Neuroligin
| neuroligin 1 | |
|---|---|
| Identifiers | |
| Symbol | NLGN1 |
| Entrez | 22871 |
| HUGO | 14291 |
| OMIM | 600568 |
| RefSeq | NP_055747 |
| UniProt | Q8N2Q7 |
| Other data | |
| Locus | Chr. 3 q26.31 |
| neuroligin 2 | |
|---|---|
| Identifiers | |
| Symbol | NLGN2 |
| Entrez | 57555 |
| HUGO | 14290 |
| OMIM | 606479 |
| RefSeq | NP_065846 |
| UniProt | Q8NFZ4 |
| Other data | |
| Locus | Chr. 17 p13.1 |
| neuroligin 3 | |
|---|---|
| Identifiers | |
| Symbol | NLGN3 |
| Entrez | 54413 |
| HUGO | 14289 |
| OMIM | 300336 |
| RefSeq | NP_001160132 |
| UniProt | Q9NZ94 |
| Other data | |
| Locus | Chr. X q13.1 |
| neuroligin 4X | |
|---|---|
| Identifiers | |
| Symbol | NLGN4X |
| Entrez | 57502 |
| HUGO | 14287 |
| OMIM | 300427 |
| RefSeq | NP_065793 |
| UniProt | Q8N0W4 |
| Other data | |
| Locus | Chr. X p22.32-22.31 |
Neuroligin (NLGN), a type I membrane protein, is a cell adhesion protein on the postsynaptic membrane that mediates the formation and maintenance of synapses between neurons. Neuroligins act as ligands for β-Neurexins, which are cell adhesion proteins located presynaptically. Neuroligin and β-neurexin "shake hands," resulting in the connection between two neurons and the production of a synapse.[2] Neuroligins also affect the properties of neural networks by specifying synaptic functions, and they mediate signalling by recruiting and stabilizing key synaptic components. Neuroligins interact with other postsynaptic proteins to localize neurotransmitter receptors and channels in the postsynaptic density as the cell matures.[3] Additionally, neuroligins are expressed in human peripheral tissues and have been found to play a role in angiogenesis.[4] In humans, alterations in genes encoding neuroligins are implicated in autism and other cognitive disorders.[5]
Structure
Neuroligins bind with the aid of Ca2+ to the α-neurexin LNS (laminin, neurexin and sex hormone-binding globulin-like folding units) domains and to the β-neurexin LNS domain which then establishes a heterophilic trans-synaptic recognition code.[6] Through the observation of the crystal structure of neuroligin-1, it was determined that neuroligin-1 forms a dimer when two neurexin-1 beta monomers bind to the neuroligin-1’s two opposite surfaces. This forms a heterotetramer, which contains an interface for binding Ca2+. The interaction of neuroligin and neurexin to form a heterotetramer is monitored by alternatively spliced sites located near the binding interface for Ca2+ in both the neuroligin-1 and the neurexin-1 beta.[7]
The extracellular domain of NLGN consists mostly of a region that is homologous to acetylcholinesterases, but the amino acids important for catalysis in AChE are not conserved in NLGN, which lack esterase activity. Furthermore, this AChE homologous region is crucial for the proper function of NLGN.[2]
Genetics
Neuroligins have been identified in both vertebrates and invertebrates, including humans, rodents, chickens, Drosophila melanogaster, Caenorhabditis elegans, honeybees, and Aplysia. Three genes for neuroligin expression have been found in mice and rats, while humans express five genes.[8] Drosophila express four genes, honeybees express five genes, and both C. elegans and Aplysia express a single gene for neuroligin.[9]
The known neuroligin genes in Homo sapiens include NLGN1, NLGN2, NLGN3, NLGN4X, and NLGN5 (also known as NLGN4Y). Each gene has been found to have unique influences on synaptic transmission.
