Simarouba amara
| Simarouba amara | |
|---|---|
 | |
| The canopy of Simarouba amara | |
| Scientific classification | |
| Kingdom: | Plantae |
| (unranked): | Angiosperms |
| (unranked): | Eudicots |
| (unranked): | Rosids |
| Order: | Sapindales |
| Family: | Simaroubaceae |
| Genus: | Simarouba |
| Species: | S. amara |
| Binomial name | |
| Simarouba amara Aubl 1775[2] | |
| Subspecies | |
| Synonyms | |
|
Quassia simarouba[4] | |
Simarouba amara is a species of tree in the Simaroubaceae family, found in the rainforests and savannahs of South and Central America and the Caribbean. It was first described by Aublet in French Guiana in 1775 and is one of six species of Simarouba. The tree is evergreen, but produces a new set of leaves once a year. It requires relatively high levels of light to grow and grows rapidly in these conditions, but lives for a relatively short time. In Panama, it flowers during the dry season in February and March, whereas in Costa Rica, where there is no dry season it flowers later, between March and July. As the species is dioecious, the trees are either male or female and only produce male or female flowers. The small yellow flowers are thought to be pollinated by insects, the resulting fruits are dispersed by animals including monkeys, birds and fruit-eating bats and the seeds are also dispersed by leaf cutter ants.
Simarouba amara has been studied extensively by scientists in an attempt to understand the tree and also to gain a better understanding of the ecology of the rainforest in general. Many of these studies were conducted on Barro Colorado Island in Panama or at La Selva Biological Station in Costa Rica. Of particular interest is how it competes with other species and with individuals of the same species at different stages in its life cycle. The seedlings are normally limited by the amount of light and nutrients found where they are growing and the saplings are considered relatively light demanding compared to other species. Young individuals are more likely to survive when they grow further away from their parents and when there are few other individuals growing near to them, which may be due to them being able to escape diseases. Plant physiologists have investigated how the leaves of the tree differ depending on their location in the forest canopy finding they are thicker in the canopy and thinner in the understory. They have also measured how the water potential of their leaves changes and when their stomata open and close during the day; the findings suggest that rather than closing their stomata to control water loss, it is controlled by the leaf area instead. Population geneticists have examined the way in which its genes vary, at both the local scale and across its range using microsatellites. It is genetically diverse, indicating gene flow occurs between populations and seeds can be dispersed up to 1 km. The leaves of S. amara are eaten by several species of caterpillar, particularly those in the Atteva genus. Several species of termite and ants live on or around the tree and lianas and epiphytes grow on the tree.
The bark of S. amara has been used by people in its range to treat dysentery and diarrhea, as well as other diseases, and was also exported to Europe in the eighteenth century to treat these illnesses. A number of compounds have since been isolated from the bark and have been shown to have antimicrobial effects. Local people use the wood of the tree for various purposes and it is also grown in plantations and harvested for its timber, some of which is exported.
Description
Simarouba amara grows to heights of up to 35 metres, with a maximum trunk diameter of 125 cm and a maximum estimated age of 121 years.[6][7][8] It has compound leaves that are each around 60 cm long, the petioles are 4–7 cm long and each leaf has 9–16 leaflets. Each leaflet is 2.5–11 cm long and 12–45 mm wide, with those towards the end of the compound leaf tending to be smaller. The flowers occur on a staminate panicle that is around 30 cm in length, which is widely branched and densely covered in flowers.[9] The flowers are unisexual, small (<1 cm long) and pale yellow in colour. They are thought to be pollinated by insects such as small bees and moths. On Barro Colorado Island (BCI), Panama, it tends to flower during the dry season from the end of January to the end of April, persisting for 11 to 15 weeks each year.[6] In Costa Rica, it flowers slightly later, between March and July, peaking in April.[10] Fruits form between 1 and 3 months after pollination occurs. The fruits are brightly colored green to purplish-black, approximately 17 mm long and contain large seeds (10–14 mm), they occur in groups of 3–5 drupes. The seeds cannot stay dormant and are dispersed by vertebrates.[6] Each seed weighs approximately 0.25 g.[7]
