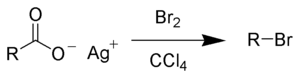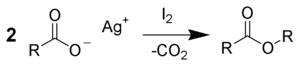Hunsdiecker reaction
The Hunsdiecker reaction (also called the Borodin reaction after Alexander Borodin) is the organic reaction of silver salts of carboxylic acids with halogens to give organic halides.[1][2][3][4] It is an example of a halogenation reaction. The reaction is named after Heinz Hunsdiecker and Cläre Hunsdiecker.
Several reviews have been published.[5][6]
Mercuric oxide will also effect this transformation.[7][8]
Reaction mechanism
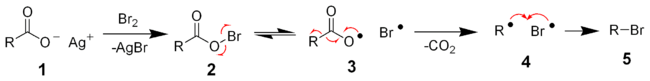
The reaction mechanism of the Hunsdiecker reaction is believed to involve organic radical intermediates. The silver salt of the carboxylic acid 1 will quickly react with bromine to form the acyl hypohalite intermediate 2. Formation of the diradical pair 3 allows for radical decarboxylation to form the diradical pair 4, which will quickly recombine to form the desired organic halide 5.
Variations
Simonini reaction
The reaction of silver salts of carboxylic acids with iodine is called the Simonini reaction, named after Angelo Simonini, a student of Adolf Lieben at the University of Vienna. The ratio of the reagents play an important role in the determination of products, namely, if 1:1 ratio of salt and iodine is used alkyl iodide is formed. On the other hand, a 2:1 ratio gives RCOOR. and 3:2 ratio gives both the products.[5][9][10]
See also
References
- ↑ Cläre Hunsdiecker, et al. U.S. Patent # 2,176,181.
- ↑ Heinz Hunsdiecker; Cläre Hunsdiecker (1942). "Über den Abbau der Salze aliphatischer Säuren durch Brom". Ber. 75 (3): 291–297. doi:10.1002/cber.19420750309.
- ↑ Borodin, A. (1861). "Ueber Bromvaleriansäure und Brombuttersäure". Ann. 119: 121–123. doi:10.1002/jlac.18611190113.
- ↑ Allen, C. F. H.; Wilson, C. V. (1955). "Methyl 5-bromovalerate". Org. Synth.; Coll. Vol. 3, p. 578
- ↑ 5.0 5.1 Johnson, R. G.; Ingham, R. K. (1956). "The Degradation of Carboxylic Acid Salts by Means of Halogen - the Hunsdiecker Reaction". Chem. Rev. 56 (2): 219–269. doi:10.1021/cr50008a002.
- ↑ Wilson, C. V. Org. React. 1957, 9, 341. (Review)
- ↑ Meek, J. S.; Osuga, D. T. (1973). "Bromocyclopropane". Org. Synth.; Coll. Vol. 5, p. 126
- ↑ Lampman, G. M.; Aumiller, J. C. (1988). "Mercury(II) oxide-modified Hunsdiecker reaction: 1-Bromo-3-chlorocyclobutane". Org. Synth.; Coll. Vol. 6, p. 179
- ↑ Simonini, A. (1892). "Über den Abbau der fetten Säuren zu kohlenstoffärmeren Alkoholen". Monatshefte für Chemie 13 (1): 320–325. doi:10.1007/BF01523646.
- ↑ Simonini, A. (1893). "Über den Abbau der fetten Säuren zu kohlenstoffärmeren Alkoholen". Monatshefte für Chemie 14 (1): 81–92. doi:10.1007/BF01517859.