Expression
Expression of neuroligins may differ between species. Neuroligin 1 is expressed specifically in the CNS at excitatory synapses. In humans, expression of neuroligin 1 is low before birth and increases between postnatal days 1-8 and remains high through adulthood. This postnatal increase during active synaptogenesis corresponds to increased expression of postsynaptic density protein-95 (PSD-95). Neuroligin 2 is mainly concentrated at inhibitory synapses in the CNS, but in mice and humans it may also be expressed in tissues such as the pancreas, lung, endothelia, uterus, and colon. Neuroligin 3 is expressed in CNS neurons, as well as a variety of glial cells in mice and rats, and the brain, heart, skeletal muscle, placenta, and pancreas in humans. Neuroligin 4X, found only in humans, is expressed in the heart, liver, skeletal muscle, pancreas, and low levels in the brain. Neuroligin 5 (or 4Y), located on the Y chromosome, is only 19 amino acids different from neuroligin 4X.[8] Neuroligin mRNA are present in human endothelial cells from large blood vessels [10] and in Dorsal Root Ganglions.[11]
Alternative splicing
Alternative splicing, a modification that occurs after transcription of mRNA, regulates neuroligins’ binding selectivity for α- or β-neurexins as well as the function of synapses. Alternative splicing in neuroligins occurs in the main functional domain, the acetylcholinesterase-homologous region.[12] Because neuroligin has two conserved splice sites in this region, sites A and B, up to four different isoforms are possible for each neuroligin gene.[8] Neurexins also undergo alternative splicing, and certain splice variants of neuroligins and neurexins are more selective for one another. Specific pairing of splice variants also affects synaptic function. For example, neuroligins lacking the B splice insert and β-neurexins with the S4 insert promote differentiation of inhibitory, GABAergic synapses. On the other hand, neuroligins with the B insert and β-neurexins lacking the S4 insert promote differentiation of excitatory, glutamatergic synapses. The A insert may promote neuroligin localization and function at inhibitory synapses, but the mechanisms are unknown.[12]
Activity with Neurexin
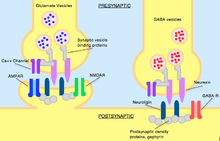
Neurexin and neuroligin work together to gather and maintain the cytoskeleton components needed to localize synaptic vesicles. Neurexin is necessary for containing the voltage-gated Ca2+ channels that are required for the release of vesicles, while neuroligin binds neurexin in order to localize the necessary neurotransmitter receptors and proteins for postsynaptic specialization. At the postsynaptic site, neuroligins are networked to specialized proteins that stimulate specific neurotransmitter receptors and channels to densely occupy specialized regions of the postsynaptic terminal during the maturation of the synapse. Because all developing synapses contain neurexins and neuroligins, developing cells can make many different connections to other cells.[3]
Synapse Formation
Neuroligin is sufficient to form new functional presynaptic terminals in vitro.[8] However, evidence suggests that additional adhesion molecules, such as immunoglobulin-domain and cadherin family proteins, mediate the initial contact between the axons and dendrites for a synapse. Neurexins and neuroligins then reinforce the contact.[12]
In addition to the selectivity of splice variants, the levels of neuroligins, neurexins, and other interacting proteins present on the pre- and postsynaptic membranes influence the differentiation and balance of synapses. As synapses form during synaptogenesis, they differentiate into one of two categories: excitatory or inhibitory. Excitatory synapses have increased probability of firing an action potential in the postsynaptic neuron and are often glutamatergic, or synapses in which the neurotransmitter glutamate is released. Inhibitory synapses have decreased probability of firing an action potential in the postsynaptic neuron and are often GABAergic, in which the neurotransmitter GABA is released. Especially during early development, neurons must receive an appropriate balance of excitatory vs. inhibitory synaptic input, referred to as the E/I ratio. In fact, an imbalance in the E/I ratio is thought to be involved in autistic spectrum disorders.[13]
Neuroligin 1 localizes at excitatory synapses, neuroligin 2 at inhibitory synapses, and neuroligin 3 at both. Reduction in the levels of neuroligins 1, 2, and 3 results in a strong reduction of inhibitory input but little reduction in excitatory input.[12] In addition, Neuroligins interacts with PSD-95, an intracellular protein that anchors synaptic proteins in the post-synaptic density of excitatory synapses, and gephyrin, the respective scaffolding protein of inhibitory post-synapses.[14] In addition, neuroligin 2 and 4 specifically interact with collybistin a protein that regulates the localization of gephyrin. The level of PSD-95 appears to influence the balance of excitatory and inhibitory inputs. An increase in the ratio of PSD-95 to neuroligin resulted in an increase in the E/I ratio, and a decrease in the PSD-95/neuroligin ratio had the opposite effect.[13] Also, overexpression of PSD-95 redirects neuroligin-2 from excitatory to inhibitory synapses, strengthening excitatory input and reducing inhibitory input.[12] These interactions of neuroligin, neurexin, and interacting proteins such as PSD-95 point to a potential regulatory mechanism that controls development and balance of excitatory and inhibitory synapses, governed by homeostatic feedback mechanisms.[13]
Clinical Significance to ASD
Neuroligin dysfunction has been implicated in Autistic spectrum disorders. Different genetic alterations have been detected in neuroligin genes in patients with ASD, including point mutations, missense mutations, and internal deletions.[10] In studies done on family members with X-linked autism, specific mutations of NLGN 3 and NLGN 4 have been identified. These mutations have been shown to affect how neuroligins function and have been shown to interfere with synaptic transmission. 19 of the 69 known proteins mutated in X-linked autism encode postsynaptic proteins, Neuroligins included.
