Cyphostemmin B
Cyphostemmin B
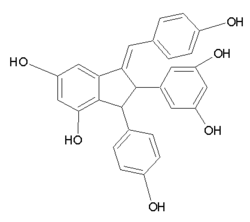 |
| Identifiers |
| Jmol-3D images |
Image |
Oc1ccc(cc1)/C=C4/c2cc(O)cc(O)c2C(c3ccc(O)cc3)C4c5cc(O)cc(O)c5
|
| Properties |
| |
C28H22O6 |
| Molar mass |
454.47 g/mol |
Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) |
|
| Infobox references |
|
|
Cyphostemmin B is an oligostilbene found in Cyphostemma crotalarioides (Vitaceae).[1] It is a resveratrol dimer.
References
- ↑ Cyphostemmins A-B, two new antifungal oligostilbenes from Cyphostemma crotalarioides (Vitaceae). Paul-Henri Ducrot, Albert Kollmann, Adil E. Bala, Amel Majira, Lucien Kerhoas, Robert Delorme and Jacques Einhorn, Tetrahedron Letters, Volume 39, Issue 52, 24 December 1998, Pages 9655–9658, doi:10.1016/S0040-4039(98)02207-2
|
|---|
|
- Diptoindonesin C
- Diptoindonesin F
- Gnetin H
- Hemsleyanol D
- Isohopeaphenol
- Laetevirenol A, B, C, D and E
- Suffruticosol A and B
- Viniferal
- E-ω-viniferin
- Z-ω-viniferin
| | | Dimers |
- Diptoindonesin G
- Jezonodione
- B
- Scirpusin A
- Tibeticanol (piceatannol dimer)
|
|---|
| | Trimers |
- Amurensin B
- Gnetin E
- Gneyulin A
- Johorenol A
- Ampelopsin E
- Vaticanol G
|
|---|
| | Tetramers: |
- Dibalanocarpol
- Gnetin J (3"-hydroxygnetin E)
- Gnetin K (3"-methoxygnetin E)
- Gnetuhainin R (isorhapontigenin tetramer)
- Laetevirenol F and G
|
|---|
| Higher polymers
(five units or more) | |
|---|
| Oligomeric forms
of resveratrol | Dimers | |
|---|
| Trimers | |
|---|
| Tetramers | |
|---|
| Pentamers | |
|---|
| Hexamers | |
|---|
| Higher polymers |
- γ-viniferin
- Valeriaphenol A
|
|---|
|
|---|
| | Glycosides or conjugates |
- Diptoindonesin A (C-glucoside of ε-viniferin)
- Foeniculoside I (glucoside of miyabenol C), II, III and IV
- Laevifonol (an ε-viniferin-ascorbic acid hybrid compound)
- Laevifoside (O-glucoside of ampelopsin A)
|
|---|
|
