BamHI
| BamHI | |||||||||
|---|---|---|---|---|---|---|---|---|---|
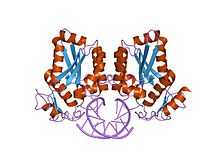 restriction endonuclease BamHI bound to a non-specific dna. | |||||||||
| Identifiers | |||||||||
| Symbol | BamHI | ||||||||
| Pfam | PF02923 | ||||||||
| Pfam clan | CL0236 | ||||||||
| InterPro | IPR004194 | ||||||||
| SCOP | 1bhm | ||||||||
| SUPERFAMILY | 1bhm | ||||||||
| |||||||||
BamHI (from Bacillus amyloliquefaciens) is a type II restriction endonuclease, having the capacity for recognizing short sequences (6 b.p.) of DNA and specifically cleaving them at a target site. This exhibit focuses on the structure-function relations of BamHI as described by Newman, et al. (1995). BamHI binds at the recognition sequence 5'-GGATCC-3', and cleaves these sequences just after the 5'-guanine on each strand. This cleavage results in sticky ends which are 4 b.p. long. In its unbound form, BamHI displays a central b sheet, which resides in between a helices. BamHI is an extraordinarily unique molecule in that it undergoes a series of unconventional conformational changes upon DNA recognition. This allows the DNA to maintain its normal B-DNA conformation without distorting to facilitate enzyme binding. BamHI is a symmetric dimer. DNA is bound in a large cleft that is formed between dimers; the enzyme binds in a "crossover" manner. Each BamHI subunit makes the majority of its backbone contacts with the phosphates of a DNA half site but base pair contacts are made between each BamHI subunit and nitrogenous bases in the major groove of the opposite DNA half site. The protein binds the bases through either direct hydrogen bonds or water-mediated H-bonds between the protein and every H-bond donor/acceptor group in the major groove. Major groove contacts are formed by atoms residing on the amino-terminus of a parallel 4 helix bundle. This bundle marks the BamHI dimer interface, and it is thought that the dipole moments of the NH2-terminal atoms on this bundle may contribute to electrostatic stabilization.
Sites of Recognition Between BamHI and DNA
The BamHI enzyme is capable of making a large number of contacts with DNA. Water-mediated hydrogen bonding, as well as both main-chain and side-chain interactions aid in binding of the BamHI recognition sequence. In the major groove, the majority of enzyme/DNA contacts take place at the amino terminus of the parallel-4-helix bundle, made up of a4 and a6 from each subunit. Although a6 from each subunit does not enter the DNA major groove, its preceding loops interact with the outer ends of the recognition site. Conversely, a4 from each subunit does enter the major groove in the center of the recognition sequence. A total of 18 bonds are formed between the enzyme and DNA across the 6 base pair recognition sequence (12 direct and 6 water mediated bonds). As discussed above, the L and R subunits bind in a cross over manner, whereby the R-subunit of BamHI contacts the left DNA half-site of the recognition sequence. The binding of each BamHI subunit is precisely the same as its symmetrical partner. The recognition site for BamHI has a palindromic sequence which can be cut in half for ease in showing bonds.
Recognition site
G G A T C C C C T A G G
As of the end of 2010, there were 5 crystal structures of BamH1 in the Protein Data Bank
References
- M. Newman, T. Strzelecka, F.D. Dorner, I. Schildkraut, A. K. Aggarwal, Science. 269, 656 (1995)
External links
- Deoxyribonuclease BamHI at the US National Library of Medicine Medical Subject Headings (MeSH)
- 5 crystal structures
This article incorporates text from the public domain Pfam and InterPro IPR004194