Mast cell
| Mast cell | |
|---|---|
 Mast cells | |
| Details | |
| Latin | mastocytus |
| Identifiers | |
| Code | TH H2.00.03.0.01010 |
| Anatomical terminology | |
A mast cell (also known as a mastocyte or a labrocyte[1]) is derived from the myeloid stem cell. It is a part of the immune system and contains many granules rich in histamine and heparin. Although best known for their role in allergy and anaphylaxis, mast cells play an important protective role as well, being intimately involved in wound healing and defense against pathogens.[2]
The mast cell is very similar in both appearance and function to the basophil, another type of white blood cell. They differ in that mast cells are tissue resident, e.g., in mucosal tissues, while basophils are found in the blood.[3]
Origin and classification


Mast cells were first described by Paul Ehrlich in his 1878 doctoral thesis on the basis of their unique staining characteristics and large granules. These granules also led him to the incorrect belief that they existed to nourish the surrounding tissue, so he named them Mastzellen (from German Mast, meaning "fattening", as of animals).[4][5] They are now considered to be part of the immune system.
Mast cells are very similar to basophil granulocytes (a class of white blood cells) in blood. Both are granulated cells that contain histamine and heparin, an anticoagulant. Both cells also release histamine upon binding to immunoglobulin E.[6] These similarities have led many to speculate that mast cells are basophils that have "homed in" on tissues. Furthermore they share a common precursor in bone marrow expressing the CD34 molecule. Basophils leave the bone marrow already mature, whereas the mast cell circulates in an immature form, only maturing once in a tissue site. The site an immature mast cell settles in probably determines its precise characteristics.[2] The first in vitro differentiation and growth of a pure population of mouse mast cells has been carried out using conditioned medium derived from concanavalin A-stimulated splenocytes.[7] Later, it was discovered that T cell-derived interleukin 3 was the component present in the conditioned media that was required for mast cell differentiation and growth.[8]
Mast cells in rodents are classically divided into two subtypes: connective tissue-type mast cells and mucosal mast cells. The activities of the latter are dependent on T-cells.[9]
Mast cells are present in most tissues characteristically surrounding blood vessels and nerves, and are especially prominent near the boundaries between the outside world and the internal milieu, such as the skin, mucosa of the lungs, and digestive tract, as well as the mouth, conjunctiva, and nose.[2]
Physiology
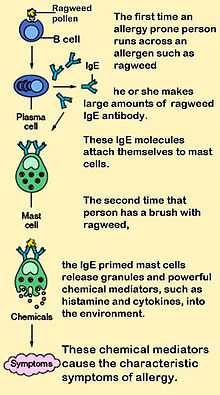
Mast cells play a key role in the inflammatory process. When activated, a mast cell rapidly releases its characteristic granules and various hormonal mediators into the interstitium. Mast cells can be stimulated to degranulate by direct injury (e.g., physical or chemical [such as opioids, alcohols, and certain antibiotics such as polymyxins]), cross-linking of immunoglobulin E (IgE) receptors, or complement proteins.[2]
Mast cells express a high-affinity receptor (FcεRI) for the Fc region of IgE, the least-abundant member of the antibodies. This receptor is of such high affinity that binding of IgE molecules is in essence irreversible. As a result, mast cells are coated with IgE, which is produced by plasma cells (the antibody-producing cells of the immune system). IgE molecules, like all antibodies, are specific to one particular antigen.
In allergic reactions, mast cells remain inactive until an allergen binds to IgE already in association with the cell (see above). Other membrane activation events can either prime mast cells for subsequent degranulation or act in synergy with FcεRI signal transduction.[10] In general, allergens are proteins or polysaccharides. The allergen binds to the antigen-binding sites, which are situated on the variable regions of the IgE molecules bound to the mast cell surface. It appears that binding of two or more IgE molecules (cross-linking) is required to activate the mast cell. The clustering of the intracellular domains of the cell-bound Fc receptors, which are associated with the cross-linked IgE molecules, causes a complex sequence of reactions inside the mast cell that lead to its activation. Although this reaction is most well-understood in terms of allergy, it appears to have evolved as a defense system against intestinal worm infestations (tapeworms, etc.).
