Lysine carbamoyltransferase
In enzymology, a lysine carbamoyltransferase (EC 2.1.3.8) is an enzyme that catalyzes the chemical reaction
- carbamoyl phosphate + L-lysine
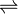 phosphate + L-homocitrulline
phosphate + L-homocitrulline
Thus, the two substrates of this enzyme are carbamoyl phosphate and L-lysine, whereas its two products are phosphate and L-homocitrulline.
This enzyme belongs to the family of transferases that transfer one-carbon groups, specifically the carboxy- and carbamoyltransferases. The systematic name of this enzyme class is carbamoyl-phosphate:L-lysine carbamoyltransferase. This enzyme is also called lysine transcarbamylase.
References
- Hommes FA, Eller AG, Scott DF, Carter AL (1983). "Separation of ornithine and lysine activities of the ornithine-transcarbamylase-catalyzed reaction". Enzyme. 29 (4): 271–7. PMID 6409607.
|
|---|
| | 2.1.1: Methyl- | |
|---|
| 2.1.2: Hydroxymethyl-,
Formyl- and Related | | Hydroxymethyltransferase | |
|---|
| | Formyltransferase | |
|---|
| | Other | |
|---|
|
|---|
| | 2.1.3: Carboxy- and Carbamoyl | |
|---|
| | 2.1.4: Amidino |
- Arginine:glycine amidinotransferase
|
|---|
|
- Biochemistry overview
- Enzymes overview
- By EC number: 1.1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 10
- 11
- 13
- 14
- 15-18
- 2.1
- 3.1
- 4.1
- 5.1
- 6.1-3
|
|
|
|
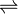 phosphate + L-homocitrulline
phosphate + L-homocitrulline