Organochloride
  |
| Two representations of the organochloride chloroform. |
An organochloride, organochlorine, chlorocarbon, chlorinated hydrocarbon, chloroalkane, or chlorinated solvent is an organic compound containing at least one covalently bonded atom of chlorine. Their wide structural variety and divergent chemical properties lead to a broad range of names and applications. Many derivatives are controversial because of the effects of these compounds on the environment and on human and animal health.
Physical properties
Chloride substituents modify the physical properties of organic compounds in several ways. They are typically denser than water due to the presence of high atomic weight of chlorine. Chloride substituents induce stronger intermolecular interactions than hydrogen substituents. The effect is illustrated by trends in boiling points: methane (−161.6 °C), methyl chloride (−24.2 °C), dichloromethane (40 °C), chloroform (61.2 °C), and carbon tetrachloride (76.72 °C). The increased intermolecular interactions is attributed to the effects of both van der Waals and polarity.
Natural occurrence
Although rare compared to non-halogenated organic compounds, many organochlorine compounds have been isolated from natural sources ranging from bacteria to humans.[1][2] Chlorinated organic compounds are found in nearly every class of biomolecules including alkaloids, terpenes, amino acids, flavonoids, steroids, and fatty acids.[1][3] Organochlorides, including dioxins, are produced in the high temperature environment of forest fires, and dioxins have been found in the preserved ashes of lightning-ignited fires that predate synthetic dioxins.[4] In addition, a variety of simple chlorinated hydrocarbons including dichloromethane, chloroform, and carbon tetrachloride have been isolated from marine algae.[5] A majority of the chloromethane in the environment is produced naturally by biological decomposition, forest fires, and volcanoes.[6] The natural organochloride epibatidine, an alkaloid isolated from tree frogs, has potent analgesic effects and has stimulated research into new pain medication.
Preparation
From chlorine
Alkanes and arylalkanes may be chlorinated under free radical conditions, with UV light. However, the extent of chlorination is difficult to control. Aryl chlorides may be prepared by the Friedel-Crafts halogenation, using chlorine and a Lewis acid catalyst.
The haloform reaction, using chlorine and sodium hydroxide, is also able to generate alkyl halides from methyl ketones, and related compounds. Chloroform was formerly produced thus.
Chlorine adds to the multiple bonds on alkenes and alkynes as well, giving di- or tetra-chloro compounds.
Reaction with hydrogen chloride
Alkenes react with hydrogen chloride (HCl) to give alkyl chlorides. For example, the industrial production of chloroethane proceeds by the reaction of ethylene with HCl:
- H2C=CH2 + HCl → CH3CH2Cl
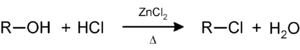
Secondary and tertiary alcohols react with the Lucas reagent (zinc chloride in concentrated hydrochloric acid) to give the corresponding alkyl halide; this reaction a method for classifying alcohols:
Other chlorinating agents
In the laboratory, alkyl chlorides are most easily prepared by reacting alcohols with thionyl chloride (SOCl2), phosphorus trichloride (PCl3), or phosphorus pentachloride (PCl5):
- ROH + SOCl2 → RCl + SO2 + HCl
- 3 ROH + PCl3 → 3 RCl + H3PO3
- ROH + PCl5 → RCl + POCl3 + HCl
In the laboratory, thionyl chloride is especially convenient, because the byproducts are gaseous.
Alternatively, the Appel reaction:
Reactions
Alkyl chlorides are versatile building blocks in organic chemistry. While alkyl bromides and iodides are more reactive, alkyl chlorides tend to be less expensive and more readily available. Alkyl chlorides readily undergo attack by nucleophiles.
Heating alkyl halides with sodium hydroxide or water gives alcohols. Reaction with alkoxides or aroxides give ethers in the Williamson ether synthesis; reaction with thiols give thioethers. Alkyl chlorides readily react with amines to give substituted amines. Alkyl chlorides are substituted by softer halides such as the iodide in the Finkelstein reaction. Reaction with other pseudohalides such as azide, cyanide, and thiocyanate are possible as well. In the presence of a strong base, alkyl chlorides undergo dehydrohalogenation to give alkenes or alkynes.
