Dihydropyrimidinase
| dihydropyrimidinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 3.5.2.2 | ||||||||
| CAS number | 9030-74-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||
In enzymology, a dihydropyrimidinase (EC 3.5.2.2) is an enzyme that catalyzes the chemical reaction
- 5,6-dihydrouracil + H2O
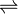 3-ureidopropanoate
3-ureidopropanoate
Thus, the two substrates of this enzyme are 5,6-dihydrouracil and H2O, whereas its product is 3-ureidopropanoate.
This enzyme belongs to the family of hydrolases, those acting on carbon-nitrogen bonds other than peptide bonds, specifically in cyclic amides. The systematic name of this enzyme class is 5,6-dihydropyrimidine amidohydrolase. Other names in common use include hydantoinase, hydropyrimidine hydrase, hydantoin peptidase, pyrimidine hydrase, and D-hydantoinase. This enzyme participates in 3 metabolic pathways: pyrimidine metabolism, beta-alanine metabolism, and pantothenate and coa biosynthesis.
Structural studies
As of late 2007, 10 structures have been solved for this class of enzymes, with PDB accession codes 1GKP, 1GKQ, 1GKR, 1NFG, 1YNY, 2FTW, 2FTY, 2FVK, 2FVM, and 2GSE.
References
- Brooks KP, Jones EA, Kim BD, Sander EG (1983). "Bovine liver dihydropyrimidine amidohydrolase: purification, properties, and characterization as a zinc metalloenzyme". Arch. Biochem. Biophys. 226 (2): 469–83. doi:10.1016/0003-9861(83)90316-8. PMID 6639068.
- EADIE GS, BERNHEIM F, BERNHEIM ML (1949). "The partial purification and properties of animal and plant hydantoinases". J. Biol. Chem. 181 (2): 449–58. PMID 15393763.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||