It is an evergreen species, with a new flush of leaves growing between January and April, during the dry season, when the highest light levels occur in the rainforest. This phenology is thought to allow S. amara to photosynthesise most effectively, since the new leaves are more efficient than those they replace.[8][10] It has visible, but indistinct growth rings that are on average 7 mm wide.[8] A study of individuals in Panama found that they grow on average 8.4 mm in diameter each year,[11] in Costa Rica, growth rates as fast as 18 mm per year have been recorded, and the stem grows constantly throughout the year.[10] The xylem vessels in mature trees range from 20 to 90 μm in diameter, with around 50 vessels present per mm2 of branch.[12] The density of the wood is between 0.37–0.44 g/cm3,[7] lower than many other species in the rainforest.[13]
It is a fast-growing, light-demanding and shade-intolerant species. Saplings are typically one straight pole, with several compound leaves and only one point of growth. This allows the sapling to achieve the greatest vertical growth with a minimum amount of biomass.[14] They start to branch once they are 2–5 m tall.[15]
A study in the forest dynamics plot on BCI found that between 1982 and 2000, around 65% of individuals died, with mortality highest amongst small individuals (<1 cm dbh). Large trees (>20 cm dbh) are relatively rare, averaging 2.4 trees per hectare, compared to 40 trees per hectare of >1 cm dbh.[6]
Taxonomy
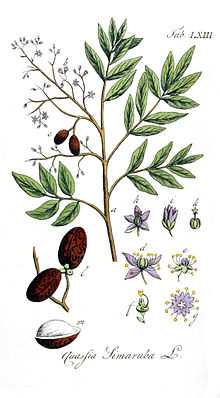
Simarouba amara was first described by Jean Baptiste Christophore Fusée Aublet in French Guiana in 1775 and is the type species of the Simarouba genus.[2][3] In 1790, William Wright described Quassia simarouba,[16] which Auguste Pyrame DeCandolle suggested was the same species as S. amara. However, because S. amara was described as monoecious by Aublet and Q. simarouba was described as dioecious by Wright, they were still regarded as separate species in 1829.[17] By 1874, when the Flora Brasiliensis was published, they were considered synonymous.[18]
Among the six species of Simarouba, two besides S. amara occur on the continent: S. glauca and S. versicolor. S. amara can be distinguished from the other continental species by having smaller flowers, anthers and fruit, and straight, rather than curved petals.[1] The leaves of Simarouba amara subsp. opaca are not glaucous (a bluish-grey or green colour) on their underside, whereas those of Simarouba amara subsp. typica are.[3]
| Structure | S. amara | S. glauca | S. versicolor |
|---|---|---|---|
| Flower | 3–5 mm long | 4–7.5 mm long | 4–7.5 mm long |
| Anthers | 0.4–1.2 mm long | 1.3–2.0 mm long | 1.0–1.5 mm long |
| Petals | Straight, dull yellow-green to white | Curved, brighter yellow with a touch of orange or red | Curved |
| Fruits | 1.0–1.5 x 0.6–1.0 cm | 2.0–2.5 x 1.2–1.5 cm | 2.0–2.5 x 1.5–2.0 cm |
Common names
Simarouba amara is known by many common names, where in the Neotropics. In Bolivia it is known as chiriuana, in Brazil as marupa, marupuaba, parahyba, paraiba and tamanqueira. In Colombia it is called simaruba, in Ecuador as cedro amargo, cuna and guitarro, in French Guiana as simarouba, in Guyana as simarupa, in Peru as marupa, in Surinam as soemaroeba and in Venezuela cedro blanco and simarouba.[19]
In Europe, it was known by various names during the nineteenth century when it was used as a medicine; these names included bitter ash,[20] bitterwood,[19] mountain damson[21] and stave-wood.[22]
Distribution
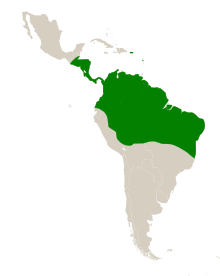
The natural range of S. amara is in the Neotropics, the ecoregion of Central and South America. Its range extends from Guatemala in the north, to Bolivia in the south and from Ecuador in the west, to the east coast of Brazil.[1] It has been introduced to the islands of Dominica and Puerto Rico in the Caribbean Sea, becoming naturalised in Puerto Rico.[23][24] On BCI, mature trees (>10 cm dbh) are found at a frequency of 5 per hectare, in Ecuador at 0.7 per hectare and in French Guiana at 0.4 per hectare. Genetic analysis of populations suggests that it has always been relatively common within its range.[25] It grows in rainforests and in savannahs.[26] The seedlings of S. amara are rare in primary forest due to their light-demanding habit.[14]