NLGN 3 Mutations
A mutated NLGN 3 gene, R451C, has been cloned. The mutant has been shown to cause defective neuroligin trafficking and retention of the mutant protein in the endoplasmic reticulum.[15] The small amount of mutant protein that reached the cell membrane demonstrated diminished binding activity for neurexin-1, consistent with a loss of function.[16] The mutant gene has been cloned and was introduced into mice, resulting in impaired social interactions, enhanced spatial learning abilities, and increased inhibitory synaptic transmission. Deleting NLGN 3 did not produce these effects, thus indicating R451C to be a gain-of-function mutation. This supports the claim that increased inhibitory synaptic transmission may contribute to human autism spectrum disorders.[17]
NLGN 4 Mutations
Mutations in NLGN 4 have also been found in persons with X-linked autism. A frame shift mutation 1186T has been found to cause an early stop codon and premature protein truncation. This mutation results in intracellular retention of mutant proteins, possibly causing impaired function of a synaptic cell adhesion molecule,[15] and modifying the binding of the neuroligin protein to its presynaptic partners, neurexins, thus interrupting essential synaptic function.[18] Other mutations of NLGN 4 found in relation to autism spectrum disorders include a 2-bp deletion, 1253delAG, in the NLGN4 gene, which causes a frameshift and a premature stop codon.[19] Another mutation is a hemizygous deletion in the NLGN4 gene encompassing exons 4, 5, and 6. The 757-kb deletion was predicted to result in a significantly truncated protein.[20]
See also
References
- ↑ Fabrichny IP, Leone P, Sulzenbacher G, Comoletti D, Miller MT, Taylor P, Bourne Y, Marchot P (December 2007). "Structural analysis of the synaptic protein neuroligin and its beta-neurexin complex: determinants for folding and cell adhesion". Neuron 56 (6): 979–91. doi:10.1016/j.neuron.2007.11.013. PMC 2703725. PMID 18093521.
- ↑ 2.0 2.1 Scheiffele P, Fan J, Choih J, Fetter R, Serafini T (June 2000). "Neuroligin expressed in nonneuronal cells triggers presynaptic development in contacting axons". Cell 101 (6): 657–69. doi:10.1016/S0092-8674(00)80877-6. PMID 10892652.
- ↑ 3.0 3.1 Purves, Dale; Augustine, George; Fitzpatrick, David; Hall, William C.; LaMantia, Anthony-Samual; White, Leonard E. (2012). Neuroscience Fifth Edition. Sunderland, MA: Sinauer Associates. pp. 521–522. ISBN 978-0-87893-695-3.
- ↑ Bottos A, Destro E, Rissone A, Graziano S, Cordara G, Assenzio B, Cera MR, Mascia L, Bussolino F, Arese M (December 2009). "The synaptic proteins neurexins and neuroligins are widely expressed in the vascular system and contribute to its functions". Proc. Natl. Acad. Sci. U.S.A. 106 (49): 20782–7. doi:10.1073/pnas.0809510106. PMC 2791601. PMID 19926856.
- ↑ Südhof TC (October 2008). "Neuroligins and neurexins link synaptic function to cognitive disease". Nature 455 (7215): 903–11. doi:10.1038/nature07456. PMC 2673233. PMID 18923512.
- ↑ Igor P. Fabrichny, Philippe Leone, Gerlind Sulzenbacher, Davide Comoletti, Meghan T. Miller, Palmer Taylor, Yves Bourne, Pascale Marchot (December 2007) Neuron. 56(6): 979–991. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2703725/PMCID: PMC2703725 NIHMSID: NIHMS109548
- ↑ Araç D, Boucard AA, Ozkan E, Strop P, Newell E, Südhof TC, Brunger AT (December 2007) Neuron. 56(6):992-1003. http://www.ncbi.nlm.nih.gov/pubmed/18093522
- ↑ 8.0 8.1 8.2 8.3 Lisé MF, El-Husseini A (August 2006). "The neuroligin and neurexin families: from structure to function at the synapse". Cell. Mol. Life Sci. 63 (16): 1833–49. doi:10.1007/s00018-006-6061-3. PMID 16794786.