The molecules released into the extracellular environment include:[2]
- preformed mediators (from the granules):
- serine proteases, such as tryptase
- histamine (2-5 pg/cell)
- serotonin
- proteoglycans, mainly heparin (active as anticoagulant)
- newly formed lipid mediators (eicosanoids):
- cytokines
- eosinophil chemotactic factor

Histamine dilates post-capillary venules, activates the endothelium, and increases blood vessel permeability. This leads to local edema (swelling), warmth, redness, and the attraction of other inflammatory cells to the site of release. It also depolarizes nerve endings (leading to itching or pain). Cutaneous signs of histamine release are the "flare and wheal"-reaction. The bump and redness immediately following a mosquito bite are a good example of this reaction, which occurs seconds after challenge of the mast cell by an allergen.[2]
The other physiologic activities of mast cells are much less-understood. Several lines of evidence suggest that mast cells may have a fairly fundamental role in innate immunity: They are capable of elaborating a vast array of important cytokines and other inflammatory mediators such as TNFa; they express multiple "pattern recognition receptors" thought to be involved in recognizing broad classes of pathogens; and mice without mast cells seem to be much more susceptible to a variety of infections.
Mast cell granules carry a variety of bioactive chemicals. These granules have been found to be transferred to adjacent cells of the immune system and neurons in a process of transgranulation via mast cell pseudopodia.[11]
Biochemistry
Structure of FcεR1
FcεR1 is a high affinity IgE-receptor that is expressed on the surface of the mast cell. FcεR1 is a tetramer made of one alpha (α) chain, one beta (β) chain, and two identical, disulfide-linked gamma (γ) chains. The binding site for the IgE is formed by the extracellular portion of the α chain that contains two domains that are similar to Ig. One transmembrane domain contains an aspartic acid residue, and one contains a short cytoplasmic tail.[12] The β chain contains, a single immunoreceptor tyrosine-based activation motif ITAM, in the cytoplasmic region. Each γ chain has one ITAM on the cytoplasmic region. The signaling cascade from the receptor is initiated when the ITAMs of the β and γ chains are phosphorylated by tyrosine. This signal is required for the activation of mast cells.[13] Type 2 helper T cells,(Th2) and many other cell types lack the β chain, so signaling is mediated only by the γ chain. This is due to the α chain containing endoplasmic reticulum retention signals that causes the α-chains to remain degraded in the ER. The assembly of the α chain with the co-transfected β and γ chains mask the ER retention and allows the α β γ complex to be exported to the golgi apparatus to the plasma membrane in rats. In humans, only the γ complex is needed to counterbalance the α chain ER retention.[12]
Allergen process
Allergen-mediated FcεR1 cross-linking signals are very similar to the signaling event resulting in antigen binding to lymphocytes. The Lyn tyrosine kinase is associated with the cytoplasmic end of the FcεR1 β chain. The antigen cross-links the FcεR1 molecules, and Lyn tyrosine kinase phosphorylates the ITAMs in the FcεR1 β and γ chain in the cytoplasm. Upon phosphorylation, the Syk tyrosine kinase gets recruited to the ITAMs located on the γ chains. This causes activation of the Syk tyrosine kinase, causing it to phosphorylate.[13] Syk functions as a signal amplifying kinase activity due to the fact that it targets multiple proteins and causes their activation.[14] This antigen stimulated phosphorylation causes the activation of other proteins in the FcεR1-mediated signaling cascade.[15]
Degranulation and fusion
An important adaptor protein activated by the Syk phosphorylation step is the linker for activation of T cells (LAT). LAT is important because it can be modified by phosphorylation to create novel binding sites.[14] Phospholipase C (PLCγ), becomes phosphorylated once bound to LAT, and is then used to catalyze Phosphatidylinositol bisphosphate breakdown to yield inositol trisphosphate (IP3) and diacyglycerol (DAG). IP3 elevates calcium levels, and DAG activates protein kinase C (PKC). This is not the only way that PKC is made. The tyrosine kinase, Fyn, phosphorylates a Grb-2-associated binder-like protein 2 (Gab2) which binds to phosphoinositide 3-kinase which activates PKC.PKC leads to the activation of myosin light-chain phosphorylation granule movements which disassembles the actin-myosin complexes to allow granules to come into contact with the plasma membrane.[13] The mast cell granule can now fuse with the plasma membrane. Soluble N-ethylmaleimide sensitive fusion Attachment Protein Rceptor"SNARE complex mediates this process. Different SNARE proteins interact to form different complexes that catalyze fusion. Rab3 guanosine triphosphatases and Rab-associated kinases and phosphatases regulate the cell granule membrane fusion in resting mast cells.
