Tert-Butyl alcohol
| tert-Butyl alcohol | |
|---|---|
 |
 |
 | |
| IUPAC name 2-Methylpropan-2-ol[1] | |
| Identifiers | |
| CAS number | 75-65-0 |
| PubChem | 6386 |
| ChemSpider | 6146 |
| UNII | MD83SFE959 |
| EC number | 200-889-7 |
| UN number | 1120 |
| DrugBank | DB03900 |
| MeSH | tert-Butyl+Alcohol |
| ChEBI | CHEBI:45895 |
| ChEMBL | CHEMBL16502 |
| RTECS number | EO1925000 |
| Beilstein Reference | 906698 |
| Gmelin Reference | 1833 |
| Jmol-3D images | Image 1 |
| |
| |
| Properties | |
| Molecular formula | C4H10O |
| Molar mass | 74.12 g mol−1 |
| Appearance | Colorless liquid |
| Odor | Camphorous |
| Density | 0.775 g/mL |
| Melting point | 25 to 26 °C; 77 to 79 °F; 298 to 299 K |
| Boiling point | 82 to 83 °C; 179 to 181 °F; 355 to 356 K |
| Solubility in water | miscible[2] |
| log P | 0.584 |
| Vapor pressure | 4.1 kPa (at 20 °C) |
| Acidity (pKa) | 17 [3] |
| Refractive index (nD) | 1.387 |
| Thermochemistry | |
| Std enthalpy of formation ΔfH |
−360.04–−358.36 kJ mol−1 |
| Std enthalpy of combustion ΔcH |
−2.64479–−2.64321 MJ mol−1 |
| Standard molar entropy S |
189.5 J K−1 mol−1 |
| Specific heat capacity, C | 215.37 J K−1 mol−1 |
| Hazards | |
| MSDS | inchem.org |
| GHS pictograms |   |
| GHS signal word | DANGER |
| GHS hazard statements | H225, H319, H332, H335 |
| GHS precautionary statements | P210, P261, P305+351+338 |
| EU Index | 603-005-00-1 |
| EU classification | |
| R-phrases | R11, R20, R36/37 |
| S-phrases | (S2), S9, S16, S46 |
| NFPA 704 |
 3
1
0
|
| Flash point | 11 °C; 52 °F; 284 K |
| Autoignition temperature | 480 °C; 896 °F; 753 K |
| Explosive limits | 2.4–8.0% |
| Related compounds | |
| Related butanols | 2-Butanol n-Butanol |
| Related compounds | 2-Methyl-2-butanol Trimethylsilanol |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
tert-Butyl alcohol (TBA), or 2-methyl-2-propanol (2M2P), is the simplest tertiary alcohol. It is one of the four isomers of butanol. tert-Butanol is a clear liquid (or a colorless solid, depending on the ambient temperature) with a camphor-like odor. It is very soluble in water and miscible with ethanol and diethyl ether. It is unique among the isomers of butanol because it tends to be a solid at room temperature, with a melting point slightly above 25 °C.
Natural occurrence
t-Butyl alcohol has been identified in beer and chickpeas.[4] It is also found in cassava[5] which is used as fermentation ingredient in certain alcoholic beverages.
Preparation
tert-Butyl alcohol is derived commercially from isobutane as a co-product of propylene oxide production. It can also be produced by the catalytic hydration of isobutylene, or by a Grignard reaction between acetone and methylmagnesium chloride.
Applications
tert-Butyl alcohol is used as a solvent, ethanol denaturant, paint remover ingredient, and gasoline octane booster and oxygenate. It is a chemical intermediate used to produce MTBE and ETBE by reaction with methanol and ethanol, and TBHP by reaction with hydrogen peroxide.
Chemistry
As a tertiary alcohol, tert-butyl alcohol is more stable to oxidation and less reactive than the other isomers of butanol.
When tert-butyl alcohol is deprotonated with a strong base, the product is an alkoxide anion. In this case, it is tert-butoxide. For example, the commonly used organic reagent potassium tert-butoxide is prepared by refluxing dry tert-butanol with potassium metal.[6]
- K + tBuOH → tBuO−K+ + 1/2 H2
The tert-butoxide species is itself useful as a strong, non-nucleophilic base in organic chemistry. It is able to abstract acidic protons from the substrate molecule readily, but its steric bulk inhibits the group from participating in nucleophilic substitution, such as in a Williamson ether synthesis or an SN2 reaction.
Conversion to alkyl halide
tert-Butyl alcohol reacts with hydrogen chloride to form tert-butyl chloride and water via an SN1 mechanism.
-

Step 1
-
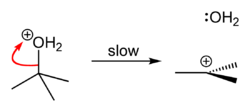
Step 2
-

Step 3
The overall reaction, therefore, is:

Because tert-butyl alcohol is a tertiary alcohol, the relative stability of the tert-butyl carbocation in the Step 2 allows the SN1 mechanism to be followed. Primary alcohols generally undergo an SN2 mechanism because the relative stability of a primary carbocation intermediate is very low. The tertiary carbocation in this case is stabilized through hyperconjugation where the neighboring C–H sigma bonds donate electrons into the empty p-orbital of the carbocation.
References
- ↑ 1.0 1.1 1.2 "tert-Butyl Alcohol - Compound Summary". PubChem Compound. National Center for Biotechnology Information. 2005-03-26. Retrieved 2012-05-19.
- ↑ http://www.inchem.org/documents/icsc/icsc/eics0114.htm
- ↑ Reeve, W.; Erikson, C.M.; Aluotto, P.F. Can. J. Chem, 1979, 57, 2747.
- ↑ http://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+50
- ↑ http://www.sc.mahidol.ac.th/scbc/bc_internet/publication/696.pdf
- ↑ Johnson, W. S.; Schneider, W. P. (1950), "β-Carbethoxy-γ,γ-diphenylvinylacetic acid", Org. Synth. 30: 18; Coll. Vol. 4: 132
External links
- International Chemical Safety Card 0114
- NIOSH Pocket Guide to Chemical Hazards 0078
- IPCS Environmental Health Criteria 65: Butanols: four isomers
- IPCS Health and Safety Guide 7: tert-Butanol
| |||||||||||||||||