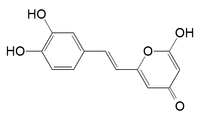
Hispidin
 | |
| Names | |
|---|---|
| IUPAC name
2-[(E)-2-(3,4-dihydroxyphenyl)ethenyl]-6-hydroxypyran-4-one | |
| Other names
6-(3,4-dihydroxystyrl)-4-hydroxy-2-pyrone | |
| Identifiers | |
| 555-55-5 | |
| ChemSpider | 13975015 |
| |
| Jmol-3D images | Image Image |
| PubChem | 5353671 |
| |
| Properties | |
| Molar mass | 246.21 g/mol |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Hispidin is a natural substance. It can also be synthetised.[1]
Hispidin 4-O-β-d-glucopyranoside can be found in Pteris ensiformis[2] whereas hispidin derivatives, such as phellibaumins, can be found in the edible mushroom Inonotus xeranticus[3] or Phellinus.[4][5]
See also
References
- ↑ Gonindard, C.; Bergonzi, C.; Denier, C.; Sergheraert, C.; Klaebe, A.; Chavant, L.; Hollande, E. (1997). "Synthetic hispidin, a PKC inhibitor, is more cytotoxic toward cancer cells than normal cells in vitro". Cell Biology and Toxicology 13 (3): 141–53. doi:10.1023/A:1007321227010. PMID 9088624.
- ↑ Identification of phenolic antioxidants from Sword Brake fern (Pteris ensiformis Burm.). Yung-Husan Chen, Fang-Rong Chang, Yih-Jer Lin, Lisu Wang, Jinn-Fen Chen, Yang-Chang Wu and Ming-Jiuan Wu, Food Chemistry, Volume 105, Issue 1, 2007, pp. 48-56, doi:10.1016/j.foodchem.2007.03.055
- ↑ Hispidin Derivatives from the Mushroom Inonotus xeranticus and Their Antioxidant Activity. In-Kyoung Lee, Soon-Ja Seok, Wan-Kyu Kim and Bong-Sik Yun, J. Nat. Prod., 2006, 69 (2), pp. 299–301, doi:10.1021/np050453n
- ↑ Lee, In-Kyoung; Yun, Bong-Sik (2007). "Highly oxygenated and unsaturated metabolites providing a diversity of hispidin class antioxidants in the medicinal mushrooms Inonotus and Phellinus". Bioorganic & Medicinal Chemistry 15 (10): 3309–14. doi:10.1016/j.bmc.2007.03.039. PMID 17387019.
- ↑ Lee, Yeon Sil; Kang, Young-Hee; Jung, Ju-Young; Lee, Sanghyun; Ohuchi, Kazuo; Shin, Kuk Hyun; Kang, Il-Jun; Park, Jung Han Yoon et al. (October 2008). "Protein glycation inhibitors from the fruiting body of Phellinus linteus" ([DEAD LINK]). Biological & Pharmaceutical Bulletin 31 (10): 1968–72. doi:10.1248/bpb.31.1968. PMID 18827365.