Orbitrap
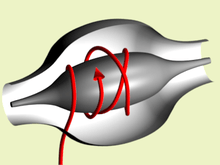
An orbitrap is a type of mass analyzer invented by Alexander Makarov. It consists of an outer barrel-like electrode and a coaxial inner spindle-like electrode that form an electrostatic field with quadro-logarithmic potential distribution.[1][2] Image current from dynamically trapped ions is detected, digitized and converted using Fourier transform into frequency and then mass spectra.
History
The orbitrap is a modification of the ion trap developed by Kingdon in the early 1920s.[3] The Kingdon trap consists of a thin central wire and an outer cylindrical electrode. A static applied voltage results in a radial logarithmic potential between the electrodes. In 1981, Knight introduced a modified outer electrode that included an axial quadrupole term that confines the ions on the trap axis.[4] Neither the Kingdon nor the Knight configurations were reported to produce mass spectra. Invention of Orbitrap analyzer and its proof-of-principle by Makarov at the end of 1990-s [1] started a sequence of technology improvements which resulted in a commercial introduction of this analyzer by Thermo Fisher Scientific as a part of hybrid LTQ Orbitrap instrument in 2005.[5][6]
Principle of operation
Trapping
In an orbitrap, ions are trapped because their electrostatic attraction to the inner electrode is balanced by centrifugal forces. Thus, ions cycle around the inner electrode on elliptical trajectories. In addition, the ions also move back and forth along the axis of the central electrode thus their trajectories in space look like helices (Fig. 1). Due to properties of quadro-logarithmic potential,[1] axial motion is harmonic, i.e. it is completely independent not only of motion around the inner electrode but also of all initial parameters of ions except their mass-to-charge ratios m/q. Its angular frequency is: ω=√(k/(m/q)) where k is the force constant of the potential, similar to the Spring Constant.
Injection
In order to inject ions from an external ion source, the field between electrodes is first reduced. As ion packets are injected tangentially into the field, the electric field is increased by ramping voltage on the inner electrode. Ions get squeezed towards the inner electrode until they reach the desired orbit inside the trap. At that moment ramping is stopped, field becomes static and detection could start. Each packet contain a multitude of ions of different velocities spread over a certain volume, therefore ions will move with different rotational frequencies but with the same axial frequency. It means that ions of a specific mass-to-charge ratio spread into rings which oscillate along the inner spindle.
Excitation
In principle, coherent axial oscillations of ion rings could be excited by applying RF waveforms to the outer electrode as demonstrated in [7] and references therein. However, if packets are injected away from the minimum of axial potential (which corresponds to the thickest part of either electrode), this automatically initiates their axial oscillations eliminating need for any additional excitation. Furthermore, absence of excitations allows to start detection process as soon as detection electronics recovers from the voltage ramp needed for ion injection.
Detection
Axial oscillations of ion rings are detected by their image current induced on the outer electrode which is split in two symmetrical pick-up sensors connected to a differential amplifier. By processing data in a manner similar to that used in Fourier transform ion cyclotron resonance mass spectrometry (FTICR-MS), the trap can be used as a mass analyzer. Like in FTICR-MS, all ions are detected simultaneously over some given period of time and resolution can be improved by increasing the strength of the field or by increasing the detection duration. Orbitrap differs from FTICR-MS by absence of magnetic field and hence significantly slower decrease of resolving power with increasing m/q.

Orbitrap Mass Spectrometry
Direct external injection
Proof-of-principle of the technology was carried out with direct injection of ions from an external laser desorption and ionization ion source.[1] This method of injection works well with pulsed sources like MALDI but cannot be interfaced to continuous ion sources like electrospray.
External injection from a trapping device
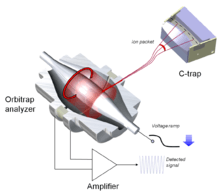
All commercial Orbitrap mass spectrometers utilize a curved linear trap for ion injection (so-called C-trap) as shown in Fig. 2. By rapidly ramping down trapping RF voltages and applying DC gradients across the C-trap, ions could be bunched into short packets similar to those from laser ion source. The C-trap is tightly integrated with the analyzer, injection optics and differential pumping.
Variants of the Orbitrap analyzer
Currently the Orbitrap analyzer exists in two variants: standard trap and compact high-field trap (Fig. 3). In practical traps, the outer electrode is sustained at virtual ground and voltage of 3.5 or 5 kV is applied to the inner electrode only. As the result, resolving power at m/z 400 and 768 ms detection time could range from 60,000 for a standard trap at 3.5 kV to 280,000 for high field trap at 5kV and with enhanced FT processing. Like in FTICR-MS the orbitrap resolving power is proportional to the number of harmonic oscillations of the ions; as a result, the resolving power is inversely proportional to the square root of m/z and proportional to acquisition time. For example, the values above would double for m/z 100 and halve for m/z 1600. For shortest transient of 96 ms these values would be reduced by 8 times while resolving power in excess of 1,000,000 was demonstrated in 3 sec transients.