Enzymes
| Enzyme | Function |
|---|---|
| Lyn tyrosine kinase | Phosphorylates the ITAMs in the FcεR1 β and γ chain in the cytoplasm. It causes Syk tyrosine kinase to get recruited to the ITAMS located on the γ chains. This causes activation of the Syk tyrosine kinase, causing it to phosphorylate |
| Syk tyrosine kinase | Targets multiple proteins and causes their activation |
| Phospholipase C | Catalyzes Phosphatidylinositol 4,5-bisphosphate |
| Inositol trisphosphate | Elevates calcium levels |
| Diacylglycerol | Activates protein kinase C |
| Fyn | Phosphorylates Grb-2-associated-binder-like-protein |
| Grb-2-associated-binder-like-protein | Binds to phosphoinositide 3-kinase |
| Phosphoinositide 3-kinase | Activates protein kinase C |
| Protein kinase C | Activates myosin light-chain phosphorylation granule movements that dissemble the actin-myosin complexes |
| Rab-associated kinases and phosphatases | Regulate cell granule membrane fusion in resting mast cells |
Role in disease
Allergic disease
Many forms of cutaneous and mucosal allergy are mediated in large part by mast cells; they play a central role in asthma, eczema, itch (from various causes), and allergic rhinitis and allergic conjunctivitis. Antihistamine drugs act by blocking histamine action on nerve endings. Cromoglicate-based drugs (sodium cromoglicate, nedocromil) block a calcium channel essential for mast cell degranulation, stabilizing the cell and preventing release of histamine and related mediators. Leukotriene antagonists (such as montelukast and zafirlukast) block the action of leukotriene mediators and are being used increasingly in allergic diseases.[2]
Calcium triggers the secretion of histamine from mast cells after previous exposure to sodium fluoride. The secretory process can be divided into a fluoride-activation step and a calcium-induced secretory step. It was observed that the fluoride-activation step is accompanied by an elevation of cAMP levels within the cells. The attained high levels of cAMP persist during histamine release. It was further found that catecholamines do not markedly alter the fluoride-induced histamine release. It was also confirmed that the second, but not the first, step in sodium fluoride-induced histamine secretion is inhibited by theophylline.[16] Vasodilation and increased permeability of capillaries are a result of both H1 and H2 receptor types.[17]
Stimulation of histamine activates a histamine (H2)-sensitive adenylate cyclase of oxyntic cells, and there is a rapid increase in cellular [cAMP] that is involved in activation of H+ transport and other associated changes of oxyntic cells.[18]
Anaphylaxis
In anaphylaxis (a severe systemic reaction to allergens, such as nuts, bee stings, or drugs), body-wide degranulation of mast cells leads to vasodilation and, if severe, symptoms of life-threatening shock.