Genetics
Populations of S. amara display high levels of heterozygosity indicating that it is genetically diverse. This is consistent with the tree outcrossing over large distances by long-distance pollen flow and that there has been sufficient long distance gene flow between populations to counteract the effects of genetic drift. A study of 478 plants from 14 populations across South America found that 24% of all alleles occurred in only one population.[25] A study of 300 plants on Barro Colorado Island found that the heterozygosity at 5 microsatellite loci varied between 0.12 and 0.75. 8 out of the 50 alleles scored occurred in only one plant.[6]
Reproduction
Individuals do not typically reproduce until they have a trunk diameter of 30 cm. Once mature, the trees produce flowers each year, but not all females produce fruit each year.[6] Their flower morphology is typical of being pollinated by generalist small insects such as bees and moths.[6] It has been reported to be pollinated by non-sphingid moths,[27] but other authors have questioned whether this is correct.[28]
Seed dispersal

The seeds of S. amara are dispersed by vertebrates, mainly large birds and mammals, including chachalacas, flycatchers, motmots, thrushes, howler monkeys, tamarins[29] and spider monkeys. Leaf cutter ants have also been observed to disperse the seeds and dense seedling carpets form in areas where they dump waste material.[6] Seeds that are eaten by monkeys are more likely to germinate than seeds that have not.[30] Fruit-eating phyllostomid bats have also been noted to disperse their seeds; this may aid the regeneration of forests as they disperse the seeds of later successional species while they feed on S. amara.[31]
Based on inverse modelling of data from seed traps on BCI, the estimated average dispersal distance for seeds is 39 m.[6] Studying seedlings and parent trees on BCI using DNA microsatellites revealed that, in fact on average, seedlings grow 392 m away from their parents, with a standard deviation of ±234 m and a range of between 9 m and 1 km. In the forest there are many seeds and seedlings beneath reproductive females; genetic data indicate that seedlings are unlikely to be from nearby adults, but rather dispersed there by vertebrates that have fed on one tree and then moved to feed on another, defecating while in the canopy and depositing the seeds.[29]
Physiology
Various aspects of the physiology or S. amara have been studied. The stomatal conductance of the leaves, an indication of the rate at which water evaporates, of mature trees at midday range from 200 to 270 mmol H2O m−2 s−1. The leaf water potentials at midday range from −0.56 to −1.85 MPa, averaging around −1.2 MPa. Cavitation is widespread in the trunk and the stomata do not close before cavitation occurs. Although this would normally be considered deleterious to the tree, it may buffer the leaf water potential and therefore be beneficial. The stomatal conductance and hydraulic conductance of the branches of taller trees (~30 m) are much higher than in the branches of smaller trees (~20 m). Phillips, Bond and Ryan suggested that this is probably due to the branches of taller trees having a lower leaf-to-sapwood ratio than those of small branches. Dye staining shows that cavitation is common in the branches of S. amara. They concluded that water flux in S. amara is controlled by structural (leaf area), rather than physiological (closing stomata) means.[12]
Leaves absorb light in the photosynthetically active radiation (PAR) spectrum at wavelengths between 400 nm and 700 nm with a high efficiency, but the efficiency decreases at longer wavelengths. Generally plants absorb PAR at efficiencies of around 85%; the higher values found in S. amara are thought to be due to the high humidity of its habitat. The reflectance and transmittance of the leaves are low at between 400 and 700 nm. The optical properties and the mass of the leaves vary depending on their location in the forest canopy, with leaves becoming thicker and more efficient as their height within the canopy increases. For their weight, however, leaves in the understory are more efficient at capturing light than leaves in the canopy.[32]
| Absorbance (400-700 nm) | Absorbance (700-750 nm) | Reflectance (400-700 nm) | Transmittance (400-700 nm) | Leaf mass (g/m2) | |
