Organogermanium compound

The main reason why the organogermanium is of limited synthetic value are the costs of germanium compounds. On the other hand germanium is advocated as a non-toxic alternative to many toxic organotin reagents and compounds like tetramethylgermanium and tetraethylgermanium are used in the microelectronics industry as precursors for germanium dioxide chemical vapor deposition.
The first organogermanium compound, tetraethylgermane, was synthesised by Winkler in 1887, by the reaction of germanium tetrachloride with diethylzinc.[2] The organogermanium compound bis (2-Carboxyethylgermanium)sesquioxide was first reported in 1966.[3]
Organogermanes
Organogermanes of the type R4Ge with alkyl (R) groups are accessed through the cheapest available germanium precursor germanium tetrachloride and alkyl nucleophiles. The following trends are observed going down the carbon group: The nucleophilicity increases Si<Ge<Sn as well as the hyperconjugation effect known as the beta-silicon effect Si<Ge<<Sn. The Si-C bond is mainly covalent and the Sn-C relatively polar, bonds with germanium are in between.
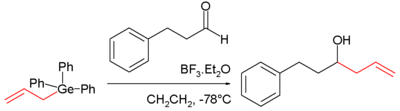
Just as with silicon the nucleophilicity of allyl germanes is high due to the intrinsic polarization of the bond (difference in electronegativity 2.55 − 2.01 = 0.54) and the combined stabilizing effect on the α-carbonion by the allyl group and the germanium atom. The germanium pendant of the Sakurai reaction was discovered in 1986:
The carbonyl group in this reaction is activated with boron trifluoride.
Germanium hydrides
Isobutylgermane (IBGe) (Me2CHCH2)GeH3 is the organogermanium hydride that is a high vapor pressure liquid germanium source for MOVPE. Isobutylgermane is currently investigated as safer and less hazardous alternative to toxic germane gas in microelectonic applications.
Tris(trimethylsilyl)germanium hydride (Me3Si)3GeH has been investigated as a non-toxic alternative to many tin hydrides such as tributyltinhydride.
Other germanium compounds
Many germanium reactive intermediates are known: germylenes (carbene pendants), germyl free radicals, germynes (carbyne pendants). Digermynnes only exist for extremely bulky substituents. Unlike alkynes, the C-Ge-Ge-C core of these Digermynnes are nonlinear, although they are planar. The Ge-Ge distance is 2.22 Å, and the Ge-Ge-C angles are 131°. Such compounds are prepared by reduction of bulky arylgermanium(II) halides.[4]
As with silicon and contrasting with carbon, compounds containing Ge=C (germenes) and Ge=Ge (digermylenes) double bonds are rare. One example is the bulky derivatives of germabenzene, an analogue of benzene.
External links
- Tetramethylgermanium Datasheet commercial supplier
- Tetraethylgermanium Datasheet commercial supplier
- Tris(trimethylsilyl)germanium hydride Datasheet commercial supplier
See also
- Compounds of carbon with other elements in the periodic table:
| CH | He | ||||||||||||||||
| CLi | CBe | CB | CC | CN | CO | CF | Ne | ||||||||||
| CNa | CMg | CAl | CSi | CP | CS | CCl | CAr | ||||||||||
| CK | CCa | CSc | CTi | CV | CCr | CMn | CFe | CCo | CNi | CCu | CZn | CGa | CGe | CAs | CSe | CBr | CKr |
| CRb | CSr | CY | CZr | CNb | CMo | CTc | CRu | CRh | CPd | CAg | CCd | CIn | CSn | CSb | CTe | CI | CXe |
| CCs | CBa | CHf | CTa | CW | CRe | COs | CIr | CPt | CAu | CHg | CTl | CPb | CBi | CPo | CAt | Rn | |
| Fr | CRa | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | |
| ↓ | |||||||||||||||||
| CLa | CCe | CPr | CNd | CPm | CSm | CEu | CGd | CTb | CDy | CHo | CEr | CTm | CYb | CLu | |||
| Ac | CTh | CPa | CU | CNp | CPu | CAm | CCm | CBk | CCf | CEs | Fm | Md | No | Lr | |||
| Core organic chemistry | Many uses in chemistry |
| Academic research, but no widespread use | Bond unknown |
References
- ↑ Main Group Metals in Organic Synthesis, Hisashi Yamamoto (Editor), Koichiro Oshima (Editor) ISBN 3-527-30508-4 2004
- ↑ Winkler, Clemens (1887). "Mittheilungen über des Germanium. Zweite Abhandlung". J. Prak. Chemie 36: 177–209. doi:10.1002/prac.18870360119. Retrieved 2008-08-20.
- ↑ "Bis(2-carboxyethylgermanium(IV) sesquioxide".
- ↑ Philip P. Power "Bonding and Reactivity of Heavier Group 14 Element Alkyne Analogues" Organometallics 2007, volume 26, pp 4362–4372. doi:10.1021/om700365p