Pyruvate carboxylase
| Pyruvate carboxylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Crystallographic structure of pyruvate carboxylase from Rhizobium etli: biotin carboxylase domain (blue); allosteric linking domain (green); biotin binding domain (red); and carboxyl transferase domain (orange)[1] | |||||||||
| Identifiers | |||||||||
| EC number | 6.4.1.1 | ||||||||
| CAS number | 9014-19-1 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||
| Pyruvate carboxyltransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | PYR_CT | ||||||||
| Pfam | PF00682 | ||||||||
| InterPro | IPR000891 | ||||||||
| PROSITE | PDOC50991 | ||||||||
| |||||||||
| Pyruvate carboxylase | |
|---|---|
| Identifiers | |
| Symbol | PC |
| Entrez | 5091 |
| HUGO | 8636 |
| OMIM | 608786 |
| RefSeq | NM_000920 |
| UniProt | P11498 |
| Other data | |
| EC number | 6.4.1.1 |
| Locus | Chr. 11 q11-q13.1 |
Pyruvate carboxylase (PC) is an enzyme of the ligase class that catalyzes the (depending on the species) irreversible carboxylation of pyruvate to form oxaloacetate (OAA).
It is an important anaplerotic reaction that creates oxaloacetate from pyruvate. The enzyme is a mitochondrial protein containing a biotin prosthetic group,[1] requiring magnesium or manganese and acetyl CoA.
Pyruvate carboxylase was first discovered in 1959 at Western Reserve University by M. F. Utter and D. B. Keech.[2][3] Since then it has been found in a wide variety of prokaryotes and eukaryotes including fungi, bacteria, plants, and animals.[4] In mammals, PC plays a crucial role in gluconeogenesis and lipogenesis, in the biosynthesis of neurotransmitters, and in glucose-induced insulin secretion by pancreatic islets. Oxaloacetate produced by PC is an important intermediate, which is used in these biosynthetic pathways.[5] In mammals, PC is expressed in a tissue-specific manner, with its activity found to be highest in the liver and kidney (gluconeogenic tissues), in adipose tissue and lactating mammary gland (lipogenic tissues), and in pancreatic islets. Activity is moderate in brain, heart and adrenal gland, and least in white blood cells and skin fibroblasts.[6]
Structure
Structural studies of PC have been conducted by electron microscopy, by limited proteolysis, and by cloning and gasa sequencing of genes and cDNA encoding the enzyme. Most well characterized forms of active PC consist of four identical subunits arranged in a tetrahedron-like structure. Each subunit contains a single biotin moiety acting as a swinging arm to transport carbon dioxide to the catalytic site that is formed at the interface between adjacent monomers. Each subunit of the functional tetramer contains four domains: the biotin carboxylation (BC) domain, the transcarboxylation (CT) domain, the biotin carboxyl carrier (BCCP) domain and the recently termed PC tetramerization (PT) domain.[7][8] From the two most complete crystal structures available, an asymmetric and symmetric form of the protein have been visualized.[9] The Staphylococcus aureus tetramer in complex with the activator Coenzyme A is highly symmetric, possessing 222 symmetry, and has been confirmed by Cryo-EM studies.[8] In contrast the Rhizobium etli, tetramer in complex with ethyl-CoA, a non-hydrolyzable analog of Acetyl-CoA, possesses only one line of symmetry.[9]
Pyruvate carboxylase uses a covalently attached Biotin cofactor which is used to catalyze the ATP– dependent carboxylation of pyruvate to oxaloacetate in two steps. Biotin is initially carboxylated at the BC active site by ATP and bicarbonate. The carboxyl group is subsequently transferred by carboxybiotin to a second active site in the CT domain, where pyruvate is carboxylated to generate oxaloacetate. The BCCP domain transfers the tethered cofactor between the two remote active sites. The allosteric binding site in PC offers a target for modifiers of activity that may be useful in the treatment of obesity or type II diabetes, and the mechanistic insights gained from the complete structural description of RePC (R. etli) permit detailed investigations into the individual catalytic and regulatory sites of the enzyme.[9]
Reaction mechanism
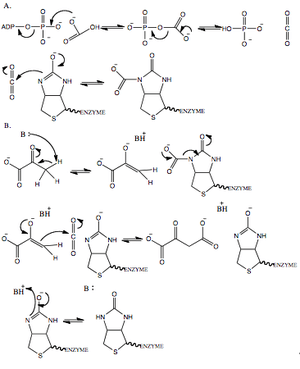
(A) ATP dependent carboxylation of biotin (BC domain);
(B) Transcarboxylation of pyruvate (CT domain).
