, Gram-positive bacteria (and some are effective against Gram-negative bacteria), but also against Mycobacterium tuberculosis, M. leprae, and M. avium intracellulare.
References
- ↑ "WHO Model List of EssentialMedicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ↑ Guidelines for the programmatic management of drug-resistant tuberculosis: emergency update 2008 (WHO/HTM/TB/2008.402). Geneva, Switzerland: World Health Organization. 2008. p. ix. ISBN 978-92-4-154758-1.
- ↑ "Two-year combination antibiotic therapy with clarithromycin, rifabutin, and clofazimine for Crohn's disease". Gastroenterology 132 (7): 2313–9. June 2007. doi:10.1053/j.gastro.2007.03.031. PMID 17570206.
- ↑ Kuenstner, J. Todd (2007). "The Australian Antibiotic Trial in Crohn’s Disease: Alternative Conclusions From the Same Study". Gastroenerology 133 (5): 1742–1743. doi:10.1053/j.gastro.2007.09.012.
External links
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||