Zinc fluoride
From Wikipedia, the free encyclopedia
| Zinc fluoride | |
|---|---|
 | |
| Other names Zinc difluoride | |
| Identifiers | |
| CAS number | 7783-49-5 13986-18-0 (tetrahydrate) |
| PubChem | 24551 |
| ChemSpider | 22957 |
| RTECS number | ZH3200000 |
| Jmol-3D images | {{#if:F[Zn]F|Image 1 |
| |
| |
| Properties | |
| Molecular formula | ZnF2 |
| Molar mass | 103.406 g/mol (anhydrous) 175.45 g/mol (tetrahydrate) |
| Appearance | white needles hygroscopic |
| Density | 4.95 g/cm3 (anhydrous) 2.30 g/cm3 (tetrahydrate) |
| Melting point | 872 °C (anhydrous) 100 °C, decomp (tetrahydrate) |
| Boiling point | 1500 °C (anhydrous) |
| Solubility in water | .000052 g/100 mL (anhydrous) 1.52 g/100 mL, 20 °C (tetrahydrate) |
| Solubility | sparingly soluble in HCl, HNO3, ammonia |
| Structure | |
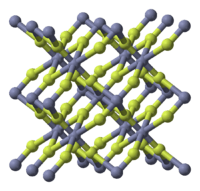
| Crystal structure | tetragonal (anhydrous), tP6 |
| Space group | P42/mnm, No. 136 |
| Hazards | |
| EU Index | Not listed |
| NFPA 704 |
 0
2
1
|
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
Zinc fluoride (ZnF2) is an inorganic chemical compound. It is encountered as the anydrous form and also as the tetrahydrate, ZnF2 · 4H2O (rhombohedral crystal structure).[1] It has a high melting point and has the rutile structure containing 6 coordinate zinc, which suggests appreciable ionic character in its chemical bonding.[2] Unlike the other zinc halides, ZnCl2, ZnBr2 and ZnI2, it is not very soluble in water.[2]
Preparation and reactions
Zinc fluoride can be synthesized several ways.
- Reaction of a fluoride salt with zinc chloride, to yield zinc fluoride and a chloride salt, in aqueous solution.[citation needed]
- The reaction of zinc metal with fluorine gas.[2]
- Reaction of hydrofluoric acid with zinc, to yield hydrogen gas (H2) and zinc fluoride (ZnF2).[2]
Zinc fluoride can be hydrolysed by hot water to form the zinc hydroxyfluoride, Zn(OH)F.[3]
References
- ↑ Perry, D. L.; Phillips, S. L. (1995). Handbook of Inorganic Compounds. CRC Press. ISBN 0-8493-8671-3.
- ↑ 2.0 2.1 2.2 2.3 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0080379419.
- ↑ Srivastava, O. K.; Secco, E. A. (1967). "Studies on Metal Hydroxy Compounds. I. Thermal Analyses of Zinc Derivatives ε-Zn(OH)2, Zn5(OH)8Cl2 · H2O, β-ZnOHCl, and ZnOHF" (pdf). Canadian Journal of Chemistry 45 (6): 579−583. doi:10.1139/v67-096.
External links
| ||||||||||||
This article is issued from Wikipedia. The text is available under the Creative Commons Attribution/Share Alike; additional terms may apply for the media files.