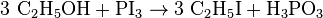Ethyl iodide
| | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Iodoethane[1] | |||
| Identifiers | |||
| 505934 | |||
| 75-03-6 | |||
| ChEBI | CHEBI:42487 | ||
| ChEMBL | ChEMBL1232588 | ||
| ChemSpider | 6100 | ||
| EC number | 200-833-1 | ||
| |||
| Jmol-3D images | Image | ||
| PubChem | 6340 | ||
| RTECS number | KI4750000 | ||
| |||
| Properties | |||
| Molecular formula |
C2H5I | ||
| Molar mass | 155.97 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 1.940 g mL−1 | ||
| Melting point | −111.10 °C; −167.98 °F; 162.05 K | ||
| Boiling point | 71.5 °C; 160.6 °F; 344.6 K | ||
| 4 g L−1 (at 20 °C) | |||
| Solubility in ethanol | Miscible | ||
| Solubility in diethyl ether | Miscible | ||
| log P | 2.119 | ||
| Vapor pressure | 17.7 kPa | ||
| Henry's law constant (kH) |
1.8 μmol Pa−1 kg−1 | ||
| Refractive index (nD) |
1.513–1.514 | ||
| Viscosity | 5.925 mPa s (at 20 °C) | ||
| Thermochemistry | |||
| Specific heat capacity (C) |
109.7 J K−1 mol−1 | ||
| Std enthalpy of formation (ΔfH |
−39.9–−38.3 kJ mol−1 | ||
| Std enthalpy of combustion (ΔcH |
−1.4629–−1.4621 MJ mol−1 | ||
| Hazards | |||
| GHS pictograms |   | ||
| GHS signal word | DANGER | ||
| H302, H315, H317, H319, H334, H335 | |||
| P261, P280, P305+351+338, P342+311 | |||
| EU classification | | ||
| R-phrases | R20, R36/37/38, R42/43 | ||
| S-phrases | S23, S26, S36/37, S45 | ||
| NFPA 704 | |||
| Flash point | 72 °C (162 °F; 345 K) | ||
| LD50 (Median lethal dose) |
330 g m−3 (oral, rat) | ||
| Related compounds | |||
| Related iodoalkanes |
|||
| Related compounds |
|||
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |||
| | |||
| Infobox references | |||
Ethyl iodide (also iodoethane) is a colorless, flammable chemical compound. It has the chemical formula C2H5I and is prepared by heating ethanol with iodine and phosphorus.[2] On contact with air, especially on the effect of light, it decomposes and turns yellow or reddish from dissolved iodine. It may also be prepared by reaction between hydroiodic acid and ethanol distilling off the ethyl iodide. Ethyl iodide should be stored in copper powder to avoid fast decomposition, though even with this method samples don't last more than 1 year.

Because iodide is a good leaving group, ethyl iodide is an excellent ethylating agent. It is also used as the hydrogen radical promoter.
Production
Ethyl iodide is prepared by using red phosphorus, ethanol and iodine. There is absolute ethanol and iodine dust used in the process, which contains solving the phosphorus and iodine in the ethanol. During the process the temperature is controlled. The phosphorus triiodide is made in situ, due to the instability of it.[3]
The crude product is purified by distillation.
References
- ↑ "iodoethane - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. Retrieved 29 February 2012.
- ↑ Merck Index of Chemicals and Drugs, 9th ed., monograph 3753
- ↑ Csámpai,A;Láng,E;Majer,Zs;Orosz,Gy;Rábai,J;Ruff,F;Schlosser,G;Szabó,D;Vass,E: Szerves Kémiai Praktikum page: 274, Eötvös kiadó 2012 ISBN 978-963-312-129-0