Xylenol
Xylenol are organic compounds with the formula (CH3)2C6H3OH. They are volatile colorless solids or oily liquids. They are derivatives of phenol with two methyl groups and a hydroxyl group. Six isomers exist, of which 2,6-xylenol with both methyl group in an ortho position with respect to the hydroxyl group is the most important. The name xylenol is a portmanteau of the words xylene and phenol.
Production
Together with many other compounds, xylenols are traditionally extracted from coal tar, the volatile materials obtained in the production of coke from coal. These residue contains a few percent by weight of xylenols as well as cresols and phenol. The main xylenols in such tar are the 3,5-, 2,4, and 2,3- isomers. 2,6-Xylenol is produced by methylation of phenol using methanol in the presence of metal oxide catalysts:[1]
- C6H5OH + 2 CH3OH → (CH3)2C6H3OH + H2O
Properties
The physical properties of the six isomeric xylenols are similar.
| Isomer | 2,6-Xylenol | 2,5-Xylenol | 2,4-Xylenol | 2,3-Xylenol | 3,4-Xylenol | 3,5-Xylenol |
| Structure |  |
 |
 |
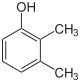 |
 |
 |
| CAS | 576-26-1 | 95-87-4 | 105-67-9 | 526-75-0 | 95-65-8 | 108-68-9 |
| Mp °C | 43-45 | 63 - 65 | 22-23 | 70-73 | 62 - 68 | 61-64 |
| Bp °C | 203 | 212 | 211-212 | 217 | 227 | 222 |
| pKa [2] | 10.59 | 10.22 | 10.45 | 10.50 | 10.32 | 10.15 |
| Density g/mL | 0.971 | 1.011 | ||||
Uses
Together with cresols and cresylic acid, xylenols are an important class of phenolics with great industrial importance. They are used in the manufacture of antioxidants. Xylenol orange is a redox indicator built on a xylenol skeleton. 2,6-Xylenol is a monomer for poly(p-phenylene oxide) (PEO) engineering resins through carbon-oxygen oxidative coupling.
References
- ↑ Helmut Fiegein "Cresols and Xylenols" in Ullmann's Encyclopedia of Industrial Chemistry, 2007; Wiley-VCH, Weinheim. doi:10.1002/14356007.a08_025
- ↑ CRC Handbook of Tables for Organic Compound Identification, Third Edition, 1984, ISBN 0-8493-0303-6.
See also
External links
- Datasheet 2,6 isomer
- Datasheet 2,5 isomer
- Datasheet 2,4 isomer
- Datasheet 2,3 isomer
- Datasheet 3,4 isomer
- Datasheet 3,5 isomer