|---|---|---|---|---|---|
| Understory (~2m) | 91.7% | 37.3% | 6.3% | 2.1% | 36.9 |
| Mid-canopy (~10m) | 92.8% | 41.6% | 6.1% | 1.2% | 55.4 |
| Canopy (>20m) | 93.1% | 46.1% | 5.2% | 0.5% | 135.2 |
The concentration of bioavailable phosphate has been found to be higher underneath female individuals than underneath males, even though the total concentration of phosphate is equal. Rhoades et al. concluded that this difference was due to females changing the availability of phosphate, rather than females only growing in areas with high phosphate availability. This is thought to be caused either by the fruit containing high levels of phosphate which would fall off the tree and rot, or by the fruits attracting animals which deposit phosphate beneath the females. It is also possible that the sexes produce different root exudates, which affect the microbial community in their rhizosphere, thereby affecting phosphate availability.[33]
The woody tissues of S. amara have been found to respire at a rate of 1.24 μmol CO2 m−2 s−1, and this rate of respiration correlates positively with the growth rate of the stem. Maintenance respiration was calculated at 31.1 μmol CO2 m−3 s−1 and this rate correlated positively with the sapwood volume.[34]
Seedling physiology
| Nutrient | Concentration (mg/g leaf)
(dry weight) |
|---|---|
| Nitrogen | 20 |
| Phosphorus | 1 |
| Potassium | 16 |
| Calcium | 2 |
| Magnesium | 5 |
Experiments on BCI where trenches were dug around seedlings of S. amara, or where gaps in the canopy were made above them, show that their relative growth rate can be increased by both. This shows that their growth is normally limited by both above-ground competition for light and by below-ground competition for nutrients and water. Competition for light is normally more important, as shown by the growth rate increasing by almost 7 times and mortality decreasing, when seedlings were placed in gaps, compared to the understory. When seedlings in gaps had a trench dug round them to prevent below-ground competition their growth increased further, by 50%, demonstrating that in gaps the seedlings are limited by below-ground competition. Trenching around seedlings in the understory did not significantly increase their growth, showing that they are normally only limited by competition for light.[35]
Larger seedlings are more likely to survive the dry season on BCI than smaller seedlings.[36] Density-dependent inhibition occurs between seedlings: they are more likely to survive in areas where fewer seedlings of S. amara are growing. A study on individuals on BCI found that this pattern may be caused by differences in soil biota rather than by insect herbivores or fungal pathogens.[36] Observations based on the distance of seedlings from their parents indicate that the Janzen-Connell hypothesis applies to seedlings of S. amara: they are more likely to survive away from their parents as they escape pests such as herbivores and plant pathogens which are more common underneath the parent trees.[29]
Sapling physiology
Saplings of S. amara are light demanding and are found in brighter areas of the rainforest compared to Pitheullobium elegans and Lecythis ampla seedlings. A study at the La Selva Biological Station found the leaves weigh approximately 30 g/m2 (dry weight), similar to P. elegans, but around double the weight of L. ampla. The photosynthetic capacity of the leaves of S. amara is higher than that of the other two species, averaging around 6 μmol m−1 s−1. Dark respiration is on average 0.72 μmol m−1 s−1, higher than that of the other two species. The maximum photosynthetic rate correlates with both stem diameter and vertical growth. Diffuse light is thought to be more important for seedling growth than sunflecks.[14] Another study of saplings at La Selva found that they grew 7 cm yr−1 in height and 0.25 mm yr−1 in diameter. On average they had nine compound leaves, a leaf area index of 0.54 and the total surface area of their leaves was 124 cm2. The saplings that had the lowest leaf area were most likely to die during the study and those with a larger leaf area grew faster than other saplings.[15]
A study of saplings between one and four centimeters in diameter on BCI found that the growth of saplings did not vary depending on which species grew near them, contrary to predictions that density-dependence inhibition occurs. A model based on these findings predicted that saplings with a diameter of 2 cm are able to grow at a maximum rate of 13 mm yr−1 and that if another tree with a diameter of 10 cm is growing within 5 m of the sapling, its growth is only reduced to 12 mm yr−1, indicating that they are not affected by crowding. Trees growing more than 15 m away from a sapling do not affect their growth.[37]
