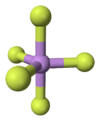
Arsenic pentafluoride
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Arsenic pentafluoride | |||
| Other names
Arsenic(V) fluoride, Arsorane, pentafluoro- | |||
| Identifiers | |||
| 7784-36-3 | |||
| ChEBI | CHEBI:30530 | ||
| ChemSpider | 74203 | ||
| |||
| Jmol-3D images | Image | ||
| PubChem | 82223 | ||
| |||
| Properties | |||
| AsF5 | |||
| Molar mass | 169.9136 g mol−1 | ||
| Appearance | colorless gas | ||
| Density | 2.138 g cm−3[1] | ||
| Melting point | -79.8 ˚C[1] | ||
| Boiling point | -52.8 ˚C[1] | ||
| Solubility | Ethanol, Dimethylether, Benzene | ||
| Hazards | |||
| EU classification | Toxic (T) Dangerous for the environment (N) | ||
| R-phrases | R23/25, R50/53 | ||
| S-phrases | (S1/2), S20/21, S28, S45, S60, S61 | ||
| NFPA 704 | |||
| Related compounds | |||
| Related group 5 fluorides |
Phosphorus pentafluoride Antimony pentafluoride Bismuth pentafluoride | ||
| Related compounds |
Arsenic pentachloride Arsenic trifluoride Arsenic pentoxide | ||
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |||
| | |||
| Infobox references | |||
Arsenic pentafluoride is a chemical compound of arsenic and fluorine. The oxidation state of arsenic is +5.
Synthesis
Arsenic pentafluoride can be prepared by direct combination of arsenic and fluorine:[2]
- 2As + 5F2 → 2AsF5
It can also be prepared by the reaction of arsenic trifluoride and fluorine:
- AsF3 + F2 → AsF5
Also it can be made by the addition of elemental fluorine
- 2As2O5 + 10F2 → 4AsF5 + 5O2
- 2As2O3 + 10F2 → 4AsF5 + 3O2
Properties
Arsenic pentafluoride is a colourless gas and has a trigonal bipyramidal structure.[2] In the solid state the axial As-F bond lengths are 171.9 pm and the equatorial 166.8 pm.[2]
Reactions
Arsenic pentafluoride forms halide complexes and is a powerful acceptor as shown by the reaction with sulfur tetrafluoride forming an ionic complex.[3]
- AsF5 + SF4 → SF3+ + AsF6−
Safety
Arsenic pentafluoride is an extremely dangerous toxin, mainly poisoning liver cells. It has an apparent smell that is similar to vinyl chloride gas.
See also
References
- ↑ 1.0 1.1 1.2 Record of Arsenic(V) fluoride in the GESTIS Substance Database of the IFA, accessed on 24/12/2007
- ↑ 2.0 2.1 2.2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0080379419.
- ↑ An investigation of the structures of the adducts of SF4 with BF3, PF5, AsF5, and SbF5 in the solid state and in solution in HF, M. Azeem, M. Brownstein, and R. J. Gillespie Can. J. Chem. 47(22): 4159–4167 (1969), doi:10.1139/v69-689
| ||||||