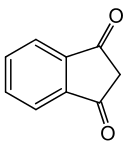
1,3-Indandione
 | |
 | |
| Names | |
|---|---|
| IUPAC name
indane-1,3-dione | |
| Other names
Indandione; 1,3-Diketohydrindene; 1,3-Dioxoindane; 1,3-Hydrindendione | |
| Identifiers | |
| 606-23-5 | |
| ChEMBL | ChEMBL283521 |
| ChemSpider | 11322 |
| |
| Jmol-3D images | Image Image |
| PubChem | 11815 |
| |
| Properties | |
| Molecular formula |
C9H6O2 |
| Molar mass | 146.14 g·mol−1 |
| Appearance | Yellow solid |
| Density | 1.37 g / cm3 |
| Melting point | 129 °C (264 °F; 402 K)[1][2] |
| slight | |
| Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| | |
| Infobox references | |
1,3-Indandione is an aromatic trans-fixed β-diketone. In standard conditions it is referred to in different sources as either a colorless or yellowish,[3] green,[4] or (most commonly) yellow solid.
Uses
In addition, 1,2-indandione ("indanedione") is used in the first stage of forensic identification of latent fingerprints. It is particularly useful for paper, and for items printed with thermal inks such as receipts. Amino acids left behind by the human hand may be developed into fingerprints by the use of it; the results, photographed with a special filter under a strong yellow-green fluorescent or green laser. It is usually the first method employed in a sequential analysis aimed at the production of evidence of a grade suitable for use in the courtroom. [5]
See also
References
- ↑ 1,3-Indandione at Sigma-Aldrich
- ↑ MSDS at Acros Organics, retrieved on June 16, 2011
- ↑ (Russian) Нейланд О. Я. Органическая химия: Учеб. для хим. спец. вузов. Москва: Высшая школа, 1990.— с. 481—490.
- ↑ Datapage, AlfaAesar, June 16, 2011
- ↑ Sequential Processing 2010 : 01 : History of Indanedione. Sequential Processing of Documents For Fingerprints (NFSTC). Retrieved August 2, 2013.
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||