Ecology

Lianas are relatively rare on mature (>20 cm dbh) individuals of S. amara, compared to other trees on BCI, with only around 25% having lianas growing on them. Putz suggested that this may be due to the trees having large leaves, but the mechanism by which this would reduce the number of lianas is unknown.[11] Smaller individuals also have fewer lianas and woody hemi-epiphytes than other species of tree in the same forests.[38]
The alianthus webworm (Atteva aurea) and other members of the Atteva genus have been recorded to eat the new shoot tips of S. amara in Costa Rica.[39] The larvae of the butterfly species, Bungalotis diophorus feed exclusively on saplings and treelets of S. amara.[40] Two termite species have been observed living on S. amara in Panama, Calcaritermes brevicollis in dead wood and Microcerotermes arboreus nesting in a gallery on a branch.[41] Bullet ants (Paraponera clavata) have been found to nest at the base of S. amara trees.[42] The Hemiptera, Enchophora sanguinea (Fulgoridae) has been found preferentially on the trunks of S. amara.[43][44]
Uses
Materials
Simarouba amara is used locally for producing paper, furniture, plywood and matches and is also used in construction.[6] It is also grown in plantations, as its bright and lightweight timber is highly sought after in European markets for use in making fine furniture and veneers.[7][45] The wood dries rapidly and is easy to work with normal tools. It is creamy white to light yellow in colour, with a coarse texture and a straight grain. It has to be treated to prevent fungi, wood borers and termites from eating it. The heartwood has a density of 0.35–0.45 g/cm3.[19] It has been noted to be one of the best species for timber that can be grown in the Peruvian Amazon, along with Cedrelinga catenaeformis, due to its rapid growth characteristics.[46] The Worldwide Fund for Nature recommend that consumers ensure S. amara timber is certified by the Forest Stewardship Council so that they do not contribute to deforestation.[47] Wood shavings of S. amara have been used in animal bedding leading to the poisoning of horses and dogs.[48]
Medical
The leaves and bark of S. amara have been used as an herbal medicine to treat dysentery, diarrhea, malaria and other illnesses in areas where it grows. In 1713, it was exported to France where it was used to treat dysentery, being an effective treatment during epidemics between 1718 and 1725. In 1918 its effectiveness was validated by a study where soldiers in a military hospital were given a tea made of the bark to treat amoebic dysentery.[medical citation needed] In a 1944 study, the Merck Institute found it was 92% effective at treating intestinal amoebiasis in humans. During the 1990s, scientists demonstrated it could kill the most common cause of dysentery, Entamoeba histolytica, and species of Salmonella and Shigella bacteria that cause diarrhea.[49]
The main biologically active compounds found in S. amara are the quassinoids, a group of triterpenes, of which the most therapeutic are ailanthinone, glaucarubinone, and holacanthone.[citation needed] These have been reported to kill protozoa, amoeba, Plasmodium (the cause of malaria) and also cancerous human cells. The anti-malarial properties were first investigated by scientists in 1947; they found that in chickens, 1 mg of bark extract per 1 kg of body weight had strong antimalarial activity.[medical citation needed] In 1997 a patent was filed in the United States for using an extract in a skin care product.[49]
References
- ↑ 1.0 1.1 1.2 Franceschinelli, E. V.; Yamamoto, K.; Shepherd, G. J. (1998). "Distinctions among Three Simarouba Species". Systematic Botany 23 (4): 479–488. doi:10.2307/2419379. JSTOR 2419379.
- ↑ 2.0 2.1 Christophore Fusée Aublet, Jean Baptiste (1775). Histoire des plantes de la Guiane Françoise. London and Paris: Pierre-François Didot. pp. 860–861.
- ↑ 3.0 3.1 3.2 3.3 Cronquist, A. (1944). "Studies in the Simaroubaceae-II. The Genus Simarouba". Bulletin of the Torrey Botanical Club 71 (3): 226–234. doi:10.2307/2481702. JSTOR 2481702.
- ↑ 4.0 4.1 4.2 Missouri Botanical Garden. "Synonyms of Simarouba amara". tropicos.org. Retrieved 2011-01-12.
