Methylenecyclopropane
| Methylenecyclopropane | |
|---|---|
 | |
| IUPAC name methylenecyclopropane | |
| Identifiers | |
| CAS number | 6142-73-0 |
| PubChem | 80245 |
| ChemSpider | 72487 |
| Jmol-3D images | Image 1 |
| |
| |
| Properties | |
| Molecular formula | C4H6 |
| Molar mass | 54.09 |
| Density | 0.8 g/cm3 |
| Boiling point | 9-12 °C |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
Methylenecyclopropane is an organic compound with the formula (CH2)2CCH2. It is a colourless easily condensed gas that is used as a reagent in organic synthesis.
Synthesis
Methylenecyclopropane can be synthesised via an intramolecular cyclisation reaction, using β-halo alkenes and a strong base such as sodium amide.[1]
Reactions
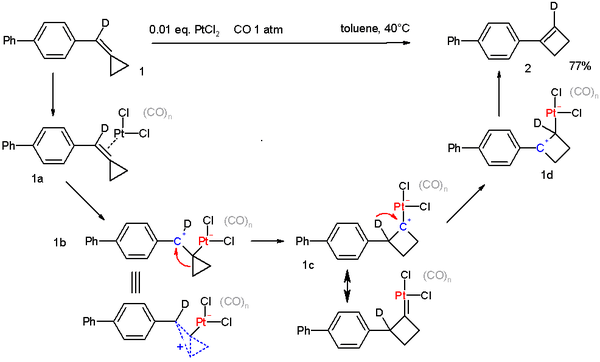
Being a strained and unsaturated molecule methylenecyclopropane undergoes many reactions, especially in the presence of metal catalysts.[2] For example methylenecyclopropanes can be converted to cyclobutenes in the presence of a Platinum catalyst.[3] This can be considered similar to the ring expansion seen in vinylcyclopropane rearrangements
Substituted methylenecyclopropanes can also be involved in trimethylenemethane cycloaddition reactions.
See also
References
- ↑ Salaun, J. R.; Champion, J.; Conia, J. M. (1977), "Cyclobutanone from methylenecyclopropane via oxaspiropentane", Org. Synth. 57: 36; Coll. Vol. 6: 320
- ↑ Nakamura, I.; Yamamoto, Y. (2002). "Transition Metal-Catalyzed Reactions of Methylenecyclopropanes". Advanced Synthesis and Catalysis 344 (2): 111–129. doi:10.1002/1615-4169(200202)344:2<111::AID-ADSC111>3.0.CO;2-0.
- ↑ PtCl2-Catalyzed Rearrangement of Methylenecyclopropanes Alois Fürstner and Christophe Aïssa J. Am. Chem. Soc.; 2006; 128(19) pp 6306 -6307; Abstract