Propellane
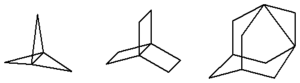
In organic chemistry, propellane is any member of a class of polycyclic hydrocarbons, whose carbon skeleton consists of three rings of carbon atoms sharing a common carbon–carbon covalent bond. The name derives from a supposed resemblance of the molecule to a propeller: namely, the rings would be the propeller's blades, and the shared C–C bond would be its axis.
Propellanes with small cycles are highly strained and unstable, and are easily turned into polymers with interesting structures, such as staffanes. Partly for these reasons, they have been the object of much research.
In the literature, the bond shared by the three cycles is usually called the "bridge"; the shared carbon atoms are then the "bridgeheads". The notation [x.y.z]propellane means the member of the family whose rings have x, y, and z carbons, not counting the two bridgeheads; or x+2, y+2, and z+2 carbons, counting them. The chemical formula is therefore C2+x+y+zH2(x+y+z). The minimum value for x, y, and z is 1, meaning a 3-carbon ring.
There is no structural ordering between the rings, so, for example, [1.3.2]propellane is the same substance as [3.2.1]propellane. Therefore it is customary to sort the indices in decreasing order, x ≥ y ≥ z.
General properties
Data can be found in Osmont et al., Energy and Fuels, Vol. 22, pp. 2241–2257 (2008).
Strain
In the propellanes with small cycles, such as [1.1.1]propellane or [2.2.2]propellane, the two carbons at the ends of the axial bond will be highly strained, and their bonds may even assume an inverted tetrahedral geometry.
The resulting steric strain causes such compounds to be unstable and highly reactive. The axial C-C bond is easily broken (even spontaneously) to yield less-strained bicyclic or even monocyclic hydrocarbons.
Surprisingly, the most strained member [1.1.1] is far more stable than the other small ring members ([2.1.1], [2.2.1], [2.2.2], [3.2.1], [3.1.1], and [4.1.1]).[1]
Polymerization
In principle, any propellane can be polymerized by breaking the axial C-C bond to yield a radical with two active centers, and then joining these radicals in a linear chain. For the propellanes with small cycles (such as [1.1.1], [3.2.1], or 1,3-dihydroadamantane), this process is easily achieved, yielding either simple polymers or alternating copolymers. For example, [1.1.1]propellane yields spontaneoulsy an interesting rigid polymer called staffane;[2] and [3.2.1]propellane combines spontaneously with oxygen at room temperature to give a copolymer where the bridge-opened propellane units [-C8H12-] alternate with [-O-O-] groups.[3]
Synthesis
The smaller-cycle propellanes are difficult to synthesize because of their strain. Larger membes are more easily obtained. R. Weber and J. Cook described in 1978 a general method which should yield [n.3.3]propellanes for any n greater than 2.[4]
Examples
True propellanes
- [1.1.1]propellane, C5H6 (K. Wiberg and F. Walker, 1982).[5] It is a highly strained molecule: the two central carbons have an inverted tetrahedron geometry, and each of the three cycles is the notoriously strained cyclopropane ring. The length of the central bond is only 160 pm. It is an unstable product that undergoes thermal isomerization to 3-methylenecylobutene at 114 °C,and spontaneously reacts with acetic acid to form a methylenecyclobutane ester.[2]
- [2.1.1]propellane, C6H8 (K. Wiberg, F. Walker, W. Pratt, and J. Michl, ????). This compound was detected by infrared spectroscopy at 30 K but has not been isolated as a stable molecule at room temperature (as of 2003). It is believed to polymerize above 50 K. The bonds of the shared carbons have an inverted tetrahedral geometry; the compound's strain energy was estimated as 106 kcal/mol.[6]
- [2.2.1]propellane, C7H10 (F Walker, K. Wiberg, and J. Michl, 1982). Obtained gas-phase dehalogenation with alkali metal atoms. Stable only in frozen gas matrix below 50K; oligomerizes or polymerizes at higher temperatures. The strain energy released by breaking the axial bond was estimated as 75 kcal/mol.[7]
- [3.2.1]propellane or tricyclo[3.2.1.01,5]octane, C8H12 (K. Wiberg and G. Burgmaier, 1969). Isolable. Has inverted tetrahedral geometry at the shared carbons. Estimated strain energy of 60 kcal/mol. Remarkably resistant to thermolysis; polymerizes in diphenyl ether solution with halflife of about 20 hours at 195°. It reacts spontaneously with oxygen at room temperature to give a copolymer with -O-O- bridges.[10][11][3][12][13]
- [2.2.2]propellane or tricyclo[2.2.2.01,4]octane, C8H12 (P. Eaton and G. Temme, 1973).[13][17] This propellane is unstable, too, due to the three cyclobutane-like rings and the highly distorted bond angles (three of them nearly 90°, the other three nearly 120°) at the axial carbons. Its strain energy is estimated to be 93 kcal/mol (390 kJ/mol).
- [3.3.3]propellane, C11H18 (R. Weber and J. Cook, 1978). It is a stable solid that melts at 130 °C.[4]
- [4.3.3]propellane, C12H20 (R. Weber and J. Cook, 1978). A stable solid that melts at 100–101°C.[4]
- [6.3.3]propellane, C14H24 (R. Weber and J. Cook, 1978). An oily liquid that boils at 275–277°C.[4]
- [10.3.3]propellane, C18H32 (S. Yang and J. Cook, 1976). A stable solid that sublimes at 33–34°C.[18]
Propellane derivatives
- 1,3-dehydroadamantane, C10H14 (R. Pincock and E.Torupka, 1969).[19] This compound is formally derived from adamantane by removing two hydrogens and adding an internal bond. It can be viewed as [1.3.3]propellane (whose axis would be the new bond), with an extra methylene bridge between its two larger "propeller blades". It is unstable and reactive and can be polymerized.