- ↑ Herbert Stone (1904). The timbers of Commerce and their identification. W. Rider. Retrieved 13 January 2011.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 6.9 Hardesty, B.; Dick, C.; Kremer, A.; Hubbell, S.; Bermingham, E. (2005). "Spatial genetic structure of Simarouba amara Aubl. (Simaroubaceae), a dioecious, animal-dispersed Neotropical tree, on Barro Colorado Island, Panama". Heredity 95 (4): 290–297. doi:10.1038/sj.hdy.6800714. PMID 16094303.
- ↑ 7.0 7.1 7.2 7.3 Schulze, M.; Grogan, J.; Landis, R.; Vidal, E. (2008). "How rare is too rare to harvest?Management challenges posed by timber species occurring at low densities in the Brazilian Amazon". Forest Ecology and Management 256 (7): 1443. doi:10.1016/j.foreco.2008.02.051.
- ↑ 8.0 8.1 8.2 Fichtler, E.; Clark, D. A.; Worbes, M. (2003). "Age and Long-term Growth of Trees in an Old-growth Tropical Rain Forest, Based on Analyses of Tree Rings and 14C". Biotropica 35 (3): 306–317. doi:10.2307/30043047. JSTOR 30043047.
- ↑ Woodson, R. E.; Schery, R. W.; Porter, D. M. (1973). "Flora of Panama. Part VI. Family 90. Simaroubaceae". Annals of the Missouri Botanical Garden 60 (1): 23–39. doi:10.2307/2394767. JSTOR 2394767.
- ↑ 10.0 10.1 10.2 O'Brien, J. J.; Oberbauer, S. F.; Clark, D. B.; Clark, D. A. (2007). "Phenology and Stem Diameter Increment Seasonality in a Costa Rican Wet Tropical Forest". Biotropica 40 (2): 151. doi:10.1111/j.1744-7429.2007.00354.x.
- ↑ 11.0 11.1 Putz, F. E. (1984). "How Trees Avoid and Shed Lianas". Biotropica (Biotropica, Vol. 16, No. 1) 16 (1): 19–23. doi:10.2307/2387889. JSTOR 2387889.
- ↑ 12.0 12.1 Phillips, N.; Bond, B. J.; Ryan, M. G. (2001). "Gas exchange and hydraulic properties in the crowns of two tree species in a Panamanian moist forest". Trees 15 (2): 123–130. doi:10.1007/s004680000077.
- ↑ Santiago, L.; Goldstein, G.; Meinzer, F.; Fisher, J.; MacHado, K.; Woodruff, D.; Jones, T. (2004). "Leaf photosynthetic traits scale with hydraulic conductivity and wood density in Panamanian forest canopy trees". Oecologia 140 (4): 543–550. doi:10.1007/s00442-004-1624-1. PMID 15232729.
- ↑ 14.0 14.1 14.2 Oberbauer, S. F.; Clark, D. B.; Clark, D. A.; Rich, P. M.; Vega, G. (2009). "Light environment, gas exchange, and annual growth of saplings of three species of rain forest trees in Costa Rica". Journal of Tropical Ecology 9: 511. doi:10.1017/S0266467400007586.
- ↑ 15.0 15.1 Sterck, F. J.; Clark, D. B.; Clark, D. A.; Bongers, F. (1999). "Light Fluctuations, Crown Traits, and Response Delays for Tree Saplings in a Costa Rican Lowland Rain Forest". Journal of Tropical Ecology 15 (1): 83–95. doi:10.1017/S0266467499000681. JSTOR 2560198.
- ↑ "Quassia simarouba". Zipcodezoo.com. Retrieved 2011-01-10.
- ↑ Andrew Duncan (1829). Supplement to The Edinburgh new dispensatory. Printed for Bell & Bradfute. pp. 85–86.
- ↑ "Flora brasiliensis". CRIA. 2010-12-31. Retrieved 2011-01-04.
- ↑ 19.0 19.1 19.2 "Tropix 6.0 – Marupa". TROPIX. CIRAD. 2009-03-24. Retrieved 2011-01-12.
- ↑ Rivière, Peter (2006). The Guiana travels of Robert Schomburgk, 1835–1844, Volume 17. p. 34. ISBN 978-0-904180-88-6.