The reaction mechanism can be subdivided into two partial reactions (see figure to the right). In the first reaction, ATP is carboxylated to produce carbonic phosphoric anhydride [−O(−O)P(=O)O–C(=O)O−] which in turn carboxylates a biotin cofactor that is covalently attached to a lysine residue of the BCCP domain.[4] Carbonic phosphoric anhydride decomposes into carbon dioxide and phosphate prior to attack by the enzyme linked biotin molecule. In most species, this reaction requires acetyl-CoA as an allosteric activator binding to the PT domain.[8] In the second reaction, occurring in the CT domain of an adjacent monomer, carbon dioxide is transferred to the acceptor molecule, pyruvate, to form oxaloacetate. The reaction proceeds via the removal of a proton from pyruvate, by an as yet unidentified active site residue, to generate an enolate intermediate. The enolate intermediate then attacks CO2 transiently released from the enzyme linked biotin molecule. The resultant oxaloacetate is released. The biotin molecule is protonated by the aforementioned active site residue and released from the active site of the CT domain to be recarboxylated.[8][9] The major regulator of enzyme activity, acetyl-CoA, stimulates the cleavage of ATP in the first partial reaction and also it has been shown to induce a conformational change in the tetrameric structure of the enzyme.[5]
Role in gluconeogenesis
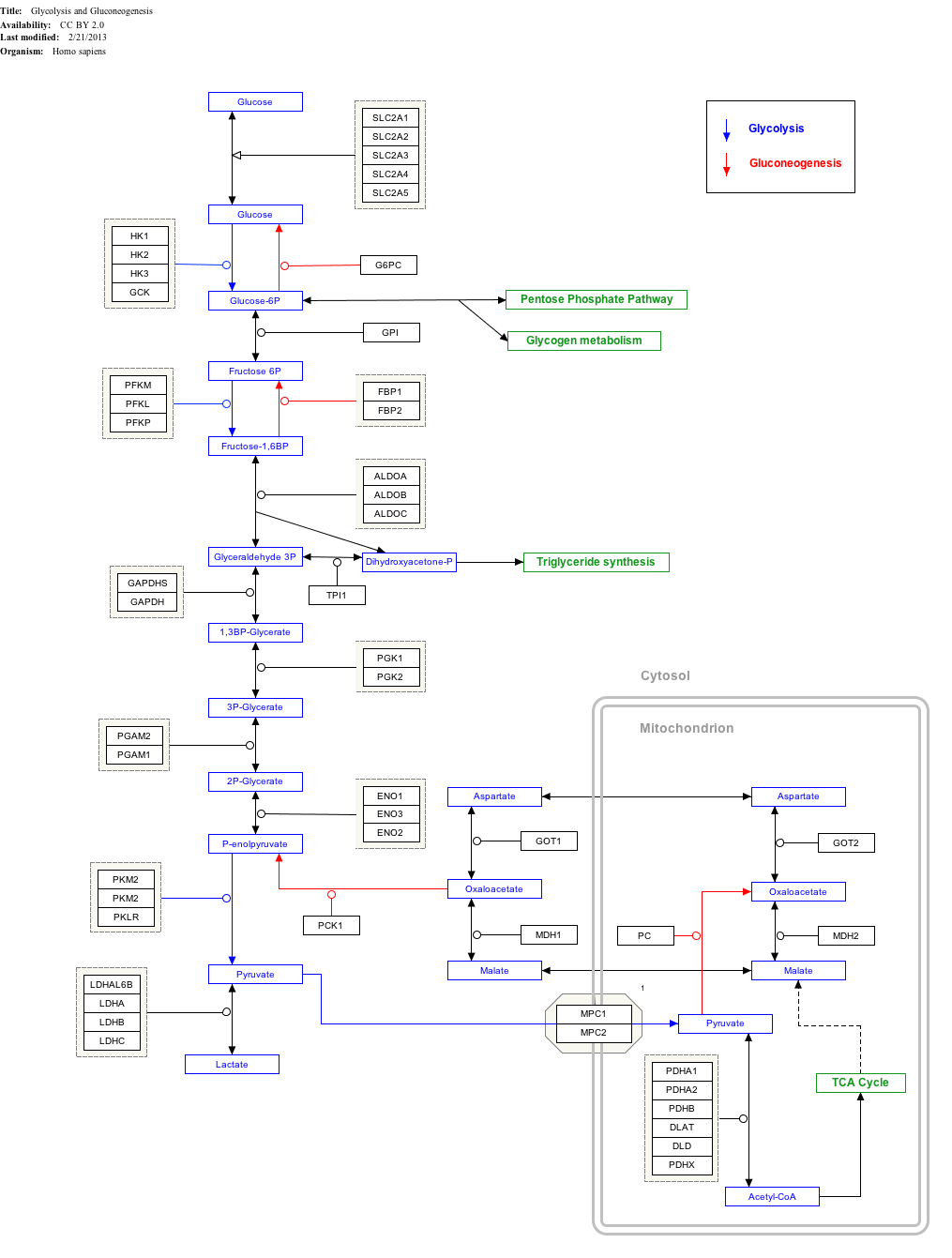
During gluconeogenesis, pyruvate carboxylase is involved in the synthesis of phosphoenolpyruvate (PEP) from pyruvate. Pyruvate is first converted by pyruvate carboxylase to oxaloacetate (OAA) in the mitochondrion requiring hydrolysis of one molecule of ATP. The OAA is then decarboxylated and simultaneously phosphorylated, which is catalyzed by one of two isoforms of phosphoenolpyruvate carboxykinase (PEPCK) either in the cytosol or in the mitochondria to produce PEP. Under ordinary gluconeogenic conditions, OAA is converted into PEP by mitochondrial PEPCK; the resultant PEP is then transported out of the mitochondrial matrix by an anion transporter carrier system,[10] and converted into glucose by cytosolic