Cimicoxib
 | |
| Names | |
|---|---|
| IUPAC name
4-[4-Chloro-5-(3-fluoro-4-methoxyphenyl)-1H-imidazol-1-yl]benzenesulfonamide | |
| Identifiers | |
| ATCvet code | QM01 |
| 265114-23-6 | |
| ChEBI | CHEBI:76127 |
| ChEMBL | ChEMBL435381 |
| ChemSpider | 184745 |
| |
| Jmol-3D images | Image |
| PubChem | 213053 |
| |
| Properties | |
| Molecular formula |
C16H13ClFN3O3S |
| Molar mass | 381.81 g·mol−1 |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
Cimicoxib (UR-8880 trade name Cimalgex) is a non-steroidal anti-inflammatory drug (NSAID) used in veterinary medicine to treat dogs for pain and inflammation associated with osteoarthritis and for the management of pain and inflammation associated with surgery.[1] It acts as a COX-2 inhibitor.
Synthesis
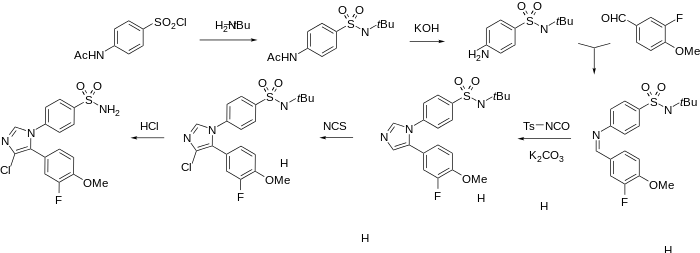
Cimicoxib synthesis:[2]
Reaction of the imine with tosylisocyanate in the presence of potassium carbonate leads to what may be viewed as 2+3 cycloaddition of the nitrogen analogue of a ketene to form the imidazole ring.
References
- ↑ "European Public Assessment Report: Cimalgex (cimicoxib)". European Medicines Agency.
- ↑ Almansa, C.; Alfón, J.; De Arriba, A. F.; Cavalcanti, F. L.; Escamilla, I.; Gómez, L. A.; Miralles, A.; Soliva, R.; Bartrolí, J.; Carceller, E.; Merlos, M.; García-Rafanell, J. N. (2003). "Synthesis and Structure−Activity Relationship of a New Series of COX-2 Selective Inhibitors: 1,5-Diarylimidazoles". Journal of Medicinal Chemistry 46 (16): 3463. doi:10.1021/jm030765s.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||