Pyrrolnitrin
 | |
| Names | |
|---|---|
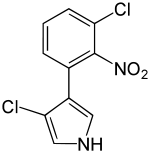
| IUPAC name
3-chloro-4-(3-chloro-2-nitrophenyl)-1H-pyrrole | |
| Identifiers | |
| ATC code | D01 |
| 1018-71-9 | |
| ChemSpider | 13314 |
| EC number | 213-812-7 |
| |
| Jmol-3D images | Image |
| KEGG | D01094 |
| MeSH | D011764 |
| PubChem | 13916 |
| |
| Properties | |
| C10H6Cl2N2O2 | |
| Molar mass | 257.07284 |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Pyrrolnitrin is an antifungal antibiotic.[1] Pseudomonas pyrrocinia and other Pseudomonas species produce pyrrolnitrin from tryptophan as secondary metabolite.[2][3]
References
- ↑ Gordee, R. S.; Matthews, T. R. (1969). "Systemic antifungal activity of pyrrolnitrin". Applied microbiology 17 (5): 690–694. PMC 377781. PMID 5785951.
- ↑ Zhu, X.; Van Pee, K. -H.; Naismith, J. H. (2010). "The Ternary Complex of PrnB (the Second Enzyme in the Pyrrolnitrin Biosynthesis Pathway), Tryptophan, and Cyanide Yields New Mechanistic Insights into the Indolamine Dioxygenase Superfamily". Journal of Biological Chemistry 285 (27): 21126–21133. doi:10.1074/jbc.M110.120485. PMC 2898318. PMID 20421301.
- ↑ Park, J. Y.; Oh, S. A.; Anderson, A. J.; Neiswender, J.; Kim, J. -C.; Kim, Y. C. (2011). "Production of the antifungal compounds phenazine and pyrrolnitrin from Pseudomonas chlororaphis O6 is differentially regulated by glucose". Letters in Applied Microbiology 52 (5): 532–537. doi:10.1111/j.1472-765X.2011.03036.x. PMID 21362001.