gluconeogenic enzymes. However, during starvation when cytosolic NADH concentration is low and mitochrondrial NADH levels are high oxaloacetate can be used as a shuttle of reducing equivalents. As such OAA is converted into malate by mitochondrial Malate dehydrogenase (MDH). After export into the cytosol, malate is converted back into OAA, with concomitant reduction of NAD+; OAA is subsequently converted to PEP which is available for gluconeogenesis in the cytosol along with the transported reducing equivalent NADH.[1]
Very high levels of PC activity, together with high activities of other gluconeogenic enzymes including PEPCK, fructose-1,6-bisphosphatase and glucose-6-phosphatase in liver and kidney cortex, suggest that a primary role of PC is to participate in gluconeogenesis in these organs. During fasting or starvation when endogenous glucose is required for certain tissues (brain, white blood cells and kidney medulla), expression of PC and other gluconeogenic enzymes is elevated.[11] In rats and mice, alteration of nutrition status has been shown to affect hepatic PC activity.[12] Fasting promotes hepatic glucose production sustained by an increased pyruvate flux, and increases in PC activity and protein concentration; diabetes similarly increases gluconeogenesis through enhanced uptake of substrate and increased flux through liver PC in mice and rats.[13][14] Similarly to other gluconeogenic enzymes, PC is positively regulated by glucagon and glucocorticoids while negatively regulated by insulin.[4] Further supporting the key role of PC in gluconeogenesis, in dairy cattle, which have hexose absorption ability at adequate nutrition levels, PC and the associated gluconeogenic enzyme PEPCK are markedly elevated during the transition to lactation in proposed support of lactose synthesis for milk production.[15]
Aside from the role of PC in gluconeogenesis, PC serves an anaplerotic role (an enzyme catalyzed reaction that can replenish the supply of intermediates in the citric acid cycle) for the tricarboxylic acid cycle (essential to provide oxaloacetate), when intermediates are removed for different biosynthetic purposes.
