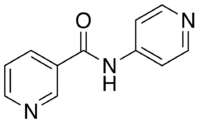
4-Pyridylnicotinamide
 | |
| Names | |
|---|---|
| IUPAC name
N-pyridin-4-ylpyridine-3-carboxamide | |
| Other names
4-pna 3-pyridinecarboxamide, N-4-pyridinyl- N-(Pyridin-4-yl)nicotinamide | |
| Identifiers | |
| 64479-79-4 | |
| ChemSpider | 2311305 |
| Jmol-3D images | Image |
| |
| Properties | |
| Molecular formula |
C11H9N3O |
| Molar mass | 199.21 g·mol−1 |
| Density | 1.287 g/cm3 |
| Boiling point | 286.08 °C (546.94 °F; 559.23 K) |
| Structure | |
| Dipole moment | 0 D |
| Hazards | |
| Flash point | 126.817 °C (260.271 °F; 399.967 K) |
| Related compounds | |
| Related compounds |
4,4'-bipyridine Pyridine Nicotinamide 4-Aminopyridine 3-Pyridylnicotinamide |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
4-Pyridylnicotinamide (4-pna), also known as N-(Pyridin-4-yl)nicotinamide, is a kinked dipodal dipyridine which was originally developed for use in chemotherapy.[1] As in its isomer 3-Pyridylnicotinamide, the nitrogen atoms on its pyridine rings can donate their electron lone pairs to metal cations, allowing it to bridge metal centers and act as a bidentate ligand in coordination polymers.[2][3][4][5] It is synthesized through the reaction of nicotinoyl chloride and 4-Aminopyridine.[1]
References
- ↑ 1.0 1.1 Gardner, T. S.; Wenis, E.; Lee, J. (1954). "The Synthesis of Compounds for the Chemotherapy of Tuberculosis. Iv. The Amide Function". The Journal of Organic Chemistry 19 (5): 753. doi:10.1021/jo01370a009.
- ↑ Kraft, P. E.; Laduca, R. L. (2012). "Catena-Poly\\tetra-μ-benzoato-κ8O:O′-dicopper(II)]-μ-\N-(pyridin-4-yl)nicotinamide]-κ2N:N′-\dibenzoato-κ2O-copper(II)]-μ-\N-(pyridin-4-yl)nicotinamide]-κ2N:N′]". Acta Crystallographica Section E Structure Reports Online 68 (8): m1049. doi:10.1107/S1600536812030437.
- ↑ Krishna Kumar, D. (2009). "Exploring the effect of chain length of bridging ligands in coordination complexes and polymers derived from mixed ligand systems of pyridylnicotinamides and dicarboxylates". Inorganica Chimica Acta 362 (6): 1767–2013. doi:10.1016/j.ica.2008.08.033.
- ↑ Kumar, D. K.; Das, A.; Dastidar, P. (2006). "One-Dimensional Chains, Two-Dimensional Corrugated Sheets Having a Cross-Linked Helix in Metal−Organic Frameworks: Exploring Hydrogen-Bond Capable Backbones and Ligating Topologies in Mixed Ligand Systems". Crystal Growth & Design 6 (8): 1903. doi:10.1021/cg0600344.
- ↑ Kumar, D. K.; Das, A.; Dastidar, P. (2006). "Exploring hydrogen-bond capable backbone and ligating topologies: Co(II) coordination polymers derived from mixed ligand systems". Journal of Molecular Structure 796: 139. doi:10.1016/j.molstruc.2006.02.033.