Trimethylsilane
From Wikipedia, the free encyclopedia
| Trimethylsilane | |
|---|---|
 |
 |
| Identifiers | |
| CAS number | 993-07-7 |
| ChemSpider | 63614 |
| Jmol-3D images | {{#if:C[SiH](C)C|Image 1 |
| |
| Properties | |
| Molecular formula | C3H10Si |
| Molar mass | 74.20 g mol−1 |
| Density | 0.638 g cm-3 |
| Melting point | −135.9 °C; −212.6 °F; 137.2 K |
| Boiling point | 6.7 °C; 44.1 °F; 279.8 K |
| Hazards | |
| EU classification | |
| R-phrases | R12, R36/37/38 |
| S-phrases | S9, S16, S26, S33 |
| NFPA 704 |
 4
2
1
|
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
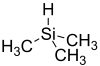
Trimethylsilane or trimethylsilyl hydride, is a compound with the formula C3H10Si, or with the structural formula (CH3)3SiH. It is very flammable. Trimethylsilane is used in the semi-conductor industry as an etchant in the plasma phase.[1]
See also
- Dimethylsilane
- Trimethylsilyl functional group
References
- ↑ Chen, Sheng-Wen; Wang, Yu-Sheng; Hu, Shao-Yu; Lee, Wen-Hsi; Chi, Chieh-Cheng; Wang, Ying-Lang (2012). "A Study of Trimethylsilane (3MS) and Tetramethylsilane (4MS) Based α-SiCN:H/α-SiCO:H Diffusion Barrier Films". Materials 5 (3): 377. doi:10.3390/ma5030377.
This article is issued from Wikipedia. The text is available under the Creative Commons Attribution/Share Alike; additional terms may apply for the media files.