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
Glycolysis and Gluconeogenesis edit
- ↑ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Clinical significance
As a crossroad between carbohydrate and lipid metabolism, pyruvate carboxylase expression in gluconeogenic tissues, adipose tissues and pancreatic islets must be coordinated. In conditions of over nutrition, PC levels are increased in pancreatic β-cells to increase pyruvate cycling in response to chronically elevated levels of glucose.[16] In contrast, PC enzyme levels in the liver are decreased by insulin;[17] during periods of overnutrition adipocyte tissue is expanded with extreme expression of PC and other lipogenic enzymes.[6][18] Hepatic control of glucose levels is still regulated in an over nutrition situation, but in obesity induced type 2 diabetes the regulation of peripheral glucose levels is no longer under regulation of insulin. In type 2 diabetic rats, chronic exposure of β-cells to glucose due to peripheral insulin resistance results in decreased PC enzyme activity and decreased pyruvate cycling[19][20] The continued overproduction of glucose by hepatocytes causes dramatic alteration of β-cell gene expression with large increases in normally suppressed genes, and equivalent decreases in expression of mRNA for insulin, ion pumps necessary for insulin secretion, and metabolic enzymes related to insulin secretion, including pyruvate carboxylase[21][22] Concurrently adipose tissue develops insulin resistance causing accumulation of triaglycerols and non-esterified fatty acids in circulation; these not only further impairing β-cell function,[22][23] but also further decreasing PC expression.[24][25] These changes result in the decline of the β-cell phenotype in decompensated diabetes.
A deficiency of pyruvate carboxylase can cause lactic acidosis as a result of lactate build up.[26] Normally, excess pyruvate is shunted into gluconeogenesis via conversion of pyruvate into oxaloacetate, but because of the enzyme deficiency, excess pyruvate is converted into lactate instead. As a key role of gluconeogenesis is in the maintenance of blood sugar, deficiency of pyruvate carboxylase can also lead to hypoglycemia.
References
- ↑ 1.0 1.1 1.2 PDB 2QF7; Jitrapakdee S, St Maurice M, Rayment I, Cleland WW, Wallace JC, Attwood PV (August 2008). "Structure, mechanism and regulation of pyruvate carboxylase". Biochem. J. 413 (3): 369–87. doi:10.1042/BJ20080709. PMC 2859305. PMID 18613815.
- ↑ Utter MF, Keech DB (May 1960). "Formation of oxaloacetate from pyruvate and carbon dioxide". J. Biol. Chem. 235: PC17–8. PMID 13840551.
- ↑ Cohen ND, Beegen H, Utter MF, Wrigley NG (March 1979). "A re-examination of the electron microscopic appearance of pyruvate carboxylase from chicken liver". J. Biol. Chem. 254 (5): 1740–7. PMID 762171.
- ↑ 4.0 4.1 4.2 Jitrapakdee S, Vidal-Puig A, Wallace JC (April 2006). "Anaplerotic roles of pyruvate carboxylase in mammalian tissues". Cell. Mol. Life Sci. 63 (7-8): 843–54. doi:10.1007/s00018-005-5410-y. PMID 16505973.