Practical Orbitrap mass spectrometers
Orbitrap analyzer is known to be interfaced to a linear ion trap (LTQ Orbitrap family of instruments), quadrupole mass filter (Q Exactive family) or directly to an ion source (Exactive instrument, all marketed by Thermo Fisher Scientific). In addition, higher-energy collision cell could be appended to the C-trap, with further addition of electron-transfer dissociation on its back.[8] Most of these instruments have atmospheric pressure ion sources though intermediate-pressure MALDI source is also used (MALDI LTQ Orbitrap). All these instruments (Fig. 4) provide a high mass accuracy (<2-3 ppm with external calibrant and <1-2 ppm with internal), a high resolving power (up to 240,000 at m/z 400), a high dynamic range and high sensitivity.[5][6]
Applications of Orbitrap mass spectrometry
Orbitrap-based mass spectrometers became a workhorse in proteomics [7][9] and are also widely used in all major applications of life science mass spectrometry such as metabolism, metabolomics, environmental, food and safety analysis.[10] Most of them are interfaced to liquid chromatography separations, though they are increasingly used also with gas chromatography and ambient ionization methods.
See also
References
- ↑ 1.0 1.1 1.2 1.3 Makarov A. (2000). "Electrostatic axially harmonic orbital trapping: A high-performance technique of mass analysis". Analytical Chemistry : AC 72 (6): 1156–62. doi:10.1021/ac991131p.
- ↑ Hu Q, Noll RJ, Li H, Makarov A, Hardman M, Graham Cooks R (2005). "The Orbitrap: a new mass spectrometer". Journal of mass spectrometry : JMS 40 (4): 430–43. doi:10.1002/jms.856. PMID 15838939.
- ↑ Kingdon KH (1923). "A Method for the Neutralization of Electron Space Charge by Positive Ionization at Very Low Gas Pressures" (subscription required). Physical Review 21 (4): 408. Bibcode:1923PhRv...21..408K. doi:10.1103/PhysRev.21.408.
- ↑ Knight, R. D. (1981). "Storage of ions from laser-produced plasmas". Applied Physics Letters 38 (4): 221–223. Bibcode:1981ApPhL..38..221K. doi:10.1063/1.92315.|url=http://link.aip.org/link/?APL/38/221/1|format=|accessdate=2007-11-30
- ↑ 5.0 5.1 Makarov A, Denisov E, Kholomeev A, Balschun W, Lange O, Strupat K, Horning S (2006). "Performance evaluation of a hybrid linear ion trap/orbitrap mass spectrometer". Anal. Chem. 78 (7): 2113–20. doi:10.1021/ac0518811. PMID 16579588.
- ↑ 6.0 6.1 Makarov A, Denisov E, Lange O, Horning S (2006). "Dynamic range of mass accuracy in LTQ Orbitrap hybrid mass spectrometer". J. Am. Soc. Mass Spectrom. 17 (7): 977–82. doi:10.1016/j.jasms.2006.03.006. PMID 16750636.
- ↑ 7.0 7.1 Perry R., Cooks G., Noll R. (2008) ”Orbitrap mass spectrometry: Instrumentation, ion motion and applications”. Mass Spectrometry Reviews, 27 (6): 661–699. DOI: 10.1002/mas.20186
- ↑ McAlister G., Berggren W., Griep-Raming J., Horning S., Makarov A., Phanstiel D., Stafford G., Swaney D., Syka J., Zabrouskov V, Coon J. (2008) “Proteomics Grade Electron Transfer Dissociation-Enabled Hybrid Linear Ion Trap-Orbitrap Mass Spectrometer”, J. Proteome Res., , 7 (8), pp 3127–3136. doi: 10.1021/pr800264t
- ↑ Scigelova M, Makarov A (2006). "Orbitrap mass analyzer - overview and applications in proteomics". Proteomics. 6 Suppl 2 (S2): 16–21. doi:10.1002/pmic.200600528. PMID 17031791.
- ↑ Makarov A., Scigelova M. (2010) “Coupling liquid chromatography to Orbitrap mass spectrometry”, J. Chromatography A, 1217 (25): 3938-3945. doi:10.1016/j.chroma.2010.02.022
External links
| ||||||||||||||||||||