Mitochondrial carrier
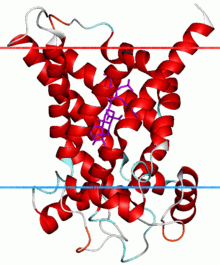 Mitochondrial ADP/ATP carrier | |||||||||
| Identifiers | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Symbol | Mito_carr | ||||||||
| Pfam | PF00153 | ||||||||
| InterPro | IPR001993 | ||||||||
| PROSITE | PDOC00189 | ||||||||
| SCOP | 1okc | ||||||||
| SUPERFAMILY | 1okc | ||||||||
| TCDB | 2.A.29 | ||||||||
| OPM superfamily | 21 | ||||||||
| OPM protein | 1okc | ||||||||
| |||||||||
Mitochondrial carriers are proteins from the solute carrier family which transfers molecules across the membranes of the mitochondria.[1]
Function
A variety of substrate carrier proteins, which are involved in energy transfer, have been found in the inner membranes of mitochondria and other eukaryotic organelles such as the peroxisome.[2][3][4][5][6][7] Such proteins include: ADP, ATP carrier protein (ADP/ATP translocase); 2-oxoglutarate/malate carrier protein; phosphate carrier protein; tricarboxylate transport protein (or citrate transport protein); Graves disease carrier protein; yeast mitochondrial proteins MRS3 and MRS4; yeast mitochondrial FAD carrier protein; and many others.
Structure
All mitochondrial carriers are encoded by nuclear genes. Most contain a primary structure exhibiting regions of 100 homologous amino acid repeats, the N and C termini face the intermembrane space and there are six definable transmembrane segments in each carrier. All carriers also contain a common sequence, referred to as the MCF motif, in each repeated region, with some variation in one or two signature sequences.[1]
Amongst the members of the mitochondrial carrier family that have been identified, it is the ADP/ATP carrier (AAC) that is responsible for importing ADP into the mitochondria and exporting ATP out of the mitochondria and into the cytosol following synthesis.[8] The AAC is an integral membrane protein that is synthesised lacking a cleavable presequence, but instead contains internal targeting information.[9] It forms a dimer of two identical subunits[10] and consists of a basket shaped structure with six transmembrane helices that are tilted with respect to the membrane, 3 of them kinked at the level of proline residues.[1]
Substrates
Examples of transported compounds include:
- citrate - SLC25A1
- ornithine - SLC25A2, SLC25A15
- phosphate - SLC25A3, SLC25A23, SLC25A24, SLC25A25
- adenine nucleotide - SLC25A4, SLC25A5, SLC25A6, SLC25A31
- dicarboxylate - SLC25A10
- oxoglutarate - SLC25A11
- glutamate - SLC25A22
Examples
Human proteins containing this domain include:
- HDMCP, LOC153328, MCART1, MCART2, MCART6, MTCH1, MTCH2
- UCP1, UCP2, UCP3
- SLC25A1, SLC25A3, SLC25A4, SLC25A5, SLC25A6, SLC25A10, SLC25A11, SLC25A12, SLC25A13, SLC25A14, SLC25A16, SLC25A17, SLC25A18, SLC25A19, SLC25A21, SLC25A22, SLC25A23, SLC25A24, SLC25A25, SLC25A26, SLC25A27, SLC25A28, SLC25A29, SLC25A30, SLC25A31, SLC25A32, SLC25A33, SLC25A34, SLC25A35, SLC25A36, SLC25A37, SLC25A38, SLC25A39, SLC25A40, SLC25A41, SLC25A42, SLC25A43, SLC25A44, SLC25A45, SLC25A46
External links
References
- ↑ 1.0 1.1 1.2 Nury H, Dahout-Gonzalez C, Trézéguet V, Lauquin GJ, Brandolin G, Pebay-Peyroula E (2006). "Relations between structure and function of the mitochondrial ADP/ATP carrier". Annu. Rev. Biochem. 75: 713–41. doi:10.1146/annurev.biochem.75.103004.142747. PMID 16756509.
- ↑ Klingenberg M (1990). "Mechanism and evolution of the uncoupling protein of brown adipose tissue". Trends Biochem. Sci. 15 (3): 108–112. doi:10.1016/0968-0004(90)90194-G. PMID 2158156.
- ↑ Walker JE (1992). "The mitochondrial transporter family". Curr. Opin. Struct. Biol. 2 (4): 519–526. doi:10.1016/0959-440X(92)90081-H.
- ↑ Kuan J, Saier Jr MH (1993). "Expansion of the mitochondrial carrier family". Res. Microbiol. 144 (8): 671–672. doi:10.1016/0923-2508(93)90073-B. PMID 8140286.
- ↑ Lawson JE, Nelson DR, Klingenberg M, Douglas MG (1993). "Site-directed mutagenesis of the yeast mitochondrial ADP/ATP translocator. Six arginines and one lysine are essential". J. Mol. Biol. 230 (4): 1159–1170. doi:10.1006/jmbi.1993.1233. PMID 8487299.
- ↑ Palmieri F (1994). "Mitochondrial carrier proteins". FEBS Lett. 346 (1): 48–54. doi:10.1016/0014-5793(94)00329-7. PMID 8206158.
- ↑ Jank B, Schweyen RJ, Link TA, Habermann B (1993). "PMP47, a peroxisomal homologue of mitochondrial solute carrier proteins". Trends Biochem. Sci. 18 (11): 427–428. PMID 8291088.
- ↑ Endres M, Neupert W, Brunner M (June 1999). "Transport of the ADP/ATP carrier of mitochondria from the TOM complex to the TIM22.54 complex". EMBO J. 18 (12): 3214–21. doi:10.1093/emboj/18.12.3214. PMC 1171402. PMID 10369662.
- ↑ Ryan MT, Müller H, Pfanner N (July 1999). "Functional staging of ADP/ATP carrier translocation across the outer mitochondrial membrane". J. Biol. Chem. 274 (29): 20619–27. doi:10.1074/jbc.274.29.20619. PMID 10400693.
- ↑ Falconi M, Chillemi G, Di Marino D, D'Annessa I, Morozzo della Rocca B, Palmieri L, Desideri A (November 2006). "Structural dynamics of the mitochondrial ADP/ATP carrier revealed by molecular dynamics simulation studies". Proteins 65 (3): 681–91. doi:10.1002/prot.21102. PMID 16988954.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||