- ↑ Robert Eglesfeld Griffith (1847). Medical botany. Lea and Blanchard. pp. 198–. Retrieved 10 January 2011.
- ↑ A. H. R. Grisebach (1864). Flora of the British West Indian islands. Lovell Reeve and Co. p. 788. Retrieved 10 January 2011.
- ↑ Moses Kairo, Bibi Ali, Oliver Cheesman, Karen Haysom and Sean Murphy (2003). "Invasive Species Threats in the Caribbean Region" (PDF). CABI. p. 122. Retrieved 2011-01-10.
- ↑ "Invasive and introduced tree species on Dominica". Food and Agriculture Organisation of the United Nations. 2009-04-01. Retrieved 2011-01-14.
- ↑ 25.0 25.1 Hardesty, B. D.; Dick, C. W.; Hamrick, J. L.; Degen, B.; Hubbell, S. P.; Bermingham, E. (2010). "Geographic Influence on Genetic Structure in the Widespread Neotropical Tree Simarouba amara (Simaroubaceae)". Tropical Plant Biology 3: 28. doi:10.1007/s12042-010-9044-3.
- ↑ Sanaiotti, T. M.; Magnusson, W. E. (1995). "Effects of Annual Fires on the Production of Fleshy Fruits Eaten by Birds in a Brazilian Amazonian Savanna". Journal of Tropical Ecology 11 (1): 53–65. doi:10.1017/S0266467400008397. JSTOR 2560140.
- ↑ Renner, S. S.; Feil, J. P. (1993). "Pollinators of Tropical Dioecious Angiosperms". American Journal of Botany 80 (9): 1100–1107. doi:10.2307/2445757. JSTOR 2445757.
- ↑ Bawa, K. S. (1994). "Pollinators of Tropical Dioecious Angiosperms: A Reassessment? No, not yet". American Journal of Botany 81 (4): 456–460. doi:10.2307/2445495. JSTOR 2445495.
- ↑ 29.0 29.1 29.2 Hardesty, B. D.; Hubbell, S. P.; Bermingham, E. (2006). "Genetic evidence of frequent long-distance recruitment in a vertebrate-dispersed tree". Ecology Letters 9 (5): 516–525. doi:10.1111/j.1461-0248.2006.00897.x.
- ↑ Stevenson, P. R.; Castellanos, M. C.; Pizarro, J. C.; Garavito, M. (2002). International Journal of Primatology 23 (6): 1187. doi:10.1023/A:1021118618936.
- ↑ Kelm, D. .; Wiesner, K. .; Von Helversen, O. . (2008). "Effects of artificial roosts for frugivorous bats on seed dispersal in a neotropical forest pasture mosaic". Conservation biology : the journal of the Society for Conservation Biology 22 (3): 733–741. doi:10.1111/j.1523-1739.2008.00925.x. PMID 18445078.
- ↑ 32.0 32.1 Poorter, L.; Oberbauer, S. F.; Clark, D. B. (1995). "Leaf Optical Properties Along a Vertical Gradient in a Tropical Rain Forest Canopy in Costa Rica". American Journal of Botany 82 (10): 1257–1263. doi:10.2307/2446248. JSTOR 2446248.
- ↑ Rhoades, C. C.; Sanford, R. L.; Clark, D. B. (1994). "Gender Dependent Influences on Soil Phosphorus by the Dioecious Lowland Tropical Tree Simarouba amara". Biotropica 26 (4): 362–368. doi:10.2307/2389229.
- ↑ Ryan, M. G.; Hubbard, R. M.; Clark, D. A.; Sanford, R. L. (1994). "Woody-Tissue Respiration for Simarouba amara and Minquartia guianensis, Two Tropical Wet Forest Trees with Different Growth Habits". Oecologia 100 (3): 213–220. doi:10.1007/BF00316947.
- ↑ 35.0 35.1 Barberis, I. M.; Tanner, E. V. J. (2005). "Gaps and Root Trenching Increase Tree Seedling Growth in Panamanian Semi-Evergreen Forest". Ecology 86 (3): 667–674. doi:10.1890/04-0677. JSTOR 3450661.
