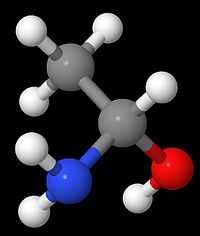
1-Aminoethanol
 | |
| Names | |
|---|---|
| Systematic IUPAC name
1-Aminoethanol | |
| Other names
Acetaldehyde ammonia, 1-Aminoethanol | |
| Identifiers | |
| 75-39-8 | |
| ChemSpider | 455852 |
| |
| Jmol-3D images | Image |
| |
| Properties | |
| Molecular formula |
C2H7NO |
| Molar mass | 61.08 g·mol−1 |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
1-Aminoethanol is the organic compound with the formula CH3CH(NH2)OH. It is classified as an alkanolamine. Specifically, it is a structural isomer of 2-aminoethanol (ethanolamine). These two compounds differ in the position of the amino group. Since the central carbon atom in 1-aminoethanol has four different substitutents, the compound has two stereoisomers. Unlike 2-aminoethanol, which is of considerable importance in commerce, 1-aminoethanol is not encountered as a pure material and is mainly of theoretical interest.
1-Aminoethanol exists in a solution of acetaldehyde and aqueous ammonia.[1]
References
- ↑ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 0-471-72091-7