Otamixaban
Otamixaban|
|
|
|
| Systematic (IUPAC) name |
|---|
|
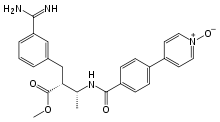
Methyl (2R,3R)-2-{3-[amino(imino)methyl]benzyl}-3-{[4-(1-oxidopyridin-4-yl)benzoyl]amino}butanoate |
| Clinical data |
|---|
| Identifiers |
|---|
|
193153-04-7  N N |
|---|
|
None |
|---|
| PubChem |
CID 5496659 |
|---|
| ChemSpider |
4593439  Y Y |
|---|
| UNII |
S173RED00L  Y Y |
|---|
| ChEMBL |
CHEMBL46618  Y Y |
|---|
| Chemical data |
|---|
| Formula |
C25H26N4O4 |
|---|
|
446.498 g/mol |
|---|
SMILES
- O=C(OC)[C@H](Cc1cc(C(=[N@H])N)ccc1)[C@H](NC(=O)c3ccc(c2cc[n+]([O-])cc2)cc3)C
|
InChI=1S/C25H26N4O4/c1-16(22(25(31)33-2)15-17-4-3-5-21(14-17)23(26)27)28-24(30)20-8-6-18(7-9-20)19-10-12-29(32)13-11-19/h3-14,16,22H,15H2,1-2H3,(H3,26,27)(H,28,30)/t16-,22-/m1/s1
 Y Y
Key:PFGVNLZDWRZPJW-OPAMFIHVSA-N
 Y Y
|
 N (what is this?) (verify) N (what is this?) (verify) |
|---|
Otamixaban (INN) is an experimental injectable anticoagulant direct factor Xa inhibitor,[1] that was investigated for the treatment for acute coronary syndrome. In 2013, Sanofi announced that it had ended development of the drug candidate after poor performance in a Phase III clinical trial.[2][3]
References
|
|---|
| | Antiplatelet drugs | | |
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| Thromboxane inhibitors |
- Thromboxane synthase inhibitors
- Receptor antagonists
|
|---|
| | |
|---|
| Other | |
|---|
|
|---|
| | Anticoagulants | | |
|---|
| Factor Xa inhibitors
(with some II inhibition) | |
|---|
| | |
|---|
| Other | |
|---|
|
|---|
| Thrombolytic drugs/
fibrinolytics | |
|---|
| | Non-medicinal | |
|---|
| |
|---|
| | Description |
- Immune system
- Cells
- Physiology
- coagulation
- proteins
- granule contents
- colony-stimulating
- heme and porphyrin
|
|---|
| | Disease |
- Red blood cell
- Monocyte and granulocyte
- Neoplasms and cancer
- Histiocytosis
- Symptoms and signs
- Blood tests
|
|---|
| | Treatment |
- Transfusion
- Drugs
- thrombosis
- bleeding
- other
|
|---|
|
|