Cinoxate
 | |
| Names | |
|---|---|
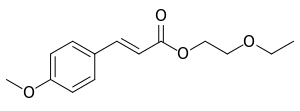
| IUPAC name
2-Ethoxyethyl 3-(4-methoxyphenyl)propenoate | |
| Other names
2-Ethoxyethyl p-methoxycinnamate | |
| Identifiers | |
| 104-28-9 | |
| ChEMBL | ChEMBL2104045 |
| ChemSpider | 4523729 |
| |
| Jmol-3D images | Image |
| KEGG | D03512 |
| PubChem | 5373773 |
| |
| Properties | |
| Molecular formula |
C14H18O4 |
| Molar mass | 250.29 g·mol−1 |
| Density | 1.102 g/cm3 |
| Melting point | −25 °C (−13 °F; 248 K) |
| Boiling point | 184 °C (363 °F; 457 K) at 2 mmHg |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Cinoxate is an organic compound used as an ingredient in some types of sunscreens. It is an ester formed from methoxycinnamic acid and 2-ethoxyethanol. It is a slightly yellow viscous liquid that is insoluble in water, but miscible with alcohols, esters, and vegetable oils.
It protects skin against the sun by absorbing UV-A and UV-B rays.
See also
- Amiloxate, another methoxycinnamate-based sunscreen
- Octyl methoxycinnamate, another methoxycinnamate-based sunscreen
References
- ↑ Merck Index, 11th Edition, 2312.
| ||||||||||||||||||||||