- ↑ Knight, D., Xie, W., Boulianne, G. (2011). "Neurexins and Neuroligins: Recent insights from invertebrates". Molecular Neurobiology 44 (3): 426–440. doi:10.1007/s12035-011-8213-1. PMC 3229692. PMID 22037798.
- ↑ 10.0 10.1 Bottos A, Rissone A, Bussolino F, Arese M (August 2011). "Neurexins and neuroligins: synapses look out of the nervous system". Cell. Mol. Life Sci. 68 (16): 2655–66. doi:10.1007/s00018-011-0664-z. PMID 21394644.
- ↑ Lorenzo LE, Godin AG, Wang F, St-Louis M, Carbonetto S, Wiseman PW, Ribeiro-da-Silva A, De Koninck Y (June 2014). "Gephyrin Clusters Are Absent from Small Diameter Primary Afferent Terminals Despite the Presence of GABAA Receptors". J. Neurosci. 34 (24): 8300–17. doi:10.1523/JNEUROSCI.0159-14.2014. PMID 24920633.
- ↑ 12.0 12.1 12.2 12.3 12.4 Craig AM, Kang Y (February 2007). "Neurexin-neuroligin signaling in synapse development". Curr. Opin. Neurobiol. 17 (1): 43–52. doi:10.1016/j.conb.2007.01.011. PMC 2820508. PMID 17275284.
- ↑ 13.0 13.1 13.2 Levinson, J.N., El-Husseini, A. (2005). "Building Excitatory and Inhibitory Synapses: Balancing Neuroligin Partnerships,". Neuron 48 (2): 171–174. doi:10.1016/j.neuron.2005.09.017. PMID 16242398.
- ↑ Poulopoulos, Alexandros; Aramuni, Gayane; Meyer, Guido; Soykan, Tolga; Hoon, Mrinalini; Papadopoulos, Theofilos; Zhang, Mingyue; Paarmann, Ingo et al. (2009). "Neuroligin 2 Drives Postsynaptic Assembly at Perisomatic Inhibitory Synapses through Gephyrin and Collybistin". Neuron 63 (5): 628–42. doi:10.1016/j.neuron.2009.08.023. PMID 19755106.
- ↑ 15.0 15.1 Chih B, Afridi SK, Clark L, Scheiffele P (July 2004). "Disorder-associated mutations lead to functional inactivation of neuroligins". Hum. Mol. Genet. 13 (14): 1471–7. doi:10.1093/hmg/ddh158. PMID 15150161.
- ↑ Comoletti D, De Jaco A, Jennings LL, Flynn RE, Gaietta G, Tsigelny I, Ellisman MH, Taylor P (May 2004). "The Arg451Cys-neuroligin-3 mutation associated with autism reveals a defect in protein processing". J. Neurosci. 24 (20): 4889–93. doi:10.1523/JNEUROSCI.0468-04.2004. PMID 15152050.
- ↑ Tabuchi K, Blundell J, Etherton MR, Hammer RE, Liu X, Powell CM, Südhof TC (October 2007). "A neuroligin-3 mutation implicated in autism increases inhibitory synaptic transmission in mice". Science 318 (5847): 71–6. doi:10.1126/science.1146221. PMC 3235367. PMID 17823315.
- ↑ Jamain S, Quach H, Betancur C, Råstam M, Colineaux C, Gillberg IC, Soderstrom H, Giros B, Leboyer M, Gillberg C, Bourgeron T (May 2003). "Mutations of the X-linked genes encoding neuroligins NLGN3 and NLGN4 are associated with autism". Nat. Genet. 34 (1): 27–9. doi:10.1038/ng1136. PMC 1925054. PMID 12669065.
- ↑ Laumonnier F, Cuthbert PC, Grant SG (February 2007). "The role of neuronal complexes in human X-linked brain diseases". Am. J. Hum. Genet. 80 (2): 205–20. doi:10.1086/511441. PMC 1785339. PMID 17236127.
- ↑ Lawson-Yuen A, Saldivar JS, Sommer S, Picker J (May 2008). "Familial deletion within NLGN4 associated with autism and Tourette syndrome". Eur. J. Hum. Genet. 16 (5): 614–8. doi:10.1038/sj.ejhg.5202006. PMID 18231125.