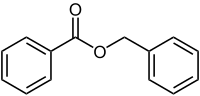
Benzyl benzoate
 | |
 | |
| Names | |
|---|---|
| Other names
Ascabin, Ascabiol, Ascarbin, Benzylate, Scabanca, Tenutex, Vanzoate, Venzoate, Benzoic acid phenylmethyl ester, Benzy alcohol benzoic ester | |
| Identifiers | |
| ATC code | P03 QP53AX11 |
| 120-51-4 | |
| ChEBI | CHEBI:41237 |
| ChEMBL | ChEMBL1239 |
| ChemSpider | 13856959 |
| DrugBank | DB02775 |
| |
| Jmol-3D images | Image |
| KEGG | D01138 |
| PubChem | 2345 |
| |
| UNII | N863NB338G |
| Properties | |
| Molecular formula |
C14H12O2 |
| Molar mass | 212.24 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | faint aromatic |
| Density | 1.118 g/cm3 |
| Melting point | 18 °C (64 °F; 291 K) |
| Boiling point | 323 °C (613 °F; 596 K) |
| insoluble | |
| Solubility | miscible in alcohol, chloroform, ether, oils soluble in acetone, benzene insoluble in glycerol |
| Refractive index (nD) |
1.5681 (21 °C) |
| Hazards | |
| EU classification | Harmful (Xn) |
| NFPA 704 | |
| Flash point | 158 °C (316 °F) (closed cup) |
| 481 °C (898 °F; 754 K) | |
| LD50 (Median lethal dose) |
1700 mg/kg (rat, oral) |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
Benzyl benzoate is the organic compound with the formula C6H5CH2O2CC6H5. It is the ester of benzyl alcohol and benzoic acid. It forms either a viscous liquid or solid flakes and has a weak, sweet-balsamic odor. It occurs in a number of blossoms (e. g. tuberose, hyacinth) and is a component of Balsam of Peru and Tolu balsam.[1][2]
It is on the World Health Organization's List of Essential Medicines, a list of the most important medication needed in a basic health system.[3]
Uses
Benzyl benzoate is used as an acaricide, scabicide, and pediculicide in veterinary hospitals. It is also a repellent for chiggers, ticks, and mosquitoes.[4] It is an effective and inexpensive topical treatment for human scabies.[5] It has vasodilating and spasmolytic effects and is present in many asthma and whooping cough drugs.[6]
Other uses of benzyl benzoate are dye carrier, solvent for cellulose derivatives, plasticizer, and fixative in the perfume industry.[1]
Side effects
Benzyl benzoate has low acute toxicity in laboratory animals. It is rapidly hydrolyzed to benzoic acid and benzyl alcohol, which are subsequently metabilized to benzoic acid. The conjugates of benzoic acid are rapidly eliminated in urine. When given in large doses to laboratory animals, benzyl benzoate can cause hyperexcitation, incoordination, ataxia, convulsions, and respiratory paralysis.[4]
Benzyl benzoate is irritant to the skin.[5]
Production
Benzyl benzoate is produced industrially by the reaction of sodium benzoate with benzyl alcohol in the presence of a base. Alternatively, it is produced by transesterification of methylbenzoate and benzyl alcohol.[6] It is also a byproduct of benzoic acid synthesis by toluene oxidation.[1] It can also be generated by the Tishchenko reaction of benzaldehyde, using sodium benzylate and aluminium benzylate as catalysts:[7]
- 2 PhCHO → PhCH2O2CPh
References
- ↑ 1.0 1.1 1.2 Takao Maki et al. (2007), "Benzoic Acid and Derivatives", Ullman's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 6
- ↑ Karl-Georg Fahlbusch et al. (2007), "Flavors and Fragrances", Ullman's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 59
- ↑ "WHO Model List of EssentialMedicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ↑ 4.0 4.1 Jamaluddin Shaikh (2005), "Benzyl Benzoate", in Philip Wexler, Encyclopedia of Toxicology 1 (2nd ed.), Elsevier, pp. 264–265
- ↑ 5.0 5.1 D.A. Burns (2010), "Diseases Caused by Arthropods and Other Noxious Animals", in Tony Burns et al., Rook's Textbook of Dermatology 2 (8th ed.), Wiley-Blackwell, p. 38.41
- ↑ 6.0 6.1 Friedrich Brühne, Elaine Wright (2007), "Benzyl Alcohol", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 7
- ↑ Friedrich Brühne, Elaine Wright (2007), "Benzaldehyde", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 3
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