Histamine is a vasodilatory substance released during anaphylaxis.[17]
Autoimmunity
Mast cells may be implicated in the pathology associated with autoimmune, inflammatory disorders of the joints. They have been shown to be involved in the recruitment of inflammatory cells to the joints (e.g., rheumatoid arthritis) and skin (e.g., bullous pemphigoid), and this activity is dependent on antibodies and complement components.[19]
Reproductive disorders
Mast cells are present within the endometrium, with increased activation and release of mediators in endometriosis.[20] In males, mast cells are present in the testes and are increased in oligo- and azoospermia, with mast cell mediators directly suppressing sperm motility in a potentially reversible manner.[20]
Mast cell disorders
Mastocytosis is a rare condition featuring proliferation of mast cells. It exists in a cutaneous and systemic form, with the former being limited to the skin and the latter involving multiple organs.[2] Mast cell tumors are often seen in dogs and cats.[21]
Autism
Research into an immunological contribution to autism suggests that autism spectrum disorder (ASD) children may present with "allergic-like" problems in the absence of elevated serum IgE and chronic urticaria, suggesting non-allergic mast cell activation in response to environmental and stress triggers. This mast cell activation could contribute to brain inflammation and neurodevelopmental problems.[22]
Histological staining
Toluidine blue: one of the most common stains for acid mucopolysaccharides and glycoaminoglycans, components of mast cells granules.[23]
Surface markers: cell surface markers of mast cells were discussed in detail by Heneberg,[24] claiming that mast cells may be inadverently included in the stem or progenitor cell isolates, since part of them is positive for the CD34 antigen. The classical mast cell markers include the high-affinity IgE receptor, CD117 (c-Kit), and CD203c (for most of the mast cell populations). Expression of some molecules may change in course of the mast cell activation.[25]
References
- ↑ "labrocytes". Memidex.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Prussin C, Metcalfe DD (February 2003). "4. IgE, mast cells, basophils, and eosinophils". The Journal of Allergy and Clinical Immunology 111 (2 Suppl): S486–94. doi:10.1067/mai.2003.120. PMID 12592295.
- ↑ http://bitesized.immunology.org/what-is-immunology
- ↑ Ehrlich, P. (1878). Beiträge zur Theorie und Praxis der histologischen Färbung [Contribution to the theory and practice of histological dyes] (Dissertation) (in German). Leipzig University. OCLC 63372150.
- ↑ http://www.mondofacto.com/facts/dictionary?mastocyte[]
- ↑ Marieb, Elaine N.; Hoehn, Katja (2007). Human Anatomy and Physiology (7th ed.). San Francisco: Pearson Benjamin Cummings. p. 659. ISBN 978-0-8053-5910-7.
- ↑ Razin E, Cordon-Cardo C, Good RA (April 1981). "Growth of a pure population of mouse mast cells in vitro with conditioned medium derived from concanavalin A-stimulated splenocytes". Proceedings of the National Academy of Sciences of the United States of America 78 (4): 2559–61. doi:10.1073/pnas.78.4.2559. PMC 319388. PMID 6166010.
- ↑ Razin E, Ihle JN, Seldin D et al. (March 1984). "Interleukin 3: A differentiation and growth factor for the mouse mast cell that contains chondroitin sulfate E proteoglycan". Journal of Immunology 132 (3): 1479–86. PMID 6198393.
- ↑ Denburg, Judah A. (1998). Allergy and allergic diseases: the new mechanisms and therapeutics. Totowa, NJ: Humana Press. ISBN 0-89603-404-6.
- ↑ Pulendran B, Ono SJ (May 2008). "A shot in the arm for mast cells". Nat. Med. 14 (5): 489–90. doi:10.1038/nm0508-489. PMID 18463655.
- ↑ Wilhelm M, Silver R, Silverman AJ (November 2005). "Central nervous system neurons acquire mast cell products via transgranulation". The European Journal of Neuroscience 22 (9): 2238–48. doi:10.1111/j.1460-9568.2005.04429.x. PMC 3281766. PMID 16262662.
- ↑ 12.0 12.1 Kinet JP (1999). "The high-affinity IgE receptor (FcεRI): from physiology to pathology". Annual Review of Immunology 17: 931–72. doi:10.1146/annurev.immunol.17.1.931. PMID 10358778.