Alkyl chlorides react with magnesium to give Grignard reagents, transforming an electrophilic compound into a nucleophilic compound. The Wurtz reaction reductively couples two alkyl halides to couple with sodium.
Applications
Vinyl chloride
The largest application of organochlorine chemistry is the production of vinyl chloride. The annual production in 1985 was around 13 billion kilograms, almost all of which was converted into polyvinylchloride (PVC).
Chloromethanes
Most low molecular weight chlorinated hydrocarbons such as chloroform, dichloromethane, dichloroethene, and trichloroethane are useful solvents. These solvents tend to be relatively non-polar; they are therefore immiscible with water and effective in cleaning applications such as degreasing and dry cleaning. Several billion kilograms of chlorinated methanes are produced annually, mainly by chlorination of methane:
- CH4 + x Cl2 → CH4−xClx + x HCl
The most important is dichloromethane, which is mainly used as a solvent. Chloromethane is a precursor to chlorosilanes and silicones. Historically significant, but smaller in scale is chloroform, mainly a precursor to chlorodifluoromethane (CHClF2) and tetrafluoroethene which is used in the manufacture of Teflon.[7]
Pesticides
Many pesticides contain chlorine. Notable examples include DDT, dicofol, heptachlor, endosulfan, chlordane, aldrin, dieldrin, endrin, mirex, kepone and pentachlorophenol. These can be either hydrophilic or hydrophobic depending on their molecular structure. Many of these agents have been banned in various countries, e.g. mirex, aldrin.[8]
Insulators
Polychlorinated biphenyls (PCBs) were once commonly used electrical insulators and heat transfer agents. Their use has generally been phased out due to health concerns. PCBs were replaced by polybrominated diphenyl ethers (PBDEs), which bring similar toxicity and bioaccumulation concerns.
Toxicity
Some types of organochlorides have significant toxicity to plants or animals, including humans. Dioxins, produced when organic matter is burned in the presence of chlorine, and some insecticides, such as DDT, are persistent organic pollutants which pose dangers when they are released into the environment. For example, DDT, which was widely used to control insects in the mid 20th century, also accumulates in food chains, and causes reproductive problems (i.e., eggshell thinning) in certain bird species.[9]
When chlorinated solvents, such as carbon tetrachloride, are not disposed of properly, they accumulate in groundwater. Some highly reactive organochlorides such as phosgene have even been used as chemical warfare agents.
However, the presence of chlorine in an organic compound does not ensure toxicity. Some organochlorides are considered safe enough for consumption in foods and medicines. For example, peas and broad beans contain the natural chlorinated plant hormone 4-chloroindole-3-acetic acid (4-Cl-IAA);[10][11] and the sweetener sucralose (Splenda) is widely used in diet products. As of 2004, there were at least 165 organochlorides approved worldwide for use as pharmaceutical drugs, including the natural antibiotic vancomycin, the antihistamine loratadine (Claritin), the antidepressant sertraline (Zoloft), the anti-epileptic lamotrigine (Lamictal), and the inhalation anesthetic isoflurane.[12]
Rachel Carson brought the issue of DDT pesticide toxicity to public awareness with her 1962 book Silent Spring. While many countries have phased out the use of some types of organochlorides such as the US ban on DDT, persistent DDT, PCBs, and other organochloride residues continue to be found in humans and mammals across the planet many years after production and use have been limited. In Arctic areas, particularly high levels are found in marine mammals. These chemicals concentrate in mammals, and are even found in human breast milk. Males typically have far higher levels, as females reduce their concentration by transfer to their offspring through breast feeding.[13]
See also
- Organic halide
References
- ↑ 1.0 1.1 Gordon W. Gribble (1998). "Naturally Occurring Organohalogen Compounds". Acc. Chem. Res. 31 (3): 141–152. doi:10.1021/ar9701777.
