Monocalcium phosphate
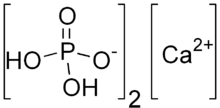 | |
| | |
| Names | |
|---|---|
| IUPAC name
Calcium dihydrogen phosphate | |
| Other names
Acid calcium phosphate Calcium acid phosphate Calcium diorthophosphate Calcium biphosphate Calcium superphosphate Monobasic calcium phosphate Monocalcium orthophosphate Phosphoric acid, calcium salt (2:1) | |
| Identifiers | |
| 7758-23-8 | |
| PubChem | 24454 |
| Properties | |
| CaH4P2O8 | |
| Molar mass | 234.05 g/mol |
| Appearance | white powder |
| Density | 2.220 g/cm3 |
| Melting point | 109 °C (228 °F; 382 K) |
| Boiling point | 203 °C (397 °F; 476 K) (decomposes) |
| 2 g/100 mL | |
| Solubility | soluble in HCl, nitric acid, acetic acid |
| Refractive index (nD) |
1.5176 |
| Structure | |
| Crystal structure | triclinic |
| Hazards | |
| EU Index | Not listed |
| NFPA 704 | |
| Flash point | Non-flammable |
| Related compounds | |
| Other anions |
Calcium pyrophosphate |
| Other cations |
Magnesium phosphate Dicalcium phosphate Tricalcium phosphate Strontium phosphate |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Monocalcium phosphate is an inorganic compound with the chemical formula Ca(H2PO4)2 ("ACMP" or "CMP-A" for anhydrous monocalcium phosphate). It is commonly found as the monohydrate (""MCP" or "MCP-M"), Ca(H2PO4)2·H2O (CAS# 10031-30-8). Both salts are colourless solids. They are used mainly as superphosphate fertilizers and are also popular leavening agents.[1]
Preparation
Material of relatively high purity, as required for baking, is produced by treating calcium hydroxide with phosphoric acid:
- Ca(OH)2 + 2 H3PO4 → Ca(H2PO4)2 + 2 H2O
Samples of Ca(H2PO4)2 tends to convert to dicalcium phosphate:
- Ca(H2PO4)2 → Ca(HPO4) + H3PO4
Applications
Use in fertilizers
Superphosphate fertilizers are produced by treatment of "phosphate rock" with acids. Using phosphoric acid, fluorapatite is converted to Ca(H2PO4)2:
- Ca5(PO4)3F + 7 H3PO4 → 5 Ca(H2PO4)2 + HF
This solid is called triple superphosphate. Several million tons are produced annually for use as fertilizers. Residual HF typically reacts with silicate minerals co-mingled with the phophate ores to produce hydrofluorosilicic acid (H2SiF6). The majority of the hexafluorosilicic acid is converted to aluminium fluoride and cryolite for the processing of aluminium.[1] These materials are central to the conversion of aluminium ore into aluminium metal.
When sulfuric acid is used, the product contains phosphogypsum ( CaSO4·2H2O) and is called single superphosphate. [2]
Use as leavening agent
Calcium dihydrogen phosphate is used in the food industry as a leavening agent, i.e., to cause baked goods to rise. Because it is acidic, when combined with an alkali ingredient, commonly sodium bicarbonate (baking soda) or potassium bicarbonate, it reacts to produce carbon dioxide and a salt. Outward pressure of the carbon dioxide gas causes the rising effect. When combined in a ready-made baking powder, the acid and alkali ingredients are included in the right proportions such that they will exactly neutralize each other and not significantly affect the overall pH of the product. AMCP and MCP are fast acting, releasing most carbon dioxide within minutes of mixing. It is popularly used in pancake mixes. In double acting baking powders, MCP is often combined with the slow acting acid sodium acid pyrophosphate (SAPP).[3]
See also
References
- ↑ 1.0 1.1 Klaus Schrödter, Gerhard Bettermann, Thomas Staffel, Friedrich Wahl, Thomas Klein, Thomas Hofmann "Phosphoric Acid and Phosphates" in Ullmann’s Encyclopedia of Industrial Chemistry 2008, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_465.pub3
- ↑ Gunnar Kongshaug et al. "Phosphate Fertilizers" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_421.pub2
- ↑ John Brodie, John Godber "Bakery Processes, Chemical Leavening Agents" in Kirk-Othmer Encyclopedia of Chemical Technology 2001, John Wiley & Sons. doi:10.1002/0471238961.0308051303082114.a01.pub2
Further reading
- Havlin, J.L., J.D. Beaton, S.L. Tisdale, and W.L. Nelson. 2005. Soil Fertility and Fertilizers. 7th edn. Pearson Prentice Hall, N.J., ISBN 0130278246