- ↑ 13.0 13.1 13.2 Abbas, Abul K.; Lichtman, Andrew H. H.; Pillai, Shiv (2011). "Role of Mast Cells, Basophils and Eosinophils in Immediate Hypersensitivity". Cellular and Molecular Immunology (7th ed.). New York, NY: Elsevier. ISBN 978-1-4377-1528-6.
- ↑ 14.0 14.1 Rivera J, Cordero JR, Furumoto Y et al. (September 2002). "Macromolecular protein signaling complexes and mast cell responses: a view of the organization of IgE-dependent mast cell signaling". Molecular Immunology 38 (16-18): 1253–8. doi:10.1016/S0161-5890(02)00072-X. PMID 12217392.
- ↑ Li W, Deanin GG, Margolis B, Schlessinger J, Oliver JM (July 1992). "FcεR1-mediated tyrosine phosphorylation of multiple proteins, including phospholipase Cγ1 and the receptor βγ2 complex, in RBL-2H3 rat basophilic leukemia cells". Molecular and Cellular Biology 12 (7): 3176–82. PMC 364532. PMID 1535686.
- ↑ Alm PE (April 1983). "Sodium fluoride evoked histamine release from mast cells. A study of cyclic AMP levels and effects of catecholamines". Agents and Actions 13 (2-3): 132–7. doi:10.1007/bf01967316. PMID 6191542.
- ↑ 17.0 17.1 Dachman WD, Bedarida G, Blaschke TF, Hoffman BB (March 1994). "Histamine-induced venodilation in human beings involves both H1 and H2 receptor subtypes". The Journal of Allergy and Clinical Immunology 93 (3): 606–14. doi:10.1016/S0091-6749(94)70072-9. PMID 8151062.
- ↑ Machen TE, Rutten MJ, Ekblad EB (February 1982). "Histamine, cAMP, and activation of piglet gastric mucosa". The American Journal of Physiology 242 (2): G79–84. PMID 6175225.
- ↑ Lee DM, Friend DS, Gurish MF, Benoist C, Mathis D, Brenner MB (September 2002). "Mast cells: a cellular link between autoantibodies and inflammatory arthritis". Science 297 (5587): 1689–92. doi:10.1126/science.1073176. PMID 12215644.
- ↑ 20.0 20.1 Menzies FM, Shepherd MC, Nibbs RJ, Nelson SM (2011). "The role of mast cells and their mediators in reproduction, pregnancy and labour". Human Reproduction Update 17 (3): 383–96. doi:10.1093/humupd/dmq053. PMID 20959350.
- ↑ "Cutaneous Mast Cell Tumors". The Merck Veterinary Manual. 2006. Retrieved 2007-07-08.
- ↑ Theoharides TC, Angelidou A, Alysandratos KD et al. (January 2012). "Mast cell activation and autism". Biochimica Et Biophysica Acta 1822 (1): 34–41. doi:10.1016/j.bbadis.2010.12.017. PMID 21193035.
- ↑ Blumenkrantz N, Asboe-Hansen G (May 1975). "A selective stain for mast cells". The Histochemical Journal 7 (3): 277–82. doi:10.1007/BF01003596. PMID 47855.
- ↑ Heneberg P (November 2011). "Mast cells and basophils: trojan horses of conventional lin- stem/progenitor cell isolates". Current Pharmaceutical Design 17 (34): 3753–71. doi:10.2174/138161211798357881. PMID 22103846.
- ↑ Lebduska P, Korb J, Tůmová M, Heneberg P, Dráber P (December 2007). "Topography of signaling molecules as detected by electron microscopy on plasma membrane sheets isolated from non-adherent mast cells". Journal of Immunological Methods 328 (1-2): 139–51. doi:10.1016/j.jim.2007.08.015. PMID 17900607.
External links
- Mast cells at the US National Library of Medicine Medical Subject Headings (MeSH)
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||