- ↑ 5.0 5.1 Jitrapakdee S, Nezic MG, Cassady AI, Khew-Goodall Y, Wallace JC (July 2002). "Molecular cloning and domain structure of chicken pyruvate carboxylase". Biochem. Biophys. Res. Commun. 295 (2): 387–93. doi:10.1016/S0006-291X(02)00651-4. PMID 12150961.
- ↑ 6.0 6.1 Jitrapakdee S, Walker ME, Wallace JC (June 1996). "Identification of novel alternatively spliced pyruvate carboxylase mRNAs with divergent 5'-untranslated regions which are expressed in a tissue-specific manner". Biochem. Biophys. Res. Commun. 223 (3): 695–700. doi:10.1006/bbrc.1996.0958. PMID 8687459.
- ↑ Kondo S, Nakajima Y, Sugio S, Yong-Biao J, Sueda S, Kondo H (March 2004). "Structure of the biotin carboxylase subunit of pyruvate carboxylase from Aquifex aeolicus at 2.2 A resolution". Acta Crystallogr. D Biol. Crystallogr. 60 (Pt 3): 486–92. doi:10.1107/S0907444904000423. PMID 14993673.
- ↑ 8.0 8.1 8.2 8.3 Yu LP, Xiang S, Lasso G, Gil D, Valle M, Tong L (June 2009). "A symmetrical tetramer for S. aureus pyruvate carboxylase in complex with coenzyme A". Structure 17 (6): 823–32. doi:10.1016/j.str.2009.04.008. PMC 2731552. PMID 19523900.
- ↑ 9.0 9.1 9.2 9.3 St Maurice M, Reinhardt L, Surinya KH, Attwood PV, Wallace JC, Cleland WW, Rayment I (August 2007). "Domain architecture of pyruvate carboxylase, a biotin-dependent multifunctional enzyme". Science 317 (5841): 1076–9. doi:10.1126/science.1144504. PMID 17717183.
- ↑ Stark R, Pasquel F, Turcu A et al. (2009). "Phosphoenolpyruvate cycling via mitochondrial phosphoenolpyruvate carboxykinase links anaplerosis and mitochondrial GTP with insulin secretion.". Journal of Biological Chemistry 284 (39): 26578–26590. doi:10.1074/jbc.M109.011775. PMC 2785346. PMID 19635791.
- ↑ Rothman DL, Magnusson I, Katz LD, Shulman RG, Shulman GI (October 1991). "Quantitation of hepatic glycogenolysis and gluconeogenesis in fasting humans with 13C NMR". Science 254 (5031): 573–6. doi:10.1126/science.1948033. PMID 1948033.
- ↑ Bizeau ME, Short C, Thresher JS, Commerford SR, Willis WT, Pagliassotti MJ (2001). "Increased pyruvate flux capacities account for diet induced increase in gluconeogenesis ‘’in vitro’’". Am. J. Physiol. Regul. Integr. Comp. Physiol. 281 (2): R427–R433. PMID 11448844.
- ↑ Salto R, Sola M, Olicer F J, Vargas A M (Dec 1996). "Effects of starvation, diabetes, and carbon tetrachloride intoxication on rat kidney cortex and liver pyruvate carboxylase levels". Arch. Physiol. Biochem. 104 (7): 845–850. doi:10.1076/apab.104.7.845.13111. PMID 9127680.
- ↑ Large V, Beylot M (June 1999). "Modifications of citric acid cycle activity and gluconeogenesis in strepozotocin induced diabetes and effects of metformin". Diabetes 48 (6): 1251–1257. doi:10.2337/diabetes.48.6.1251. PMID 10342812.
- ↑ Greenfield RB, Cecava MJ, Donkin SS (2002). "Changes in mRNA expression for gluconeogenic enzymes in the liver of dairy cattle during transition to lactation". Journal of Dairy Science 82 (6): 1228–1236. doi:10.3168/jds.S0022-0302(00)74989-7. PMID 10877388.
