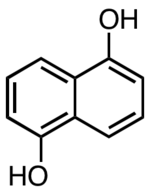
1,5-Dihydroxynaphthalene
 | |
| Names | |
|---|---|
| IUPAC name
Naphthalene-1,5-diol | |
| Other names
Azurol; 1,5-Naphthalenediol | |
| Identifiers | |
| 83-56-7 | |
| |
| Jmol-3D images | Image |
| PubChem | 6749 |
| |
| Properties | |
| Molecular formula |
C10H8O2 |
| Molar mass | 160.17 g·mol−1 |
| Appearance | Colorless solid |
| Melting point | 259–261 °C (498–502 °F; 532–534 K) |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
1,5-Dihydroxynaphthalene is an organic compound with the formula C10H6(OH)2. It is one of several isomers of dihydroxynaphthalene. It exists as colorless crystals that are soluble in polar organic solvents. It is a precursor to certain dyes.
Preparation and use
1,5-Dihydroxynaphthalene is prepared from naphthalene-1,5-disulfonic acid by hydrolysis with strong base followed by acidification.
It couples with various aryl diazonium salts to give diazo dyes. Oxidation with chromium trioxide gives juglone, a naturally occurring dye.[1]
References
- ↑ Gerald Booth "Naphthalene Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_009.