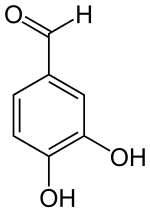
Protocatechuic aldehyde
 | |
| Names | |
|---|---|
| IUPAC name
3,4-dihydroxybenzaldehyde | |
| Other names
Protocatechualdehyde 3,4-Dihydroxybenzaldehyde Rancinamycin IV 3,4-Dihydroxybenzyl aldehyde | |
| Identifiers | |
| 139-85-5 | |
| ChEMBL | ChEMBL222021 |
| ChemSpider | 8438 |
| |
| Jmol-3D images | Image |
| PubChem | 8768 |
| |
| Properties | |
| C7H6O3 | |
| Molar mass | 138.12 g/mol |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Protocatechuic aldehyde is a phenolic aldehyde, a compound released from cork stoppers into wine.[1]
This molecule can be used as a precursor in the vanillin synthesis by biotransformation by cell cultures of Capsicum frutescens, a type of Chili pepper.[2] It is also found in the mushroom Phellinus linteus.[3]
References
- ↑ Polyphenolic Composition of Quercus suber Cork from Different Spanish Provenances. Elvira Conde, Estrella Cadahía, María Concepción García-Vallejo and Brígida Fernández de Simón, J. Agric. Food Chem., 1998, 46 (8), pp 3166–3171 doi:10.1021/jf970863k
- ↑ Biotransformation of protocatechuic aldehyde and caffeic acid to vanillin and capsaicin in freely suspended and immobilized cell cultures of Capsicum frutescens. Sathuluri Ramachandra Rao and Gokare Aswathanarayana Ravishankar, Journal of Biotechnology, Volume 76, Issues 2-3, 21 January 2000, Pages 137-146 doi:10.1016/S0168-1656(99)00177-7
- ↑ Lee YS, Kang YH, Jung JY et al. (October 2008). "Protein glycation inhibitors from the fruiting body of Phellinus linteus" ([DEAD LINK]). Biological & Pharmaceutical Bulletin 31 (10): 1968–72. doi:10.1248/bpb.31.1968. PMID 18827365.
See also
- Phenolic compounds in wine