- ↑ 36.0 36.1 Mangan, S.; Schnitzer, S.; Herre, E.; Mack, K.; Valencia, M.; Sanchez, E.; Bever, J. (2010). "Negative plant-soil feedback predicts tree-species relative abundance in a tropical forest". Nature 466 (7307): 752–755. Bibcode:2010Natur.466..752M. doi:10.1038/nature09273. PMID 20581819.
- ↑ Uriarte, M. A.; Condit, R.; Canham, C. D.; Hubbell, S. P. (2004). "A Spatially Explicit Model of Sapling Growth in a Tropical Forest: Does the Identity of Neighbours Matter?". Journal of Ecology 92 (2): 348–360. doi:10.1111/j.0022-0477.2004.00867.x. JSTOR 3599599.
- ↑ Clark, D. B.; Clark, D. A. (1990). "Distribution and Effects on Tree Growth of Lianas and Woody Hemiepiphytes in a Costa Rican Tropical Wet Forest". Journal of Tropical Ecology 6 (3): 321–331. doi:10.1017/S0266467400004570. JSTOR 2559832.
- ↑ Wilson, J.; Landry, J. F. O.; Janzen, D.; Hallwachs, W.; Nazari, V.; Hajibabaei, M.; Hebert, P. (2010). "Identity of the ailanthus webworm moth (Lepidoptera, Yponomeutidae), a complex of two species: evidence from DNA barcoding, morphology and ecology". ZooKeys 46. doi:10.3897/zookeys.46.406.
- ↑ Jeff, Miller; Daniel H. Janzen, Winifred Hallwachs (2009). One hundred butterflies and moths. Harvard University Press. ISBN 978-0-674-02334-5.
- ↑ Roisin, Y. .; Dejean, A. .; Corbara, B. .; Orivel, J. .; Samaniego, M. .; Leponce, M. . (2006). "Vertical stratification of the termite assemblage in a neotropical rainforest". Oecologia 149 (2): 301–311. doi:10.1007/s00442-006-0449-5. PMID 16791633.
- ↑ Belk, M. C.; Black, H. L.; Jorgensen, C. D.; Hubbell, S. P.; Foster, R. B. (1989). "Nest Tree Selectivity by the Tropical Ant, Paraponera clavata". Biotropica (Biotropica, Vol. 21, No. 2) 21 (2): 173–177. doi:10.2307/2388707. JSTOR 2388707.
- ↑ Johnson, L. K.; Foster, R. B. (1985). "Associations of Large Homoptera and Trees in a Tropical Forest". Journal of the Kansas Entomological Society 58 (3): 565. doi:10.2307/25084689. JSTOR 25084689.
- ↑ Naskrecki, P.; Nishida, K. (2007). "Novel trophobiotic interactions in lantern bugs (Insecta: Auchenorrhyncha: Fulgoridae)". Journal of Natural History 41 (37–40): 2397. doi:10.1080/00222930701633570.
- ↑ Granier, A.; Huc, R.; Colin, F. (1992). "Transpiration and stomatal conductance of two rain forest species growing in plantations (Simarouba amara and Goupia glabra) in French Guyana". Annales des Sciences Forestières 49 (1): 17. doi:10.1051/forest:19920102.
- ↑ McClain, Michael; Reynaldo L. Victoria, Jeffrey Edward Richey (2001). The biogeochemistry of the Amazon Basin. Oxford University Press. p. 111. ISBN 0-19-511431-0.
- ↑ "Photographic guide to identify your timber". Worldwide Fund for Nature (WWF). Retrieved 2011-01-04.
- ↑ Declercq, J. (2004). "Suspected wood poisoning caused by Simarouba amara (marupá/caixeta) shavings in two dogs with erosive stomatitis and dermatitis". Veterinary dermatology 15 (3): 188–193. doi:10.1111/j.1365-3164.2004.00377.x. PMID 15214956.
- ↑ 49.0 49.1 The Virtual Field Herbarium. "Simaroubaceae Simarouba amara Aublet". Oxford University Herbarium. Retrieved 2011-01-11.
External links
- Simarouba amara discoverlife.org
- Simarouba amara Missouri Botanical Garden
- Photographs of wood structure