- ↑ Liu YQ, Han J, Epstein PN, Long YS (Dec 2005). "Enhanced rat β-cell proliferation in 60% pancreatectomized islets by increased glucose metabolic flux through pyruvate carboxylase pathway". Am. J. Physiol. Endocrinol. Metab 288 (3): E471–E478. doi:10.1152/ajpendo.00427.2004. PMID 15507531.
- ↑ Desvergne B, Michalik L, Wahli W (April 2006). "Transcriptional regulation of metabolism". Physiol. Rev 86 (2): 465–514. doi:10.1152/physrev.00025.2005. PMID 16601267.
- ↑ Lynch CJ, McCall KM, Billingsley ML, Bohlen LM, Hreniuk SP, Martin LF, Witters LA, Vannucci SJ (May 1992). "Pyruvate carboxylase in genetic obesity". Am. J. Physiol 262 (5 Pt 1): E608–E618. PMID 1375435.
- ↑ MacDonald MJ, Tang J, Polonsky KS (Nov 1996). "Low mitochondrial glycerol phosphate dehydrogenase and pyruvate carboxylase in pancreatic islets of Zucker diabetic fatty rats". Diabetes 45 (11): 1626–1630. doi:10.2337/diabetes.45.11.1626. PMID 8866570.
- ↑ McDonald MJ, Efendic S, Ostenson CG (July 1996). "Normalization by insulin of low mitochondrial glycerol phosphate dehydrogenase and pyruvate carboxylase in pancreatic islets of the GK rat". Diabetes 45 (7): 886–890. doi:10.2337/diabetes.45.7.886. PMID 8666138.
- ↑ Laybutt DR, Glandt M, Xu G, Ahn YB, Trivedi N, Bonner-Weir S, Weir GC (Jan 2003). "Critical reduction in β-cell mass results in two distinct outcomes over time. Adaption with impaired glucose tolerance or decompensated diabetes". J. Biol. Chem. 278 (5): 2997–3005. doi:10.1074/jbc.M210581200. PMID 12438314.
- ↑ 22.0 22.1 Poitout V, Robertson R P (Feb 2002). "Secondary ß-cell failure in type 2 diabetes-a convergence of glucotoxicity and lipotoxicity". Endocrinology 143 (2): 339–342. doi:10.1210/en.143.2.339. PMID 11796484.
- ↑ Boucher A, Lu D, Burgess SC, Telamaque-Potts S, Jensen M V, Mulder H, Wang M Y, Unger R H, Sherry A D, Newgard C B (2004). "Biochemical mechanism of lipid-induced impairment of glucose-stimulated insulin secretion and reversal with a malate analogue". J. Biol. Chem. 279 (26): 27263–27271. doi:10.1074/jbc.M401167200. PMID 15073188.
- ↑ Busch AK, Cordery D, Denyer GS, Biden TJ (Apr 2002). "Expression profiling of palmitate- and oleate-regulated genes provides novel insights into the effects of chronic exposure on pancreatic β-cell function". Diabetes 51 (4): 977–987. doi:10.2337/diabetes.51.4.977. PMID 11916915.
- ↑ Iizuka K, Nakajima H, Namba M, Miyagawa J, Mijazaki J, Hanafusa T, Matsuzawa Y (Jan 2002). "Metabolic consequences of long-term exposure of pancreatic β-cells to free fatty acid with special reference to glucose insensitivity". Biochim. Biophys. Acta 1586 (1): 23–31. doi:10.1016/s0925-4439(01)00082-5. PMID 11781146.
- ↑ García-Cazorla A, Rabier D, Touati G, Chadefaux-Vekemans B, Marsac C, de Lonlay P, Saudubray JM (January 2006). "Pyruvate carboxylase deficiency: metabolic characteristics and new neurological aspects". Ann. Neurol. 59 (1): 121–7. doi:10.1002/ana.20709. PMID 16278852.
External links
See also
| |||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||