- ↑ Gordon W. Gribble (1999). "The diversity of naturally occurring organobromine compounds". Chemical Society Reviews 28 (5): 335. doi:10.1039/a900201d.
- ↑ Kjeld C. Engvild (1986). "Chlorine-Containing Natural Compounds in Higher Plants". Phytochemistry 25 (4): 7891–791.
- ↑ Gribble, G. W. (1994). "The Natural production of chlorinated compounds". Environmental Science and Technology 28 (7): 310A–319A. doi:10.1021/es00056a001. PMID 22662801.
- ↑ Gribble, G. W. (1996). "Naturally occurring organohalogen compounds - A comprehensive survey". Progress in the Chemistry of Organic Natural Products 68 (10): 1–423. doi:10.1021/np50088a001. PMID 8795309.
- ↑ Public Health Statement - Chloromethane, Centers for Disease Control, Agency for Toxic Substances and Disease Registry
- ↑ M. Rossberg et al. "Chlorinated Hydrocarbons" in Ullmann's Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_233.pub2
- ↑ Robert L. Metcalf "Insect Control" in Ullmann's Encyclopedia of Industrial Chemistry Wiley-VCH, Wienheim, 2002. doi:10.1002/14356007.a14_263
- ↑ Connell, D. et al. (1999). Introduction to Ecotoxicology. Blackwell Science. p. 68. ISBN 0-632-03852-7.
- ↑ Pless, Tanja; Boettger, Michael; Hedden, Peter; Graebe, Jan (1984). "Occurrence of 4-Cl-indoleacetic acid in broad beans and correlation of its levels with seed development". Plant Physiology 74 (2): 320–3. doi:10.1104/pp.74.2.320. PMC 1066676. PMID 16663416.
- ↑ Magnus, Volker; Ozga, Jocelyn A; Reinecke, Dennis M; Pierson, Gerald L; Larue, Thomas A; Cohen, Jerry D; Brenner, Mark L (1997). "4-chloroindole-3-acetic and indole-3-acetic acids in Pisum sativum". Phytochemistry 46 (4): 675–681. doi:10.1016/S0031-9422(97)00229-X.
- ↑ MDL Drug Data Report (MDDR), Elsevier MDL, version 2004.2
- ↑ Marine Mammal Medicine, 2001, Dierauf & Gulland
External links
- "Formation of Chlorinated Hydrocarbons in Weathering Plant Material" article at SLAC website
- "The oxidation of chlorinated hydrocarbons" article from The Institute for Green Oxidation Chemistry at the Carnegie Mellon University website
| CH | He | ||||||||||||||||
| CLi | CBe | CB | CC | CN | CO | CF | Ne | ||||||||||
| CNa | CMg | CAl | CSi | CP | CS | CCl | CAr | ||||||||||
| CK | CCa | CSc | CTi | CV | CCr | CMn | CFe | CCo | CNi | CCu | CZn | CGa | CGe | CAs | CSe | CBr | CKr |
| CRb | CSr | CY | CZr | CNb | CMo | CTc | CRu | CRh | CPd | CAg | CCd | CIn | CSn | CSb | CTe | CI | CXe |
| CCs | CBa | CHf | CTa | CW | CRe | COs | CIr | CPt | CAu | CHg | CTl | CPb | CBi | CPo | CAt | Rn | |
| Fr | CRa | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | |
| ↓ | |||||||||||||||||
| CLa | CCe | CPr | CNd | CPm | CSm | CEu | CGd | CTb | CDy | CHo | CEr | CTm | CYb | CLu | |||
| Ac | CTh | CPa | CU | CNp | CPu | CAm | CCm | CBk | CCf | CEs | Fm | Md | No | Lr | |||
| Core organic chemistry | Many uses in chemistry |
| Academic research, but no widespread use | Bond unknown |