See also
References
- ↑ 1.0 1.1 1.2 Josef Michl, George J. Radziszewski, John W. Downing, Kenneth B. Wiberg, Frederick H. Walker, Robert D. Miller, Peter Kovacic, Mikolaj Jawdosiuk, and Vlasta Bonačić-Koutecký (1983), Highly strained single and double bonds. Pure & Appl. Chem., Vol.55, No.2, pp.315—321
- ↑ 2.0 2.1 Piotr Kaszynski and Josef Michl (1988), [n]Staffanes: a molecular-size "Tinkertoy" construction set for nanotechnology. Preparation of end-functionalized telomers and a polymer of [1.1.1]propellane J. Am. Chem. Soc.; volume 110 issus 15, pp. 5225 – 5226; doi:10.1021/ja00223a070
- ↑ 3.0 3.1 Kenneth B. Wiberg and George J. Burgmaier (1972), Tricyclo[3.2.1.01,5]octane. A 3,2,1-Propellane. Journal of the American Chemical Society, volume 94 issue 21, pp. 7396–7401 doi:10.1021/ja00776a022
- ↑ 4.0 4.1 4.2 4.3 Robert W. Weber, James M. Cook (1978), General method for the synthesis of [n.3.3]propellanes, n ≥ 3. Canadian Journal of Chemistry, volume 56, pages 189–192.
- ↑ [1.1.1]Propellane Kenneth B. Wiberg and Frederick H. Walker J. Am. Chem. Soc.; 1982; 104(19) pp 5239 – 5240; doi:10.1021/ja00383a046
- ↑ Oliver Jarosch and Günter Szeimies (2003), Thermal Behavior of [2.1.1]Propellane: A DFT/ab Initio Study. J. Org. Chem., volume 68 issue 10, pp. 3797–3801 doi:10.1021/jo020741d
- ↑ Frederick H. Walker, Kenneth B. Wiberg, Josef Michl (1982), [2.2.1]Propellane. Journal of the American Chemical Society volume 104, p. 2056.
- ↑ P.G. Gassman and G.S. Proehl (1980), ??. Journal of the American Chemical Society, volume 102, p. 6862
- ↑ K. Mlinaric-Majerski and Z. Majerski (1980), ??. Journal of the American Chemical Society, volume 102, p. 1418.
- ↑ Kenneth B. Wiberg, George J. Burgmaier (1969), Tricyclo[3.2.1.01,5]octane. Tetrahedron Letters Volume 10, Issue 5, pp. 317–319
- ↑ Paul G. Gassman, Alwin Topp, and John W. Keller (1969), Tricyclo[3.2.1.01,5]octane – a highly strained "propellerane". Tetrahedron Letters Volume 10, Issue 14 pp. 1093–1095
- ↑ D.H. Aue and R.N. Reynolds (1974), J. Org. Chem. volume 39, p. 2315
- ↑ 13.0 13.1 Kenneth B. Wiberg, William E. Pratt, William F. Bailey (1977), Reaction of 1,4-diiodonorbornane, 1,4-diiodobicyclo[2.2.2]octane, and 1,5-diiodobicyclo[3.2.1]octane with butyllithium. Convenient preparative routes to the [2.2.2]- and [3.2.1]propellanes. Journal of the American Chemical Society, volume 99, pp. 2297–2302. doi:10.1021/ja00449a045
- ↑ David P. G. Hamon, V. Craige Trenerry (1981), Carbenoid insertion reactions: formation of [4.1.1]propellane. Journal of the American Chemical Society, volume 103, pp. 4962–4965 doi:10.1021/ja00406a059
- ↑ Ursula Szeimies-Seebach, J. Harnish, Guenter Szeimies, M.V. Meerssche, G. Germain, and J.P. Declerq (1978), ?. Angewandte Chemie Int. Ed. Engl. volume 17, p. 848
- ↑ Ursula Szeimies-Seebach, Guenter Szeimies (1978), A facile route to the [4.1.1]propellane system. Journal of the American Chemical Society, volume 100, pp. 3966–3967 doi:10.1021/ja00480a072
- ↑ Philip E. Eaton and George H. Temme (1973), [2.2.2]Propellane system J. Am. Chem. Soc., volume 95 issue 22, pp. 7508–7510; doi:10.1021/ja00803a052
- ↑ (Daisy?) S. Yang, James M. Cook (1976), Reactions of dicarbonyl compounds with dimethyl β-ketoglutarate: II. Simple synthesis of compounds of the [10.3.3]- and [6.3.3]-propellane series. Journal of Organic Chemistry volume 41, issue 11, pp. 1903–1907; doi:10.1021/jo00873a004
- ↑ Richard E. Pincock and Edward J. Torupka (1969), Tetracyclo[3.3.1.13,7.01,3]decane. Highly reactive 1,3-dehydro derivative of adamantane. Journal of the American Chemical Society, volume 91 issue 16, pp. 4593–4593; doi:10.1021/ja01044a072