Procyanidin A2
| Procyanidin A2 | ||
|---|---|---|
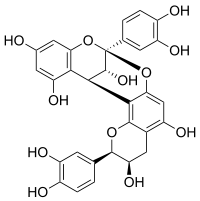 | ||
| IUPAC name (2R,3R,8S,14R,15R)-2,8-bis(3,4-dihydroxyphenyl)-2,3,4,14-tetrahydro-8,14-methanobenzo[7,8][1,3]dioxocino[4,5-h]chromene-3,5,11,13,15-pentaol | ||
| Other names Dimeric catechin | ||
| Identifiers | ||
| CAS number | 41743-41-3 | |
| PubChem | 124025 | |
| ChEBI | CHEBI:28472 | |
| Jmol-3D images | {{#if:O[C@@H]1[C@@H](C2=C(O[C@H](C3=CC(O)=C(O)C=C3[C@H](O)C4)C4=C(O)C=C2O5)C6=C(C=C(O)C=C6O)O[C@]15C7=CC(O)=C(O)C=C7|Image 1 | |
| ||
| Properties | ||
| Molecular formula | C30H24O12 | |
| Molar mass | 576.50 g mol−1 | |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | ||
| Infobox references | ||
Procyanidin A2 is a A type proanthocyanidin.
It can be found in horse chestnut (Aesculus hippocastanum),[1] in cranberry juice concentrate,[2] in peanut[2] skins[3] in Cinchona cinchona and Cinchona pubescens rind, bark and cortex, in Cinnamomum verum (Ceylon cinnamon) rind, bark and cortex, in the fruit of Persea americana (avocado or alligator pear),[4] in Urvillea ulmaceae,[5] and in Ecdysanthera utilis.[6]
Synthesis
Procyanidin B2 can be converted into procyanidin A2 by radical oxidation using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals under neutral conditions.[7]
References
- ↑ Facino, R. Maffei; Carini, M.; Brambilla, A.; Bombardelli, E.; Morazzoni, P. (1996). "Proanthocyanidin-A2: a new polyphenol". Cosmetics & Toiletries.
- ↑ 2.0 2.1 Koerner Jayma, Hsu Victor, Lee Jungmin, Kennedy, James, (2009). "Determination of Proanthocyanidin A2 Content in Phenolic Polymer Isolates by Reversed-Phase High Performance Liquid Chromatography". Journal of Chromatography A 1216 (9): 1403–1409. doi:10.1016/j.chroma.2008.12.086. PMID 19168185.
- ↑ Hongxiang Lou; Yamazaku Y.; Sasaku T.; Uchida M.; Tanaka H.; Oka S. (1999). "A-type proanthocyanidins from peanut skins". Phytochemistry 51 (2): 297–308. doi:10.1016/S0031-9422(98)00736-5.
- ↑ Proanthocyanidin-A-2 on liberherbarum.com
- ↑ Dias, Suziane A.; Cardoso (Gazio), Flávia P.; Santin, Silvana M. O.; Da Costa, Willian F.; Vidotti, Gentil J.; De Souza, Maria Conceição; Sarragiotto, Maria Helena (2009). "Free radical scavenging activity and chemical constituents ofUrvillea ulmaceae". Pharmaceutical Biology 47 (8): 717. doi:10.1080/13880200902933336.
- ↑ Lin, Lie-Chwen; Kuo, Yuh-Chi; Chou, Cheng-Jen (2002). "Immunomodulatory Proanthocyanidins fromEcdysantherautilis". Journal of Natural Products 65 (4): 505–8. doi:10.1021/np010414l. PMID 11975489.
- ↑ Conversion of procyanidin B-type (catechin dimer) to A-type: evidence for abstraction of C-2 hydrogen in catechin during radical oxidation. Kazunari Kondo, Masaaki Kurihara, Kiyoshi Fukuhara, Takashi Tanaka, Takashi Suzuki, Naoki Miyata and Masatake Toyoda, Tetrahedron Letters, 22 January 2000, Volume 41, Issue 4, Pages 485–488, doi:10.1016/S0040-4039(99)02097-3
| |||||||||||