Oxazole
From Wikipedia, the free encyclopedia
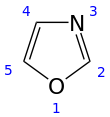
| Oxazole | ||
|---|---|---|
 |
 | |
 |
 | |
| IUPAC name 1,3-oxazole | ||
| Identifiers | ||
| CAS number | 288-42-6 | |
| PubChem | 9255 | |
| Jmol-3D images | Image 1 | |
| ||
| Properties | ||
| Molecular formula | C3H3NO | |
| Molar mass | 69.06 g mol−1 | |
| Density | 1.050 g/cm3 | |
| Boiling point | 69-70 °C | |
| Acidity (pKa) | 0.8 (of conjugate acid) [1] | |
| Supplementary data page | ||
| Structure and properties |
n, εr, etc. | |
| Thermodynamic data |
Phase behaviour Solid, liquid, gas | |
| Spectral data | UV, IR, NMR, MS | |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | ||
| Infobox references | ||
Oxazole is the parent compound for a vast class of heterocyclic aromatic organic compounds. These are azoles with an oxygen and a nitrogen separated by one carbon.[2] Oxazoles are aromatic compounds but less so than the thiazoles. Oxazole is a weak base; its conjugate acid has a pKa of 0.8, compared to 7 for imidazole.
Preparation
Classical oxazole synthetic methods in organic chemistry are
- the Robinson–Gabriel synthesis by dehydration of 2-acylaminoketones
- the Fischer oxazole synthesis from cyanohydrins and aldehydes
- the Bredereck reaction with α-haloketones and formamide
- the Van Leusen reaction with aldehydes and TosMIC
Other methods are reported in literature.
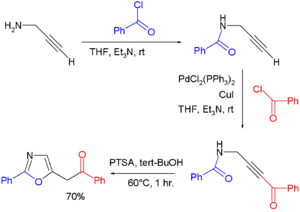
- Oxazolines can also be obtained from cycloisomerization of certain propargyl amides. In one study[3] oxazoles were prepared via a one-pot synthesis consisting of the condensation of propargyl amine and benzoyl chloride to the amide, followed by a Sonogashira coupling of the terminal alkyne end with another equivalent of benzoylchloride, and concluding with p-toluenesulfonic acid catalyzed cycloisomerization:
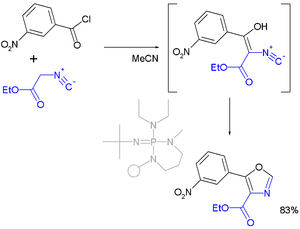
- In one reported oxazole synthesis the reactants are a nitro-substituted benzoyl chloride and an isonitrile:[4] [5]
Biosynthesis
In biomolecules, oxazoles result from the cyclization and oxidation of serine or threonine nonribosomal peptides:
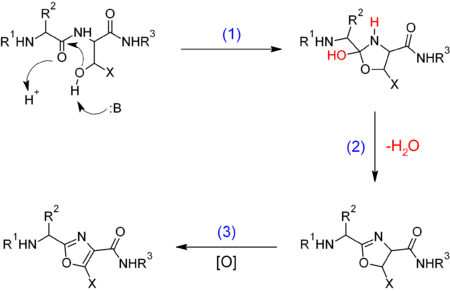 Where X = H, CH
Where X = H, CH
3 for serine and threonine respectively, B = base.
(1) Enzymatic cyclization. (2) Elimination. (3) [O] = enzymatic oxidation.
Oxazoles are not as abundant in biomolecules as the related thiazoles with oxygen replaced by a sulfur atom.
Reactions
- Deprotonation of oxazoles at C2 is often accompanied by ring-opening to the isonitrile.
- Electrophilic aromatic substitution takes place at C5 requiring activating groups.
- Nucleophilic aromatic substitution takes place with leaving groups at C2.
- Diels-Alder reactions with oxazole dienes can be followed by loss of oxygen to form pyridines.
- The Cornforth Rearrangement of 4-acyloxazoles is a thermal rearrangement reaction with the organic acyl residue and the C5 substituent changing positions.
- Various oxidation reactions. One study[6] reports on the oxidation of 4,5-diphenyloxazole with 3 equivalents of CAN to the corresponding imide and benzoic acid:
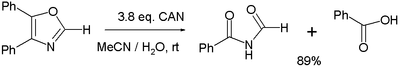
- In the balanced half-reaction three equivalents of water are consumed for each equivalent of oxazoline, generating 4 protons and 4 electrons (the latter derived from CeIV).
See also
- Isoxazole, an analog with the nitrogen atom in position 2.
- Imidazole, an analog with the oxygen replaced by a nitrogen.
- Thiazole, an analog with the oxygen replaced by a sulfur.
- Benzoxazole, where the oxazole is fused to another aromatic ring.
- Pyrrole, an analog without the oxygen atom.
- Furan, an analog without the nitrogen atom.
- Oxazoline, which has one double bond reduced.
- Oxazolidine, which has both double bonds reduced.
- Oxadiazoles with two nitrogens instead of one (e.g. furazan).
- Oxazolone, an analog with a carbonyl group
References
- ↑ Zoltewicz, J. A. & Deady, L. W. Quaternization of heteroaromatic compounds. Quantitative aspects. Adv. Heterocycl. Chem. 22, 71-121 (1978).
- ↑ Heterocyclic Chemistry TL Gilchrist, The Bath press 1985 ISBN 0-582-01421-2
- ↑ A new consecutive three-component oxazole synthesis by an amidation–coupling–cycloisomerization (ACCI) sequence Eugen Merkul and Thomas J. J. Müller Chem. Commun., 2006, 4817 - 4819, doi:10.1039/b610839c
- ↑ Fully Automated Continuous Flow Synthesis of 4,5-Disubstituted Oxazoles Marcus Baumann, Ian R. Baxendale, Steven V. Ley, Christoper D. Smith, and Geoffrey K. Tranmer Org. Lett.; 2006; 8(23) pp 5231 - 5234; (Letter) doi:10.1021/ol061975c
- ↑ They react together in the first phase in a continuous flow reactor to the intermediate enol and then in the second phase in a phosphazene base (PS-BEMP) induced cyclization by solid-phase synthesis.
- ↑ Ceric Ammonium Nitrate Promoted Oxidation of Oxazoles David A. Evans, Pavel Nagorny, and Risheng Xu Org. Lett.; 2006; 8(24) pp 5669 - 5671; (Letter) doi:10.1021/ol0624530
This article is issued from Wikipedia. The text is available under the Creative Commons Attribution/Share Alike; additional terms may apply for the media files.