Molecular sensor
A molecular sensor or chemosensor is a molecule that interacts with an analyte to produce a detectable change. Molecular sensors combine molecular recognition with some form of reporter so the presence of the guest can be observed.[1] The term supramolecular analytical chemistry has recently been coined to describe the application of molecular sensors to analytical chemistry.[2]
Early examples of molecular sensors are crown ethers with large affinity for sodium ions but not for potassium and forms of metal detection by so-called complexones which are traditional pH indicators retrofitted with molecular groups sensitive to metals. This receptor-spacer-reporter concept is a recurring theme often with the reporter displaying photoinduced electron transfer.[3] One example is a sensor sensitive to heparin.
 |  |
| ||
| Heparin binding | Tannic acid binding | Saxitoxin binding | ||
Other receptors are sensitive not to a specific molecule but to a molecular compound class. One example is the grouped analysis of several tannic acids that accumulate in ageing Scotch whisky in oak barrels. The grouped results demonstrated a correlation with the age but the individual components did not. A similar receptor can be used to analyze tartrates in wine.
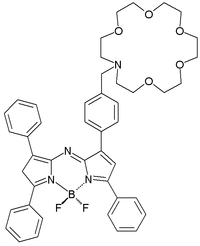
The compound saxitoxin is a neurotoxin found in shellfish and a chemical weapon. An experimental sensor for this compound is again based on PET. Interaction of saxitoxin with the sensor's crown ether moiety kills its PET process towards the fluorophore and fluorescence is switched from off to on.[4] The unusual boron moiety makes sure the fluorescence takes place in the visible light part of the electromagnetic spectrum
In another strategy called indicator-displacement assay (IDA) an analyte such as citrate or phosphate ions displace a fluorescent indicator in an indicator-host complex. The so-called UT taste chip (University of Texas) is a prototype electronic tongue and combines supramolecular chemistry with charge-coupled devices based on silicon wafers and immobilized receptor molecules.
See also
- Molecular recognition
- Boronic acids in supramolecular chemistry: Saccharide recognition
- Host-guest chemistry
- Molecular machine
References
- ↑ Luminescent molecular sensors based on analyte coordination to transition-metal complexes Cerrie W. Rogers and Michael O. Wolf Coordination Chemistry Reviews; 2002; 233-24 pp 341 - 350; (Review) doi:10.1016/S0010-8545(02)00023-1
- ↑ Supramolecular Analytical Chemistry Eric V. Anslyn J. Org. Chem.; 2007; 72(3) pp 687 - 699; (Perspective) doi:10.1021/jo0617971
- ↑ Design principles of fluorescent molecular sensors for cation recognition Bernard Valeur and Isabelle Leray Coordination Chemistry Reviews; 2000; 205 pp 3 - 40; (Review) doi:10.1016/S0010-8545(00)00246-0
- ↑ Visible Fluorescence Chemosensor for Saxitoxin Robert E. Gawley, Hua Mao, M. Mahbubul Haque, John B. Thorne, and Jennifer S. Pharr J. Org. Chem.; 2007; 72(6) pp 2187 - 2191; (Article) doi:10.1021/jo062506r